What Is Cytokine MS Quantification?
Cytokines are small signaling proteins that mediate intercellular communication in the immune system. Their precise quantification is essential for understanding inflammatory responses, immune regulation, and disease mechanisms in immuno-oncology, autoimmune disorders, infectious diseases, and vaccine development.
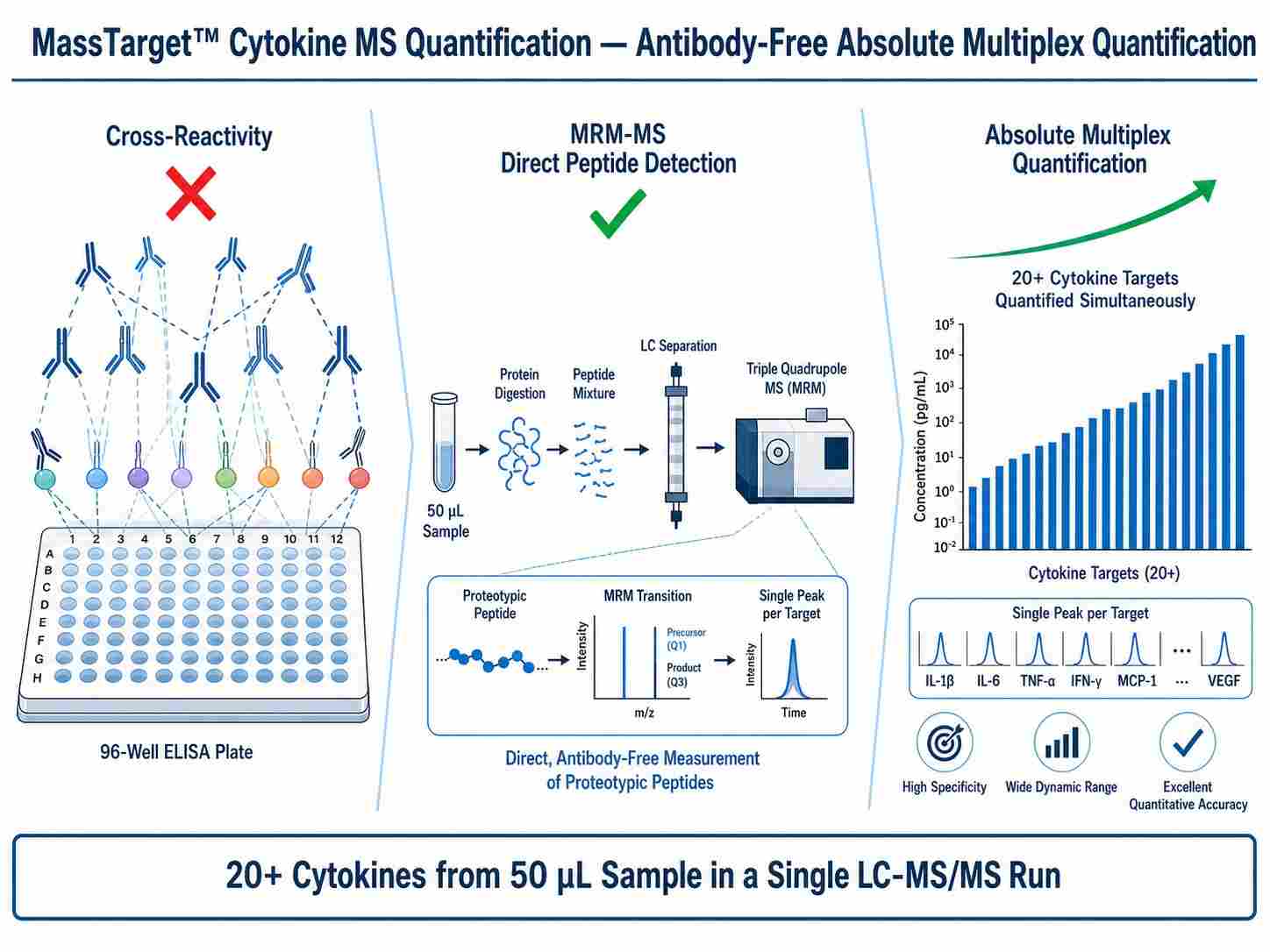
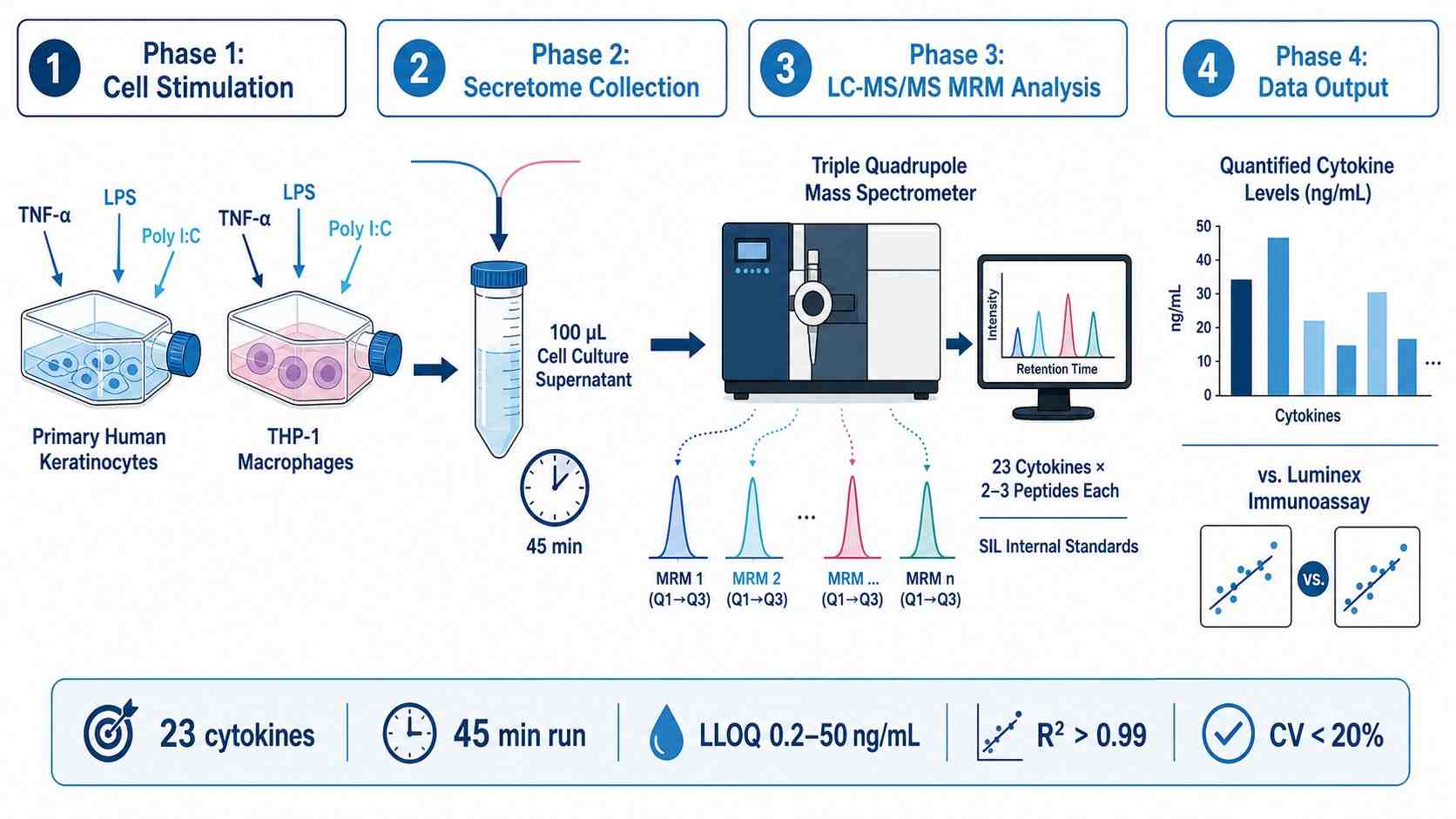
MassTarget™ Cytokine MS Quantification uses liquid chromatography-tandem mass spectrometry (LC-MS/MS) in multiple reaction monitoring (MRM) mode to achieve antibody-free, absolute quantification of cytokines from biological matrices. Unlike traditional immunoassays that rely on antibody binding, MRM-MS directly measures signature peptides derived from each cytokine after proteolytic digestion, using stable isotope-labeled (SIL) peptide standards for absolute quantification.
