Starch Content Analysis
Starch is the most important poly-glucide and the main source of digestible car-bohydrate in the human diet. It is formed from α-D-gluco-pyranose with a high polymerization degree of thousands order. The importance of starch for the food industry makes it necessary to develop easy, fast, economic and accurate methodologies for its quantification. In some cases, determination of starch content is not made directly. It is calculated as the difference between the dry matter and all the other components (e.g. proteins, lipids, sugars, fibers and minerals).
The starch content in plants varies from 14% to 75 % depending on the plant species. Several chemical and physical methods have been developed to estimate starch contents, such as headspace gas chromatographic method, differential thermal analysis, and determination of the solution poalrimetric deviation. However, the most efficient and easiest method is the enzymatic methods. This method is based on a complete enzymatic conversion of the starch into glucose, followed by an enzymatic determination of the glucose. Gelatinized starch is partly hydrolyzed with thermostable α-amylase. A complete degradation of starch oligomers into glucose is performed using amyloglucosidase. The released amount of glucose is determined quantitatively by an enzymatic method using glucose oxidase. The enzymatic hydrolysis and glucose colorimetric determination allows the quantification of starch content.
Starch is a nutrient stored in plant bodies, primarily found in seeds and tubers. Starch content varies among different plant species, and it is generally abundant in various types of plants. When determining the total starch content using conventional dual-wavelength or sulfuric acid methods, stability and accuracy are often compromised. Additionally, biological samples typically contain a substantial amount of soluble sugars, which can interfere with the accurate measurement of starch content.
Creative Proteomics employs a method that involves the removal of soluble sugars from the sample, followed by enzymatic hydrolysis and colorimetric techniques to measure the starch content. This approach ensures high accuracy and stability in the results obtained.
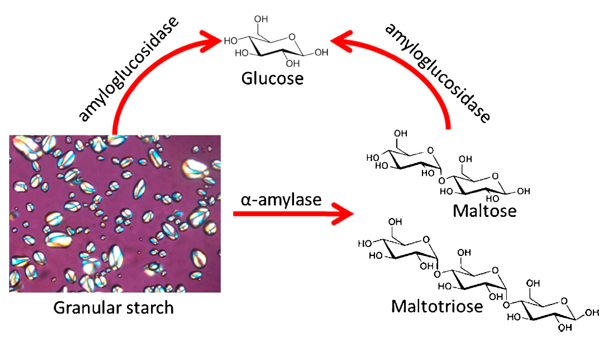 Figure 1. Schematic showing a reaction scheme of alpha-amylase and amyloglucosidase on starch [2]
Figure 1. Schematic showing a reaction scheme of alpha-amylase and amyloglucosidase on starch [2]
Amylose Content Analysis
Starch is formed by α-glucose binding chains and is comprised of two glucose polymers, amylopectin, a large branched molecule comprising chains of α-(1→4) linked anhydroglucose resides linked by α-(1→6) branch points, and amylose, an essentially linearpolymer made of α-D-glucose units, bonded to each other through α-(1→4) glycosidic bonds. Amylose chain can form helical coils which traps iodine and turn blue.
Starch from different sources contains different amount of amylose and amylopectin, which generally affects physicochemical properties of starch such as paste viscosity, gelatinization, and water absorption. For those reasons, the amylose content must be measured accurately.
Starch-iodine colorimetry is a simple and rapid method for the measurement of amylose content. Starch-iodine binding leads to a blue solution that mainly depends on the amylose/amylopectin ratio in starch. Amylose in starch is responsible for the formation of a deep blue color in the presence of iodine. This amylose-iodide complex has a distinct blue-black coloration, as opposed to the orange-red color of iodine. This method is rapid, simple, accurate and does not require the use of multi-component analysis of spectra. It has been widely used to determine amylose content.
Technical Advantages
- Enzymatic hydrolysis combined with GOPOD colorimetry, ensuring high stability with an RSD (Relative Standard Deviation) of less than 5% for samples in the same batch.
- Elimination of the interference from soluble sugars, resulting in more reliable outcomes.
- High throughput, capable of simultaneously analyzing up to 96 samples.
- Applicable for both whole powder samples and starch samples.
Sample Requirements:
Sample Types: Fresh or dried samples are both acceptable.
Biological Replicates: It is recommended to perform three biological replicates.
Sample Quantity: For fresh samples, more than 2 grams are required, and for dried samples, more than 1 gram is needed.
Sample Preparation
Before sampling, establish control and experimental groups with a recommendation of at least three biological replicates for each group.
Sampling should be done rapidly, aiming to maintain consistency in timing.
Excessive pre-treatment is generally discouraged, and it is preferred for our company to handle most of the pre-treatment steps. However, customers should ensure that liquid samples are securely packaged, avoid using glass containers, prepare suitable buffers, protect against vibrations and impact, and prevent sample leakage during shipping. For solid dried samples, minimal or preliminary grinding may be necessary. For solid fresh samples, no special treatment is needed when shipping at room temperature with ice packs.
When shipping with dry ice, samples must be cut into small pieces of less than 1 cm³.
Applications
Crop nutritional quality research
Crop breeding research
Food processing research
Polymer material research
Starch synthesis and metabolism research
Plant stress resistance research
Plant growth and development research
Technical Workflow

As one of the leading companies in the omics field with over years of experience in omics study, Creative Proteomics provides glycomics analysis service customized to your needs. Contact us to discuss your project.
How to place an order:

*If your organization requires signing of a confidentiality agreement, please contact us by email.
References
- DEMIATE, Ivo Motim, KONKEL, Francisco Enéias, & PEDROSO, Ricardo Alexandre. (2001). ENZYMATIC DETERMINATION OF STARCH IN DOCE DE LEITE USING DIALYSIS. Food Science and Technology, Vol21(3), 339-342.
- Frederick J. Warren, Bin Zhang, Gina Waltzer, Michael J. Gidley, Sushil Dhital (2015) The interplay of α-amylase and amyloglucosidase activities on the digestion of starch in in vitro enzymic systems. Carbohydrate Polymers, Vol117(6), 192-200.
- McGrance, S.J., Cornell, H.J. and Rix, C.J. (1998), A Simple and Rapid Colorimetric Method for the Determination of Amylose in Starch Products. Starch/Stärke, 50: 158-163.




