CYP450 Inhibition Panel — LC-MS/MS Cocktail Assay for Drug-Drug Interaction Screening
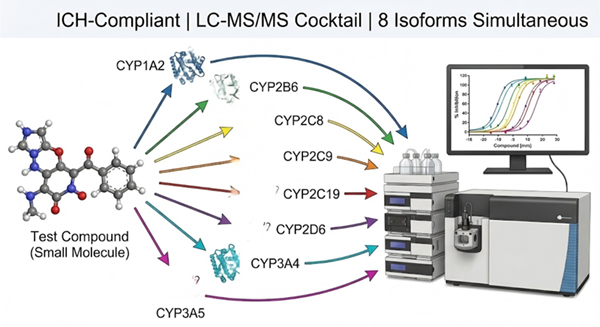
ICH-compliant CYP450 inhibition screening that simultaneously evaluates your compound's inhibitory potential against 7-8 major hepatic isoforms in a single LC-MS/MS cocktail incubation.
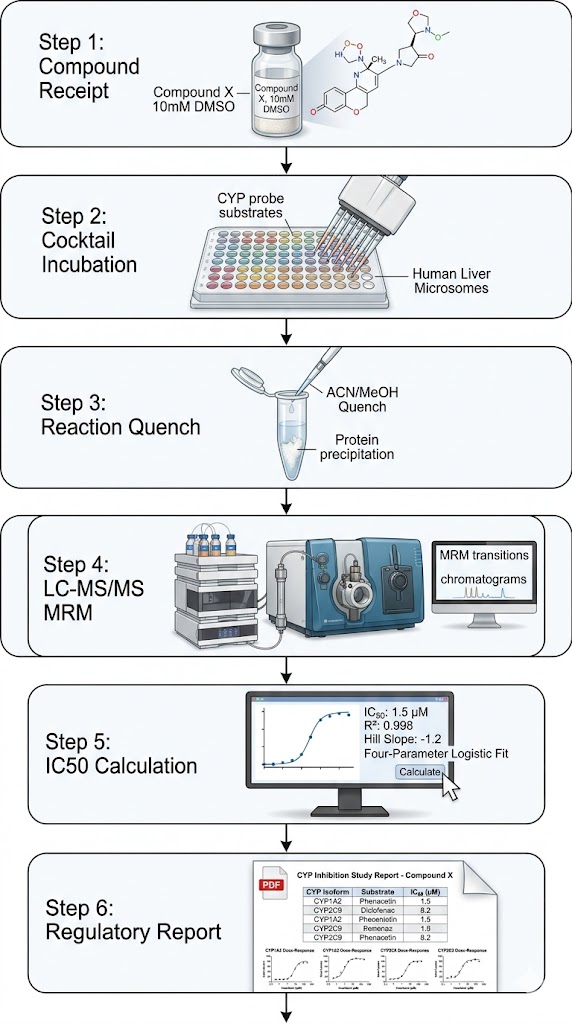
At Creative Proteomics, our CYP450 inhibition panel service is designed to address the regulatory requirement for in vitro DDI assessment before first-in-human dosing — delivering regulatory-ready IC50 data free from fluorescence interference.
Leveraging our validated LC-MS/MS cocktail approach with pooled human liver microsomes, we deliver multi-isoform IC50 results from a single incubation, reducing compound consumption by up to 8-fold compared to individual probe assays.
Key Advantages:
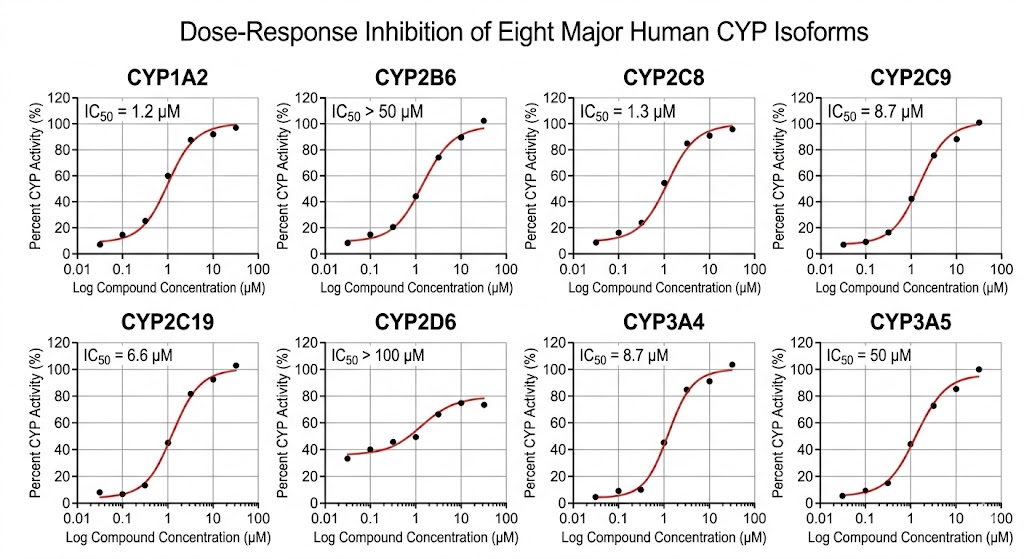
- ICH-compliant CYP panel covering 7 major hepatic isoforms (CYP1A2, 2B6, 2C8, 2C9, 2C19, 2D6, 3A4/5) plus optional CYP2E1.
- LC-MS/MS cocktail approach — 8 probe substrates in a single incubation, 8 IC50 curves from one LC-MS/MS run.
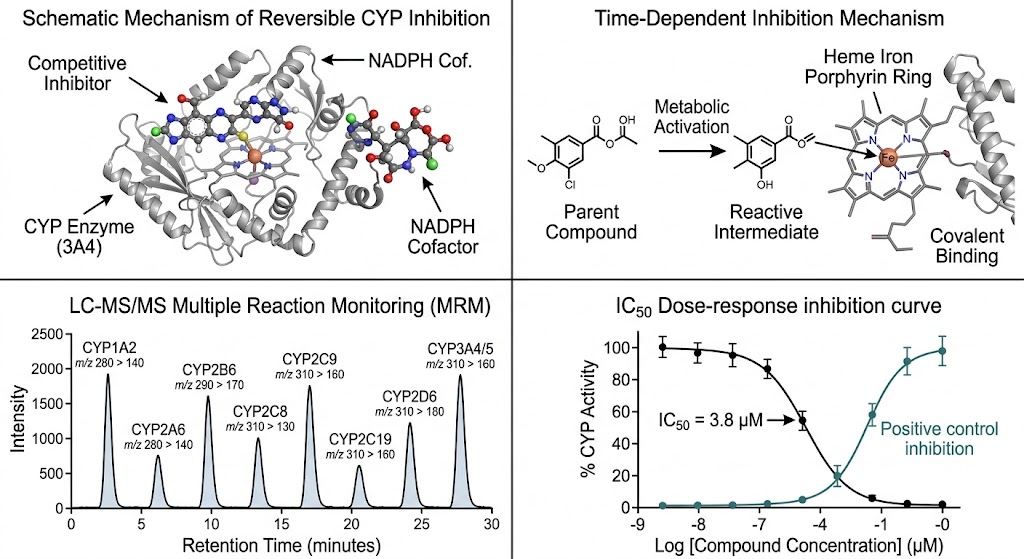
- No fluorescence interference — mass-based MRM detection eliminates autofluorescence, quenching, and inner-filter effect artifacts.
- Time-dependent inhibition (TDI) add-on with ±NADPH pre-incubation design for mechanism-based inactivation risk assessment.