Acoustic Ejection Mass Spectrometry (AEMS / Echo-MS) Service for Ultra-Fast Label-Free Screening
We deliver sub-2-second cycle time label-free screening by acoustic ejection mass spectrometry — enabling direct-to-biology reaction monitoring, enzyme kinetics, and ultra-HTS at nanoliter sample consumption.
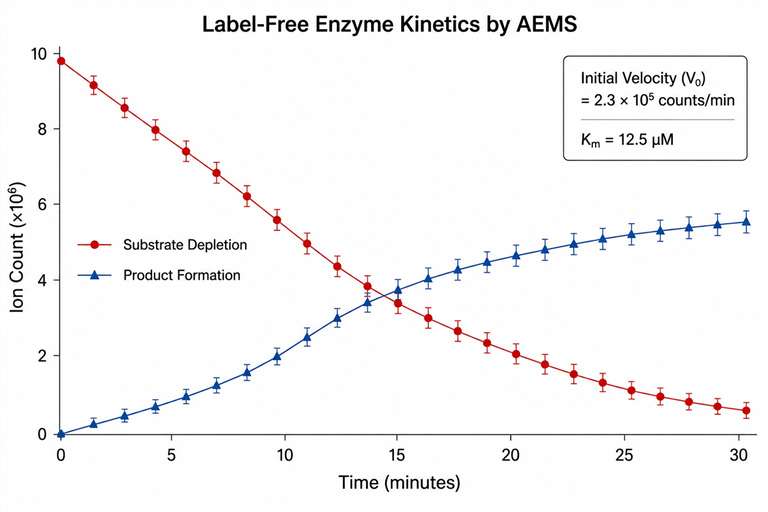
Acoustic ejection mass spectrometry (AEMS), also known as Echo-MS, is an ultra-high-throughput analytical platform that uses focused acoustic energy to eject nanoliter droplets directly from a multiwell plate into an open-port interface (OPI), where they are carried by a solvent stream into an electrospray ionization (ESI) mass spectrometer. The entire process — from well to mass spectrum — takes less than 2 seconds per sample.
Key Advantages:
- Sub-2-second cycle time — Acoustic droplet ejection + open-port interface MS achieves the fastest sample-to-result time of any MS-based screening platform.
- Nanoliter sample consumption — Each acoustic ejection uses only ~2.5 nL of sample, preserving precious compounds and enabling repeated analyses.
- Direct-to-biology screening — Analyse crude reaction products without purification, accelerating the design-make-test cycle.
- No carryover, no cross-contamination — Contactless acoustic ejection eliminates sample-to-sample carryover inherent in autosampler-based systems.