Native MS Fragment Screening Service
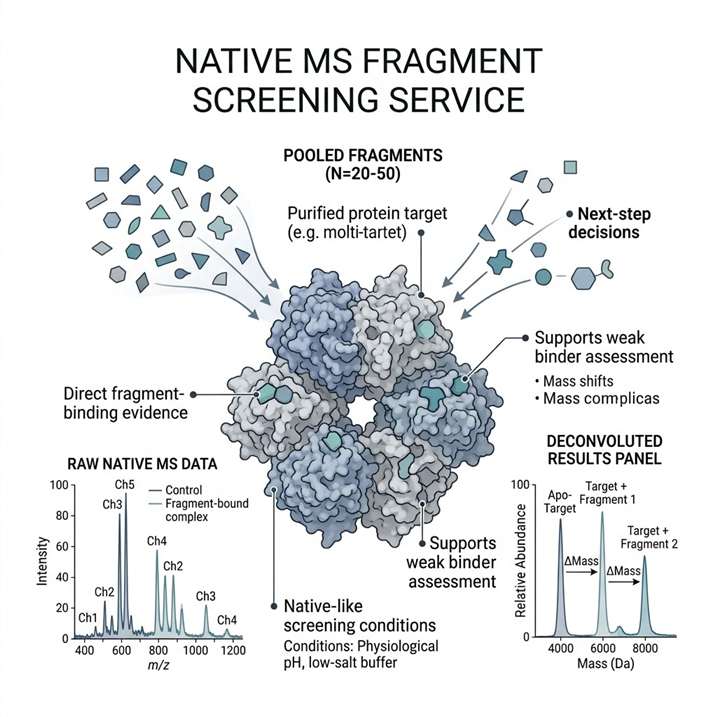
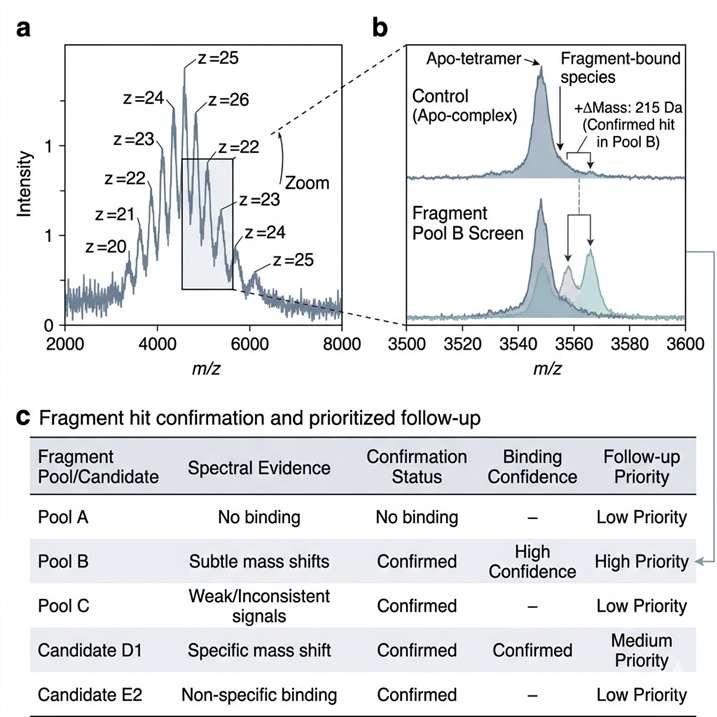
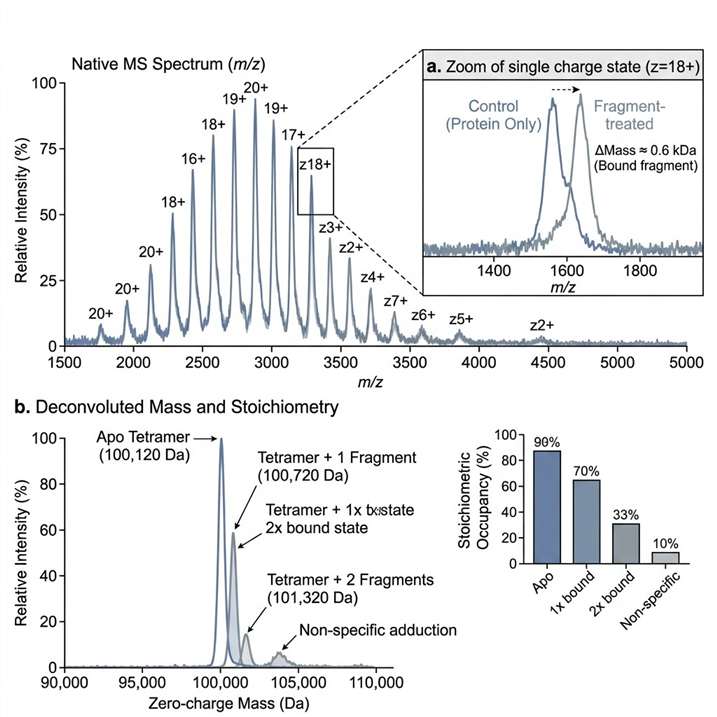
Direct fragment-binding evidence under native-like conditions.
