Background
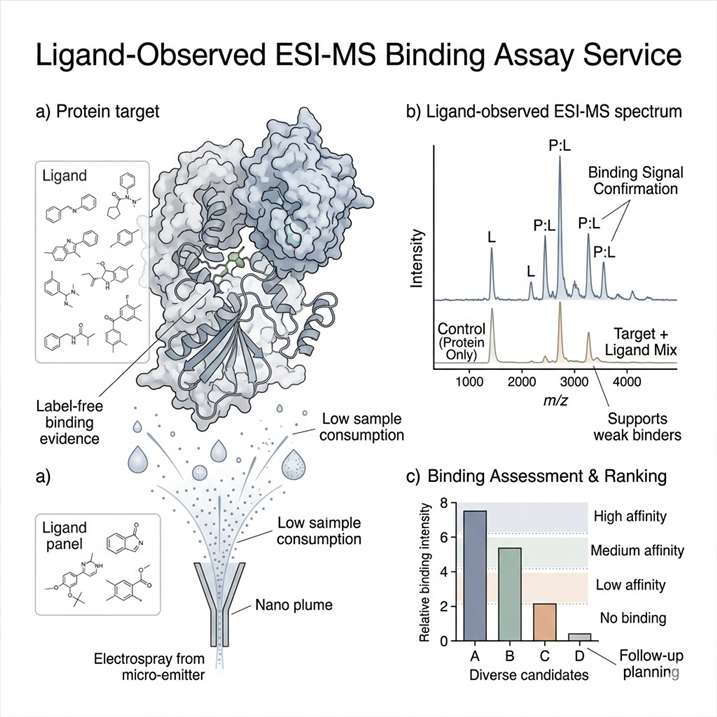
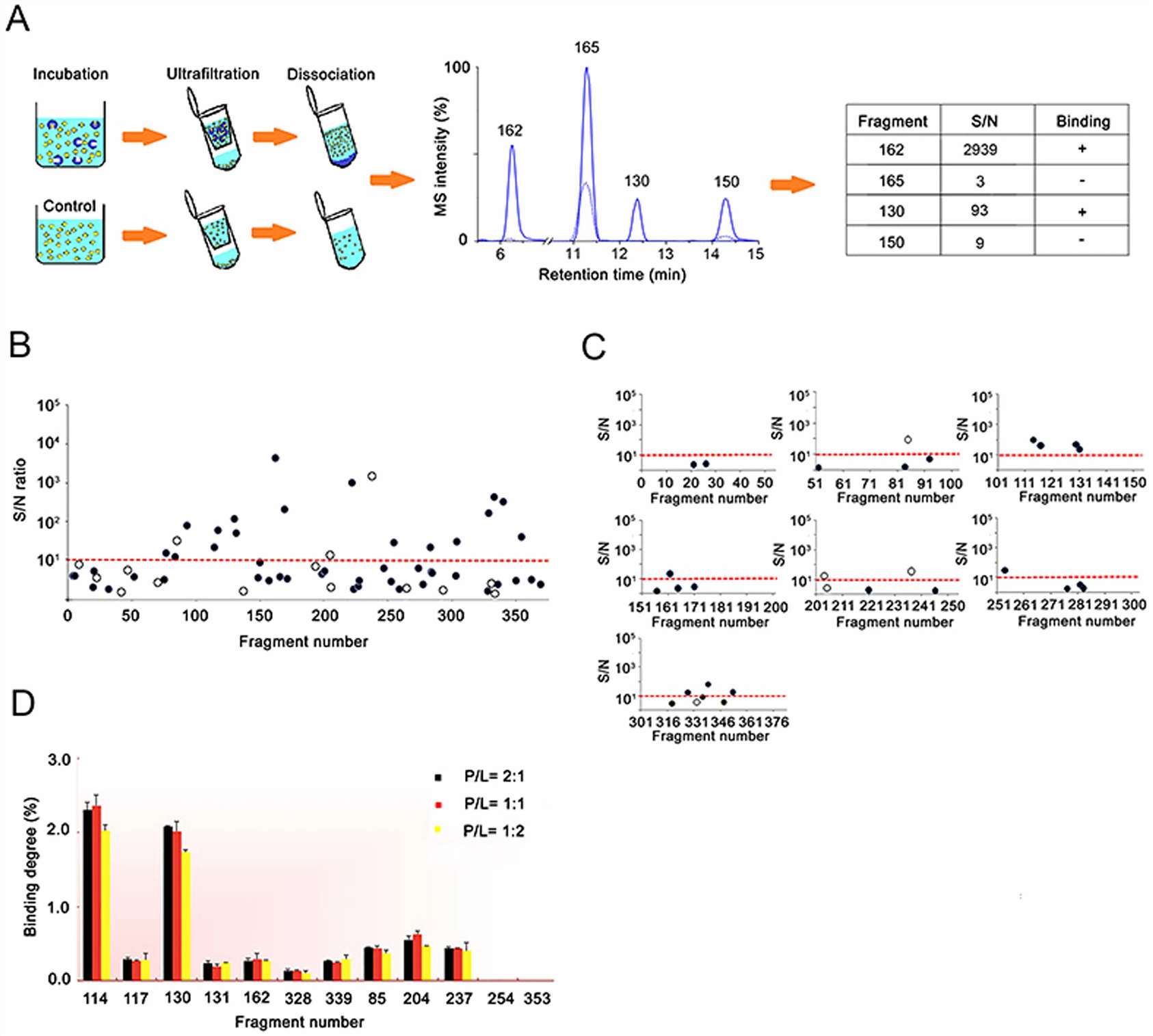
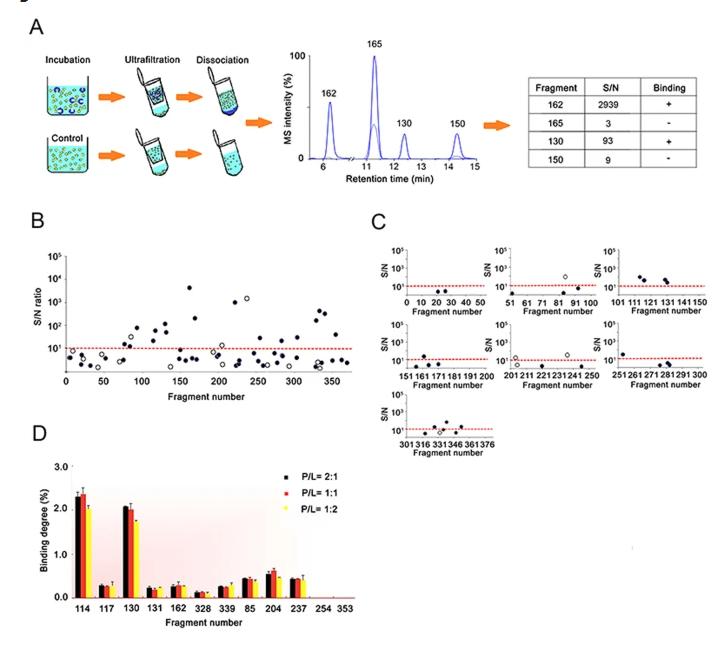
Fragment-based lead discovery depends on reliable early evidence that a fragment is interacting with the target. Because weak binders can be difficult to confirm with a single platform, ligand-observed mass spectrometry was investigated as a way to strengthen