Drug-Resistance Mechanism Analysis Service — MS-Based Multi-Omics Profiling
Elucidate the molecular mechanisms driving drug resistance with integrated proteomics, metabolomics, and phosphoproteomics — from in vitro models to patient-derived specimens.
Drug resistance remains the single greatest barrier to effective cancer therapy. Whether in targeted therapy, chemotherapy, or immunotherapy, resistant clones inevitably emerge — yet the underlying molecular mechanisms often remain a black box. Genomic approaches alone cannot capture the post-translational signaling rewiring, metabolic reprogramming, and proteome-level adaptations that drive resistance.
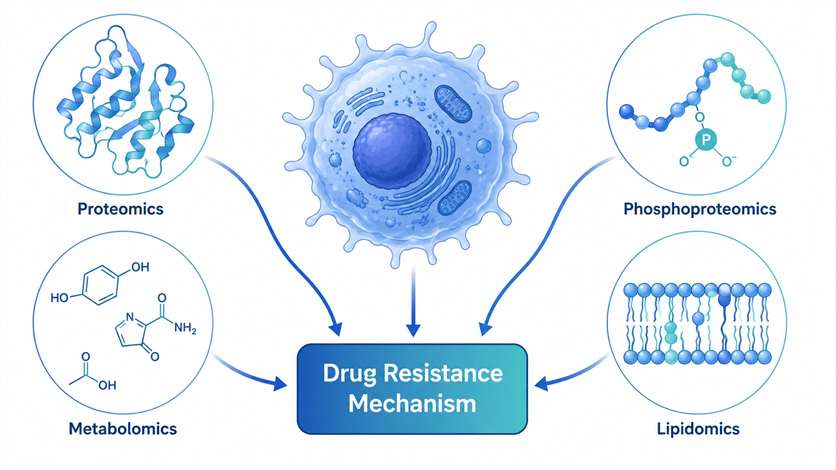
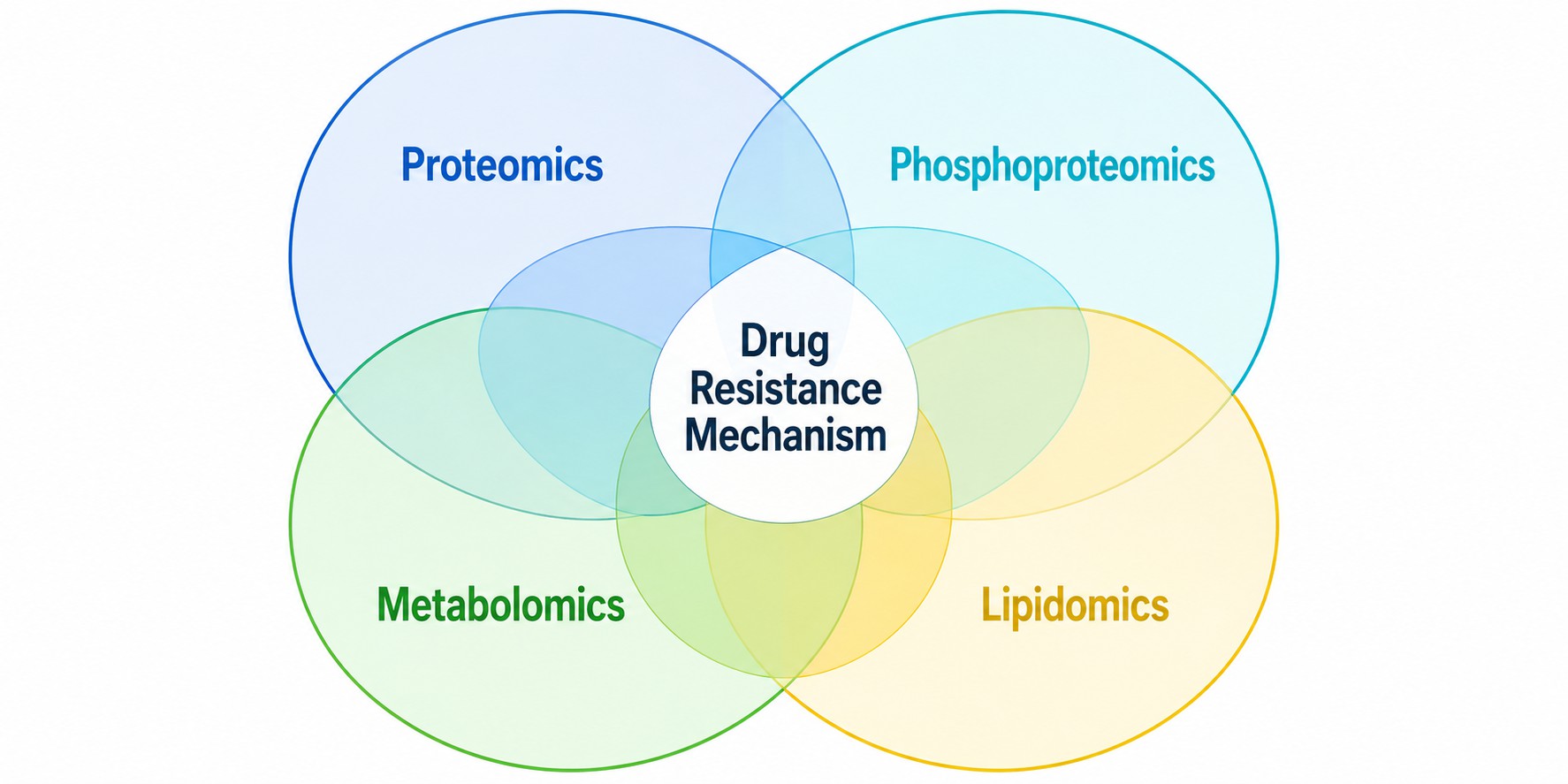
MassTarget™ offers a comprehensive Drug-Resistance Mechanism Analysis Service that integrates mass spectrometry-based proteomics, phosphoproteomics, metabolomics, and lipidomics to systematically map the molecular landscape of drug resistance. Our multi-omics approach covers the full spectrum of resistance mechanisms including drug target alterations, signaling pathway rewiring, metabolic reprogramming, efflux pump upregulation, epithelial-mesenchymal transition, apoptosis evasion, and DNA damage repair activation.
By combining multiple MS-based analytical modalities under one roof, we provide an integrated view of resistance mechanisms that no single-omics approach can deliver — enabling you to identify actionable targets for combination therapy and biomarker strategies.