MALDI-TOF HTS for Label-Free Biochemical Drug Screening
We deliver label-free, high-throughput biochemical screening by MALDI-TOF mass spectrometry — enabling direct substrate-to-product readout for enzyme inhibition, fragment screening, and IC50 determination without fluorescent labels or assay development bottlenecks.
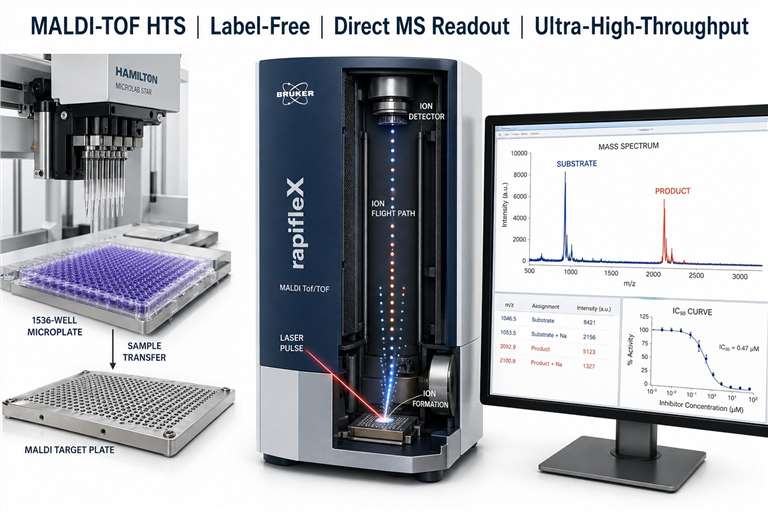
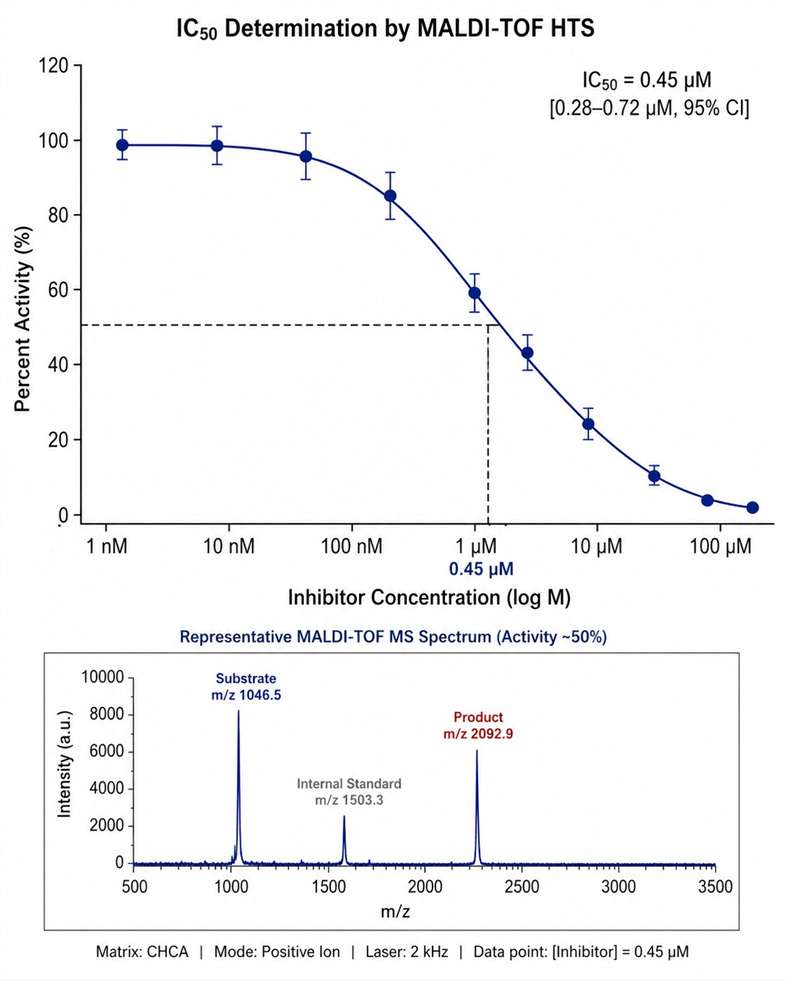
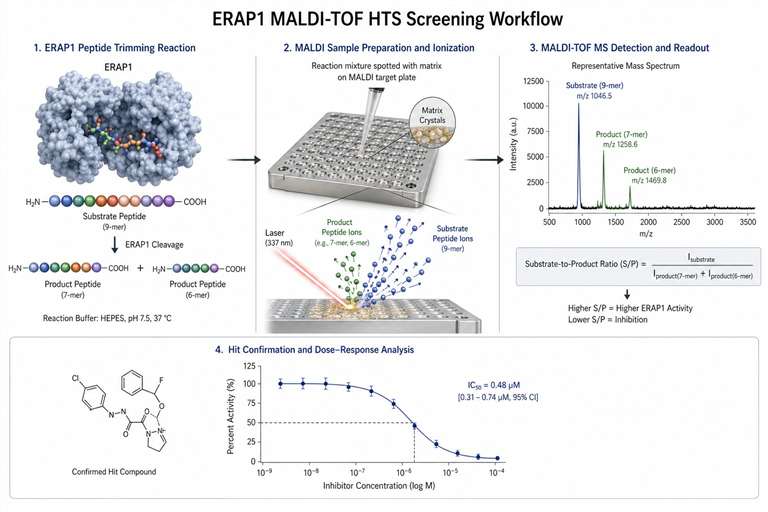
MALDI-TOF high-throughput screening (MALDI-TOF HTS) is a label-free mass spectrometry approach that uses matrix-assisted laser desorption/ionization time-of-flight MS to directly measure enzymatic substrate-to-product conversion, binding events, or biochemical activity in a high-throughput format. Unlike conventional fluorescence-based HTS, MALDI-TOF HTS detects analytes by their intrinsic mass, eliminating the need for fluorescent labels, antibody reagents, or reporter systems.
At Creative Proteomics, our MALDI-TOF HTS service is designed for discovery-stage teams that need label-free, direct-measurement screening without the assay development burden of traditional HTS. We provide end-to-end support — from assay feasibility assessment through full screening campaigns to hit list delivery.
Key Advantages:
- True label-free detection — Direct mass measurement of substrates and products. No fluorescent tags, no FRET pairs, no antibody reagents.
- Ultra-high-throughput capability — 1536-well plate analysed in under 2 minutes with modern MALDI-TOF instrumentation.
- Broad target compatibility — Enzyme inhibition, fragment screening, binding assays, and direct-to-biology screening of crude reaction mixtures.
- Minimal assay development — Days instead of weeks or months compared to fluorescence-based HTS optimisation.