The Creative Proteomics Advantage: Solving the "Sticky" Problem
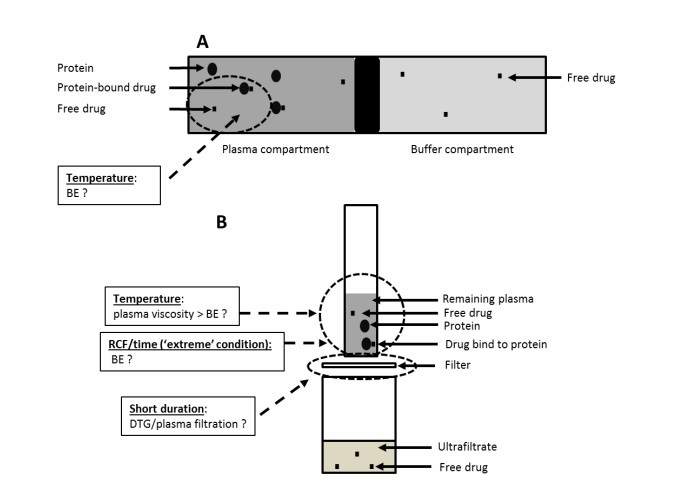
Every drug discovery team eventually encounters the "sticky" compound problem. As medicinal chemists design more complex, highly lipophilic (fat-soluble) molecules to hit challenging targets, these compounds tend to stick to everything—plastic tubes, assay plates, and filtration membranes. In standard assays, this Non-Specific Binding (NSB) leads to artificially low recovery rates, skewed binding data, and ultimately, incorrect dosing predictions. When hydrophobic interactions dominate, standard screening methods simply cannot be trusted.
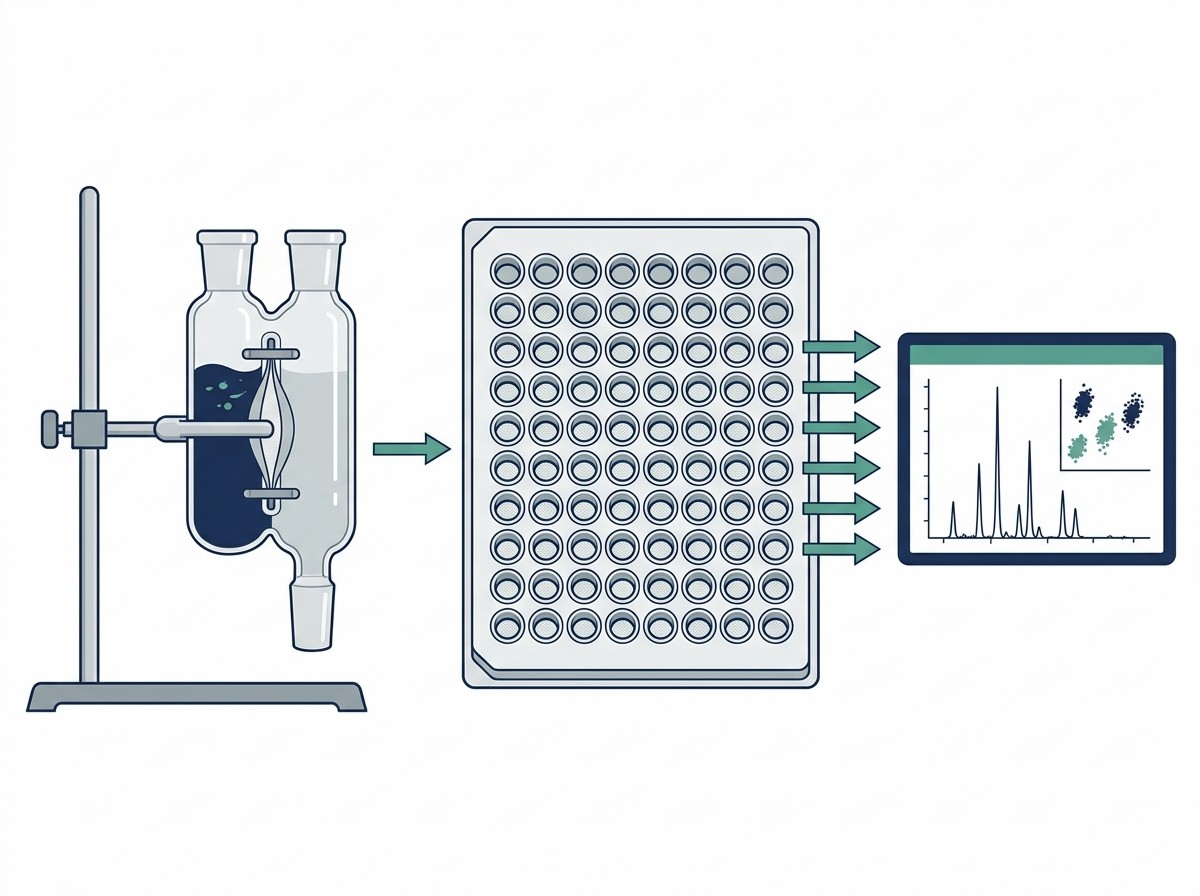
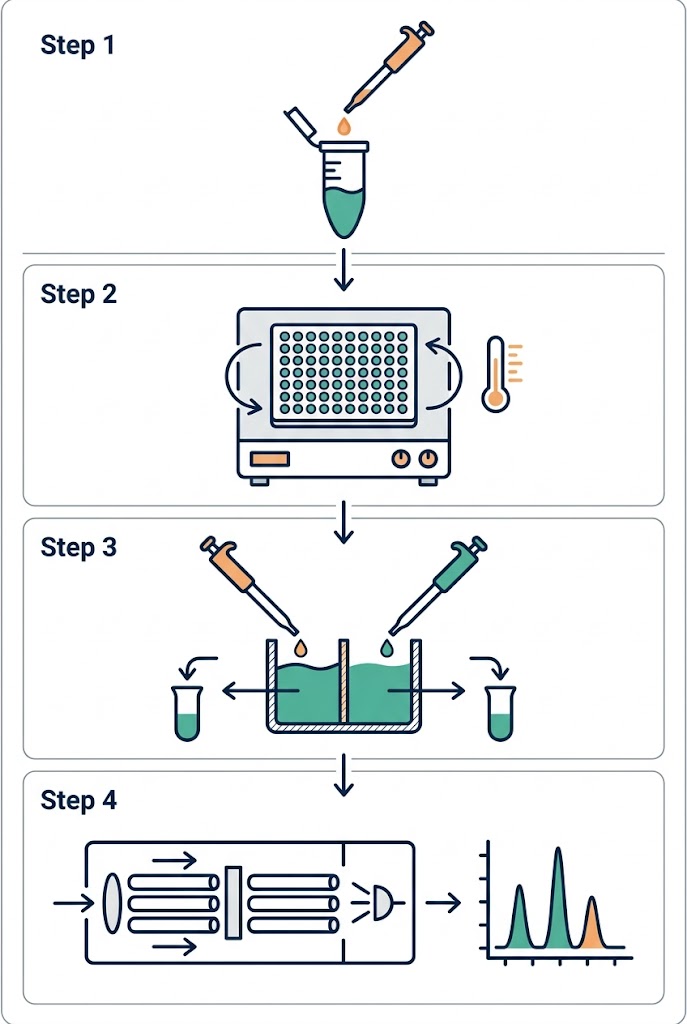
We understand how frustrating it is to synthesize a promising analog, only to have the data ruined by poor assay methodology. At Creative Proteomics, our ADME and pharmacokinetics team specializes in rescuing these difficult projects. We utilize the industry-leading Rapid Equilibrium Dialysis (RED) format, utilizing 96-well plates equipped with