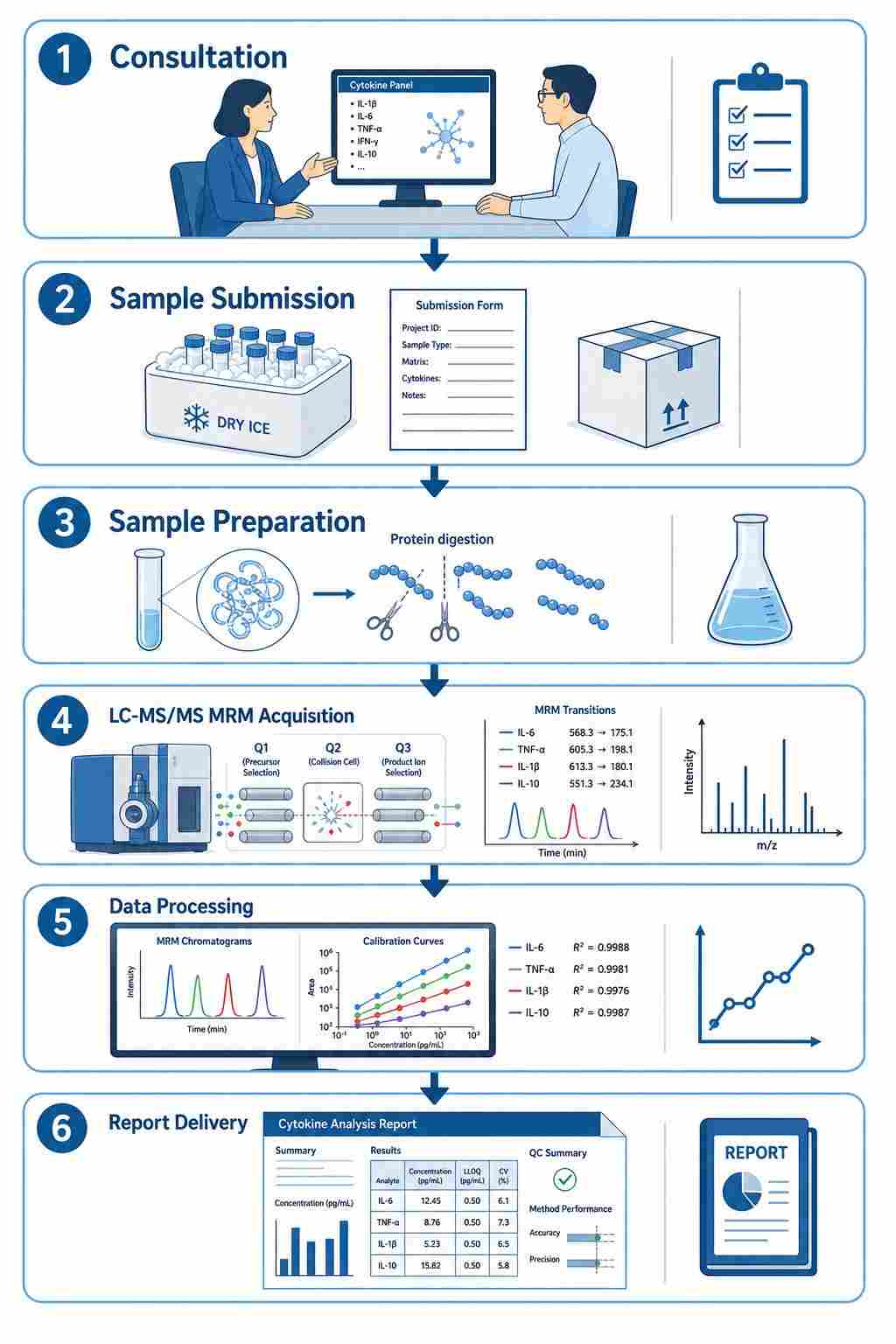
The core workflow involves:
- Sample preparation — Protein extraction and digestion from serum, plasma, cell culture supernatant, or tissue lysate
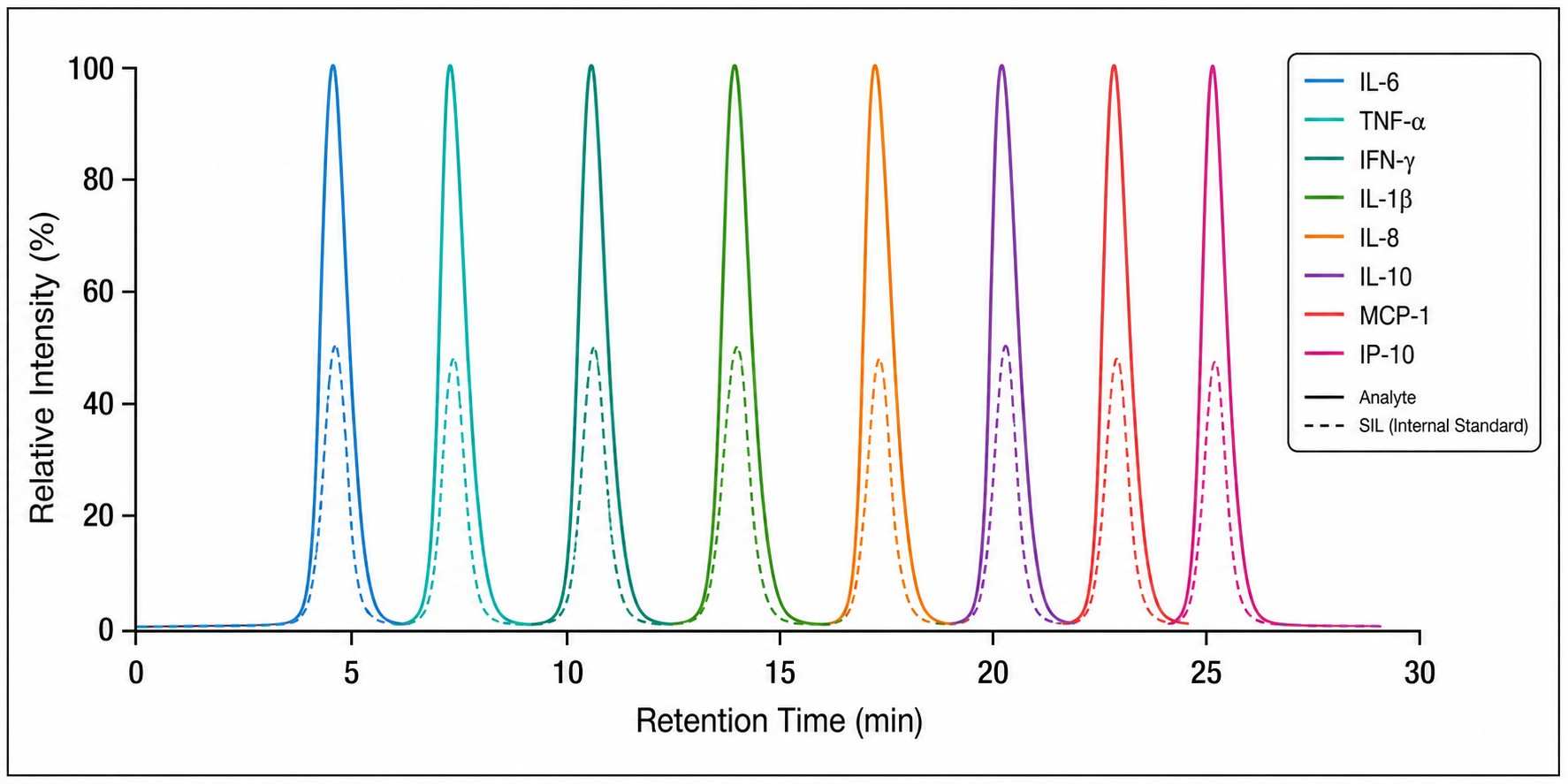
- LC-MS/MS MRM acquisition — Targeted detection of signature peptides for each cytokine using scheduled MRM transitions