the early screening and confirmation workflow for the HCV RNA polymerase NS5B target.
Methods
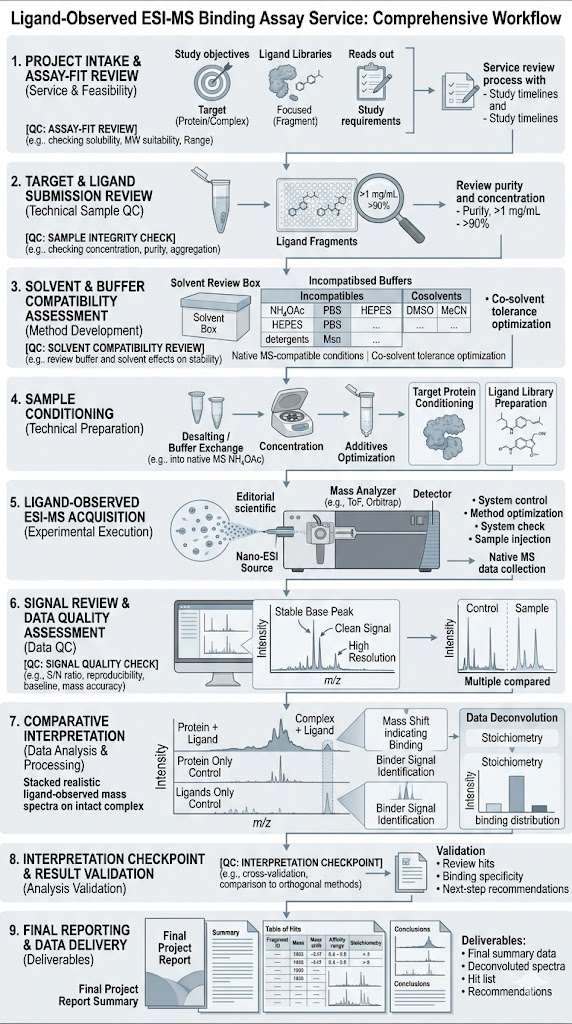
The study integrated a ligand-observed mass spectrometry workflow into an FBLD campaign. A 384-member fragment library was screened through two independent screens of complex cocktails, followed by a validation assay to confirm the resulting candidates.