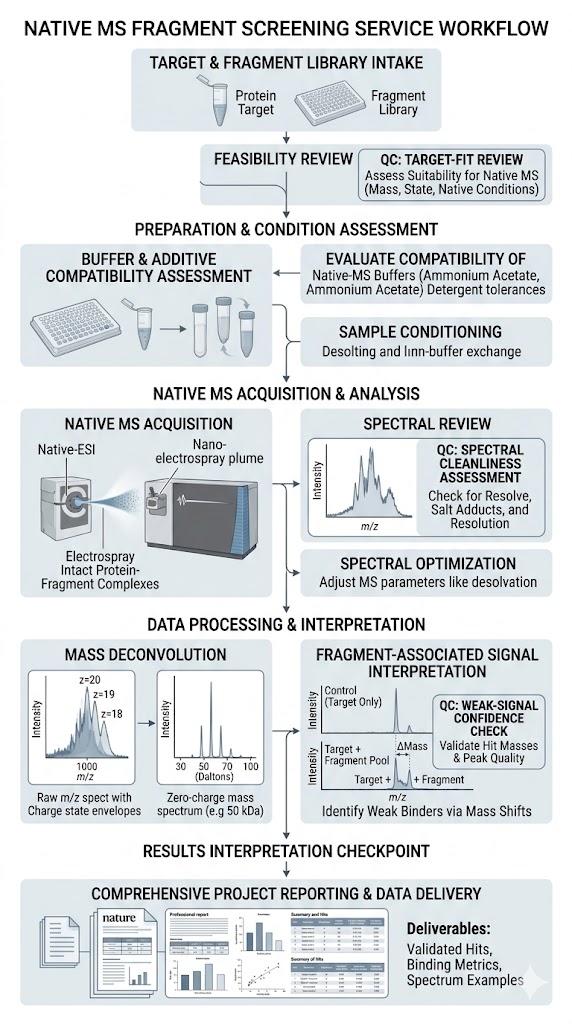
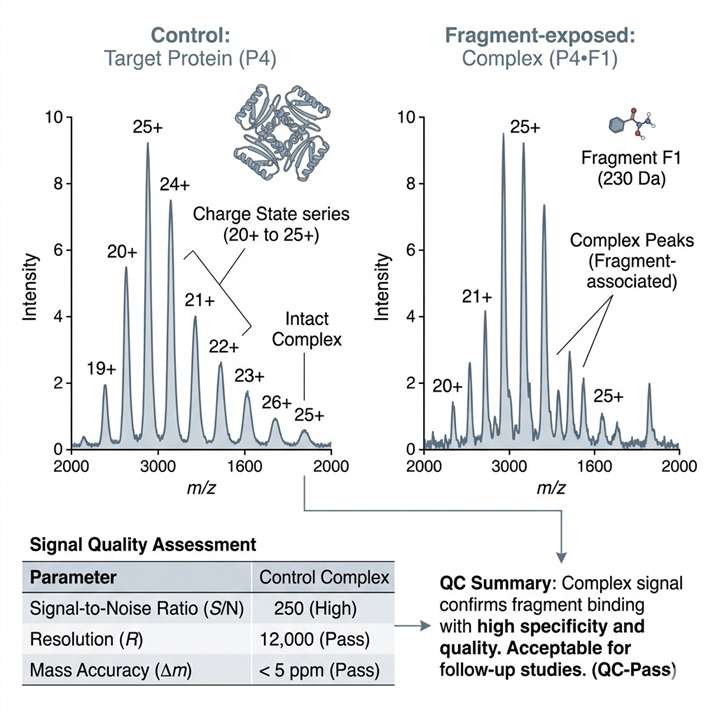
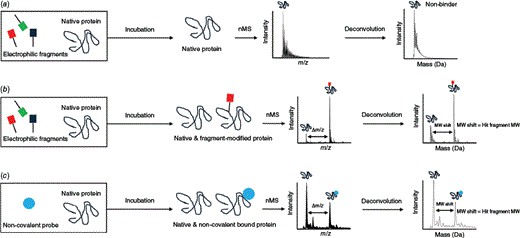
Native MS fragment screening helps us detect fragment binding under native-like conditions and turn spectral evidence into clear next-step decisions. We use this service to support hit triage, weak binder assessment, target-fit review, and orthogonal follow-up planning in early discovery programs.
Key Advantages:
- Direct fragment-binding evidence.
- Supports weak binder assessment.
- Native-like screening conditions.
- Useful for target-fit review.
- Built for triage decisions.