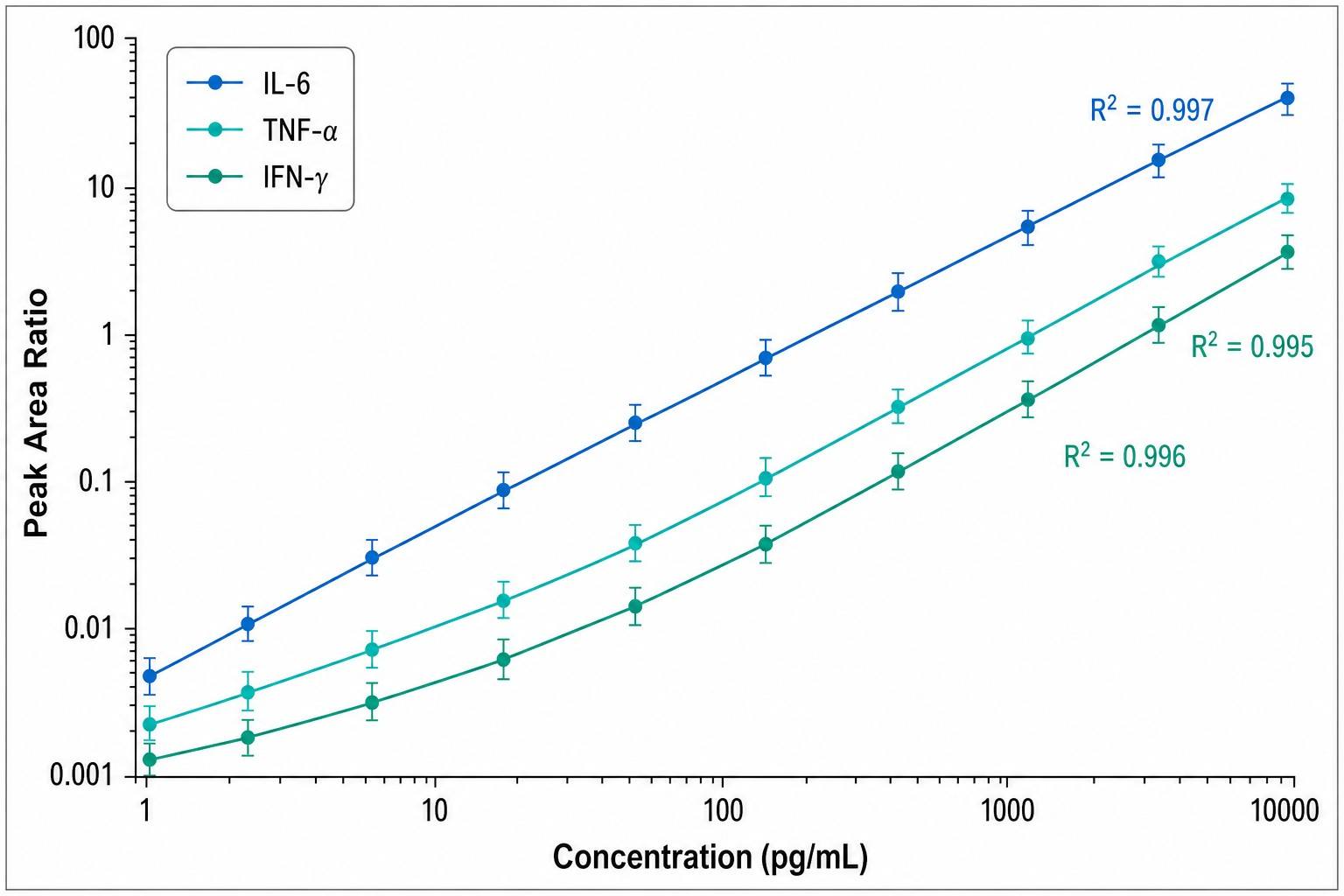
- Absolute quantification — Comparison of endogenous peptide signal against SIL internal standard calibration curves
- Data processing — Peak integration, quality control, and concentration reporting in ng/mL
This approach delivers several fundamental advantages: it eliminates antibody cross-reactivity, provides true absolute concentrations rather than relative fluorescence units, enables multiplexing of 20+ cytokines in a single injection, and allows isoform-specific quantification that antibody-based methods cannot achieve.
Learn more about LC-MS MRM quantification →