Cell Permeability Assessment by LC-MS/MS
Direct, quantitative measurement of drug transport across living cell monolayers — Papp, efflux ratio, and intracellular concentration delivered via high-sensitivity LC-MS/MS.
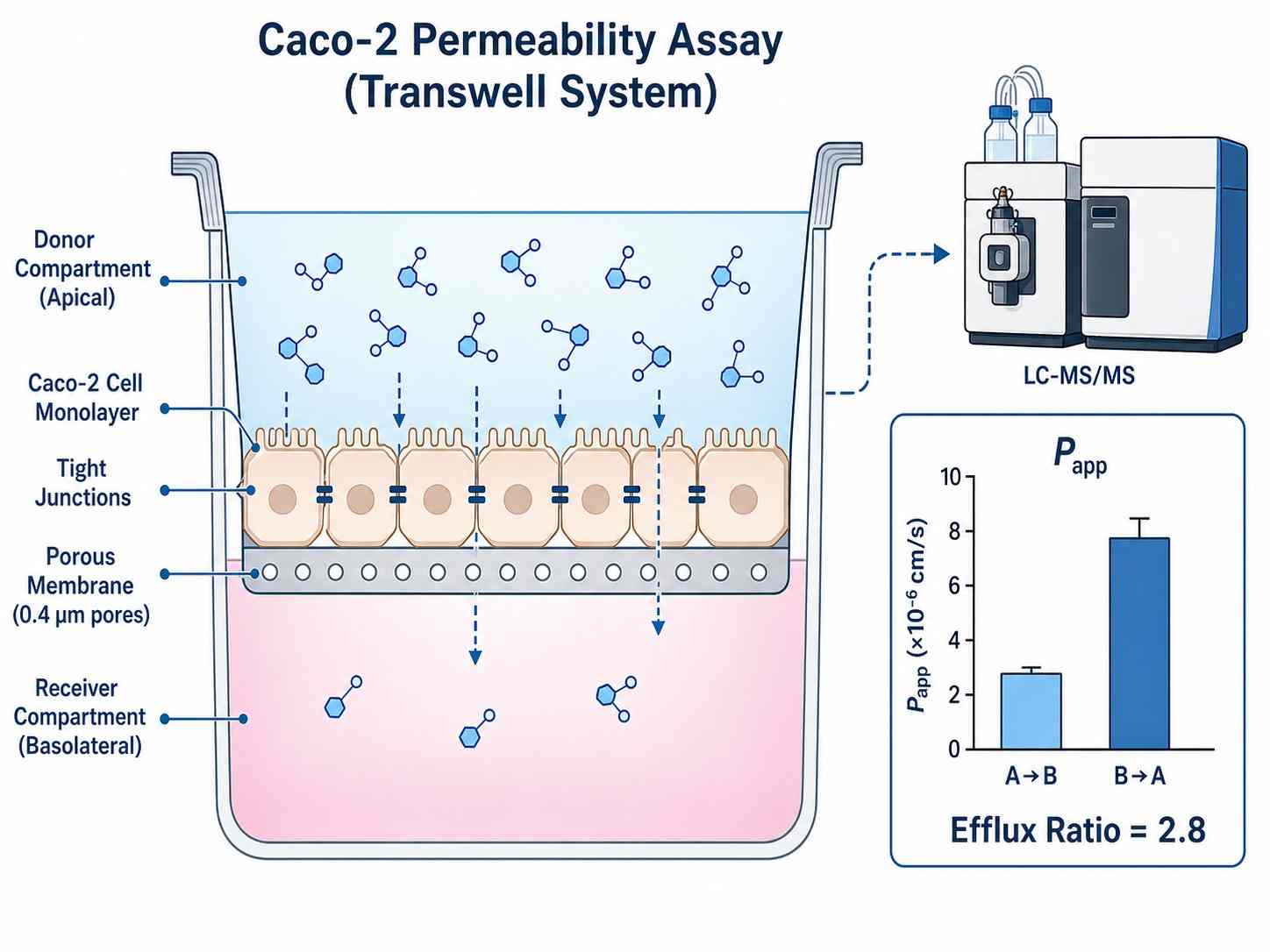
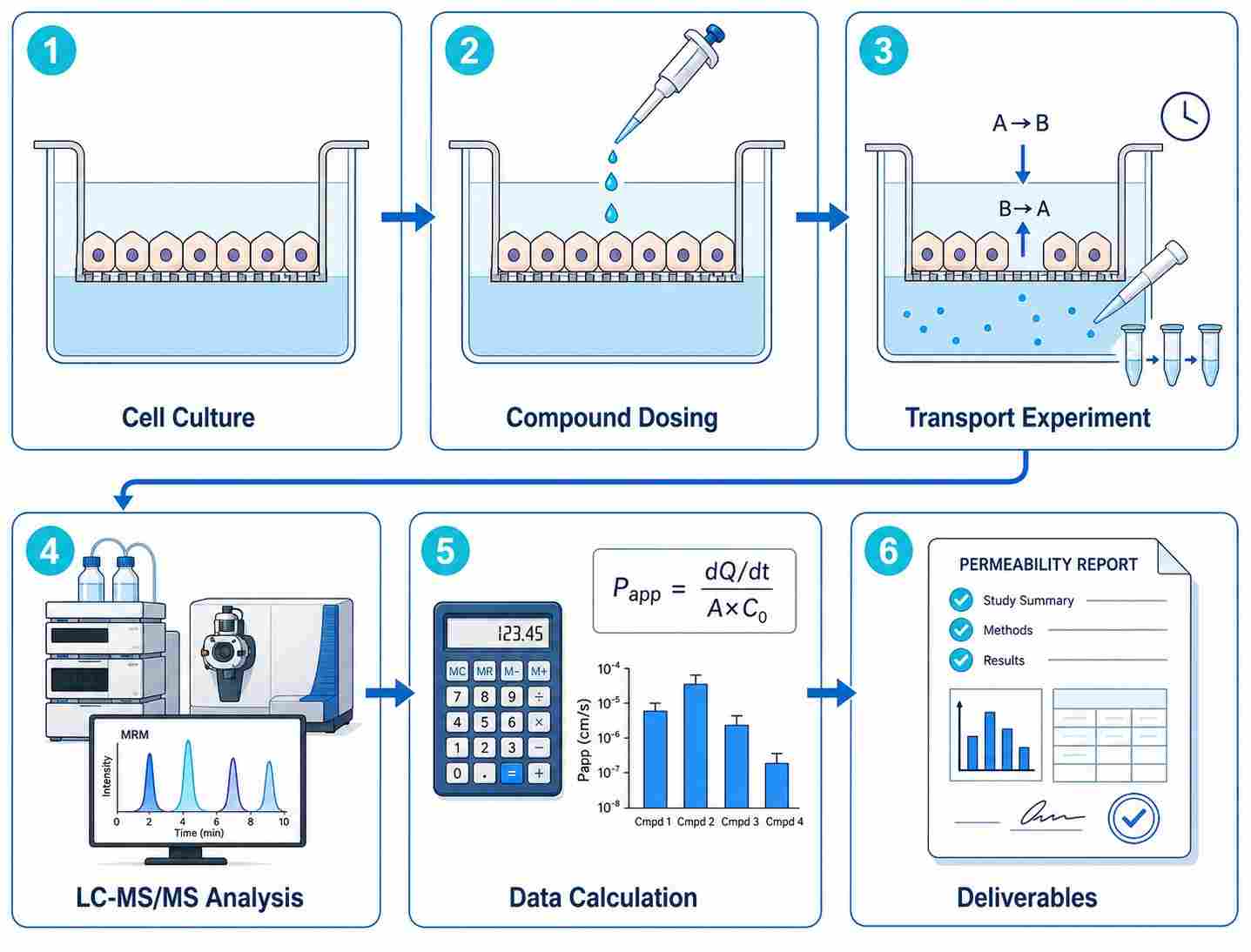
Cell permeability is one of the most critical determinants of oral drug bioavailability and intracellular target engagement. Yet for decades, drug discovery teams have relied on indirect estimates — logD predictions, artificial membrane assays (PAMPA), or computational models — that often fail to predict real cellular permeability. Our Cell Permeability Assessment by LC-MS/MS service fills this gap by providing direct, quantitative measurement of drug transport across living cell monolayers using high-sensitivity liquid chromatography-tandem mass spectrometry.
We deliver apparent permeability coefficients (Papp), efflux ratios, recovery rates, and intracellular concentrations for your compounds, enabling data-driven decisions during lead optimization.
Key Capabilities:
- Direct LC-MS/MS quantification of apical-to-basolateral and basolateral-to-apical transport
- Multiple validated cell models: Caco-2, MDCK-MDR1, MDCK-WT, primary cells, and custom models
- Papp values, efflux ratios (ER), recovery rates, and intracellular concentrations
- High-throughput capacity: 100+ compounds per week via automated sample preparation
- Bidirectional transport assays with P-gp, BCRP, and MRP substrate assessment
- Compatible with small molecules, PROTACs, macrocycles, peptides, and beyond-Rule-of-5 compounds
- Integrated with our cell-based MS drug screening platform