Bio-Layer Interferometry (BLI) Service
Real-time, label-free binding analysis for affinity, kinetics, competition, and clone-ranking decisions in drug discovery.
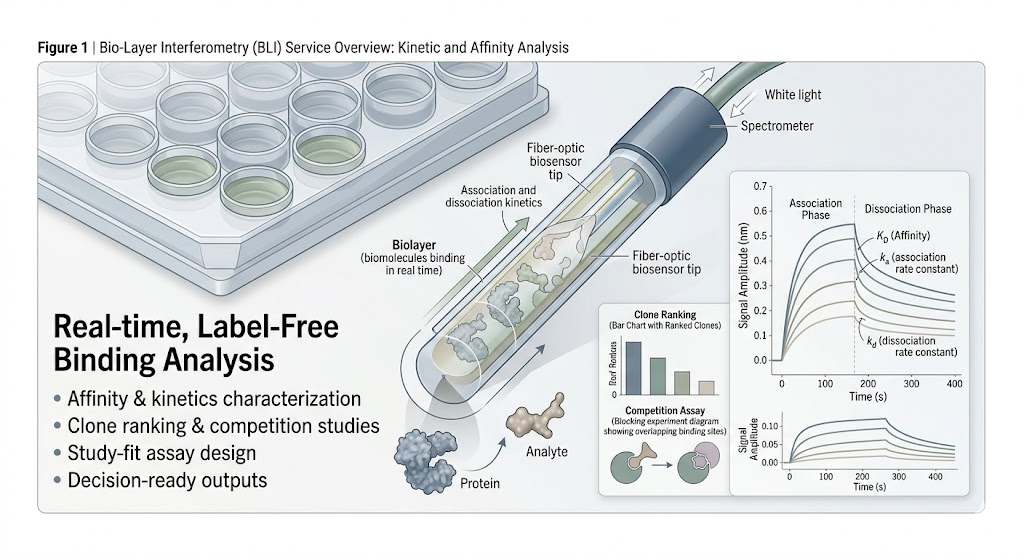
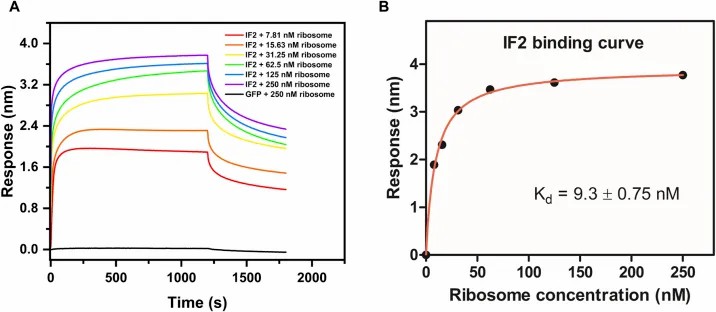
Bio-Layer Interferometry (BLI) Service gives you a real-time, label-free way to study molecular interactions when the key question is not only whether binding happens, but how strong it is, how fast it forms, how fast it dissociates, and how candidates compare across a discovery workflow.
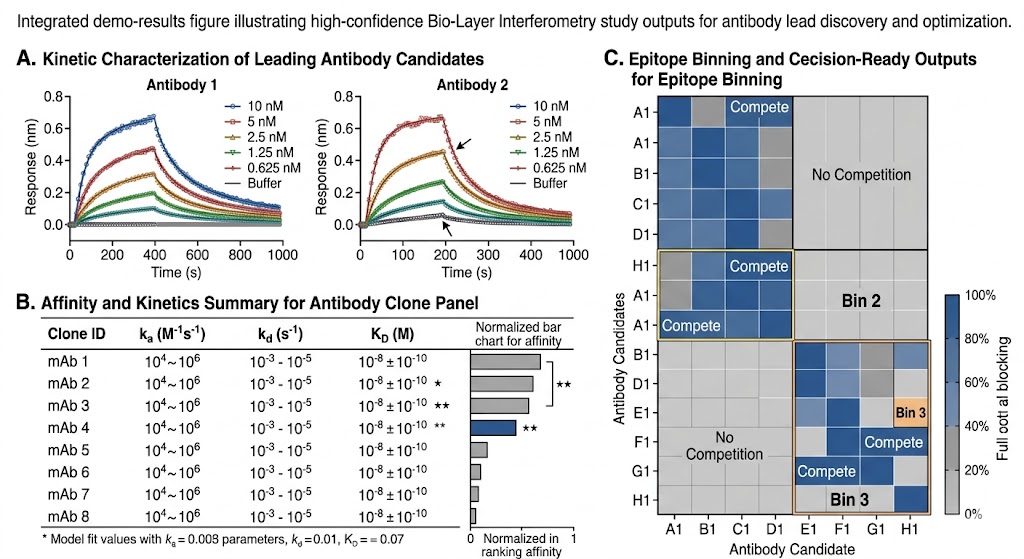
We use BLI to support affinity and kinetics characterization, clone ranking, competition studies, and epitope binning in projects where clear interaction data can help move a program forward with stronger technical confidence.
Whether you are comparing antibody clones, evaluating a receptor-ligand interaction, or deciding whether BLI or an adjacent method is the better fit, we frame the project around the scientific question you need the data to answer.
Key Advantages:
- Real-time, label-free interaction analysis for practical project decisions.
- Affinity and kinetics can be reviewed within the same study design.
- Useful for ranking, competition, and clone differentiation workflows.
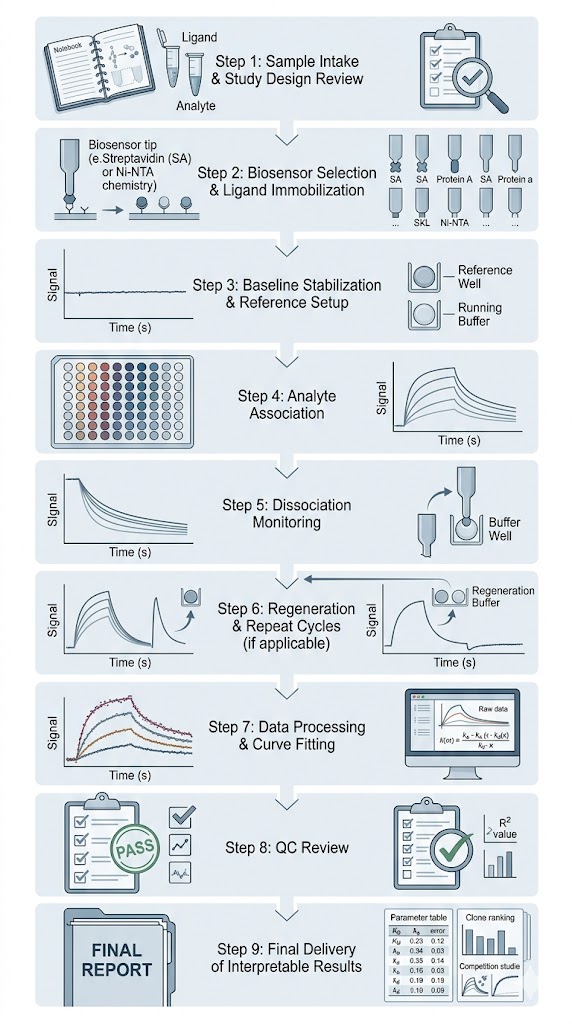
- Built around study-fit assay design and clear QC checkpoints.
- Delivered as decision-ready outputs, not raw curves alone.