Results
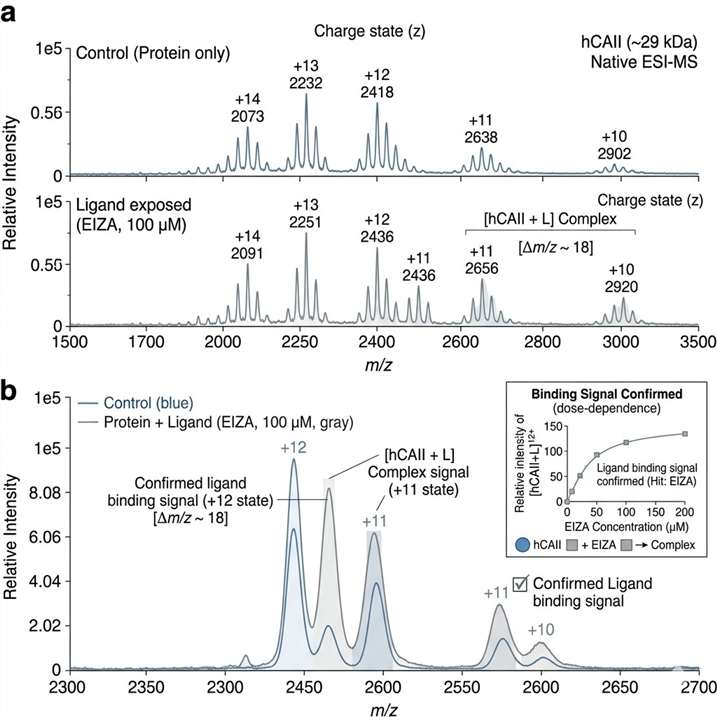
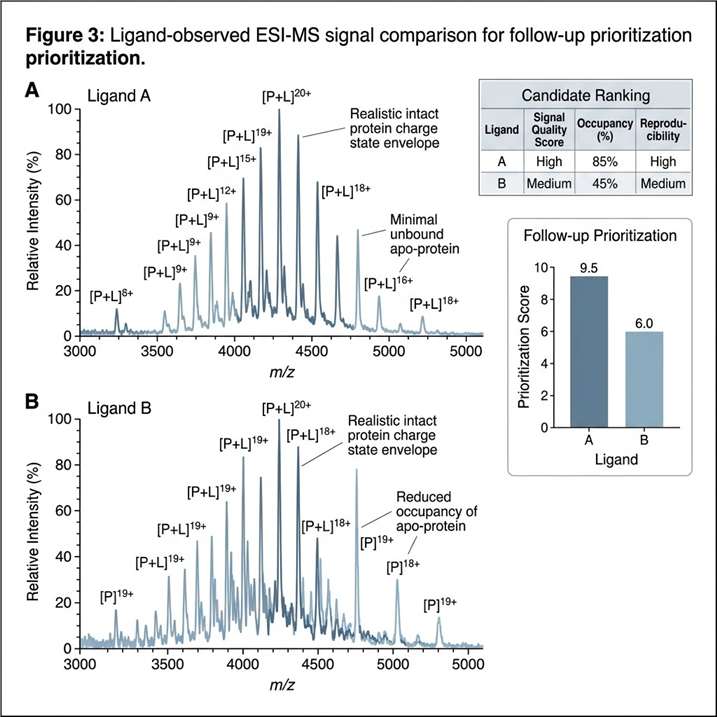
The ligand-observed MS workflow identified 10 hits from the 384-member fragment library. The study also showed that the method could be used for quantitative measurement of weak binding affinities, and that these measurements were generally consistent with SPR analysis.
Conclusion
This case shows that ligand-observed MS can be integrated into a fragment-based discovery workflow as a practical method for early binding evidence, hit refinement, and follow-up planning. For projects involving weak binders or early-stage candidates, it supports a route that is fast, label-free, and compatible with broader orthogonal validation strategies.