specialized low-binding regenerated cellulose membranes. This high-throughput setup minimizes the surface area-to-volume ratio where drugs can get trapped, significantly reducing NSB risks while maximizing sample recovery. By closely monitoring the thermodynamics of the system, we ensure that your compound remains in the solution phase where it belongs.
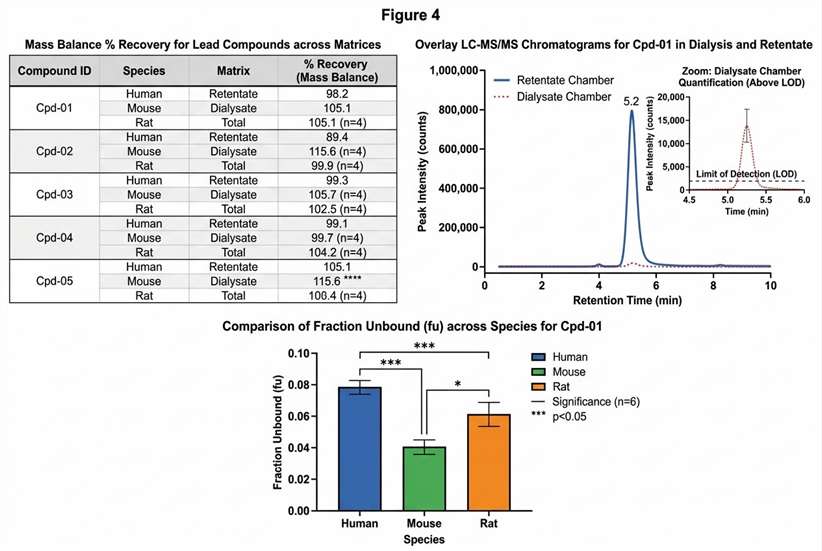
Furthermore, accurately calculating the fu of a drug that is >99% bound requires extreme analytical sensitivity, because the amount of free drug floating in the buffer chamber is minuscule. Our platform leverages state-of-the-art QQQ and Q-TOF mass spectrometers, allowing us to accurately detect and quantify trace levels of your compound that other laboratories might miss entirely. If your project is still in the earliest stages of hit identification and you need to screen thousands of compounds before optimizing for ADME properties, you can also explore our high-throughput Affinity Selection Mass Spectrometry (ASMS) solutions to rapidly filter your initial compound libraries.