Intracellular Drug Uptake & Retention Analysis by LC-MS/MS
Direct, quantitative measurement of intracellular compound accumulation, retention kinetics, and subcellular distribution — label-free LC-MS/MS quantification for any small molecule.
A compound's ability to enter cells and remain at a sufficient concentration over time is one of the most critical determinants of its pharmacological activity. Yet for decades, drug discovery teams have relied on a critical but incomplete surrogate — cell permeability (Papp) — to estimate intracellular exposure. While permeability tells you how fast a compound crosses a membrane, it does not tell you how much compound actually accumulates inside cells, how long it stays there, or what fraction is free to engage its target.
The disconnect between permeability and intracellular concentration is well documented. A compound with high Papp may be rapidly effluxed, metabolized intracellularly, or sequestered in lysosomes, resulting in a free intracellular concentration far below what permeability data would suggest. Conversely, a compound with moderate permeability but low efflux and high retention may achieve superior intracellular exposure.
At Creative Proteomics, our Intracellular Drug Uptake & Retention Analysis by LC-MS/MS service fills this critical gap by providing direct, quantitative measurement of intracellular compound concentration — enabling data-driven decisions during lead optimization, target engagement correlation, and drug resistance mechanism studies.
Key Capabilities:
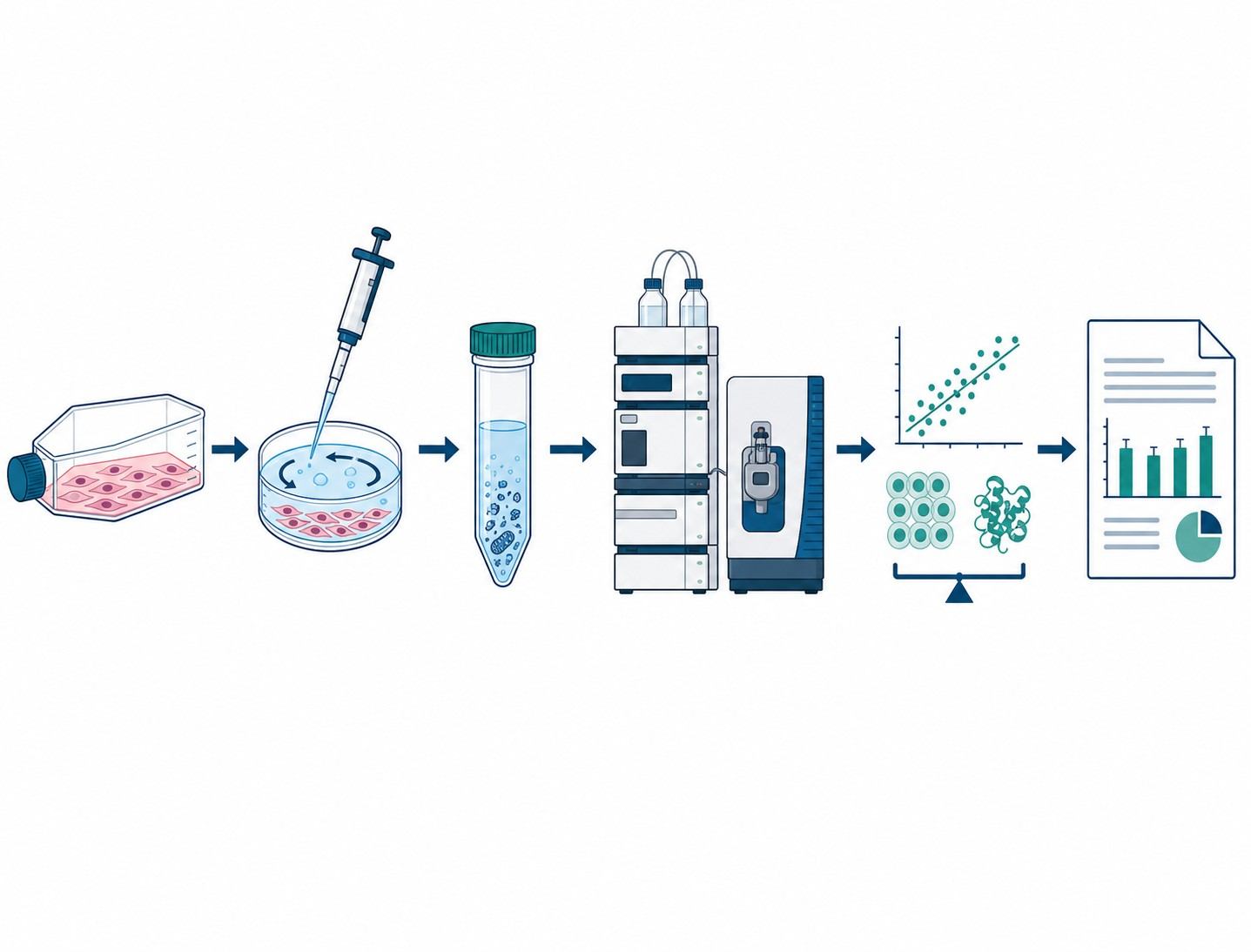
- Direct LC-MS/MS quantification of intracellular compound concentration — label-free, compound-specific, compatible with any small molecule
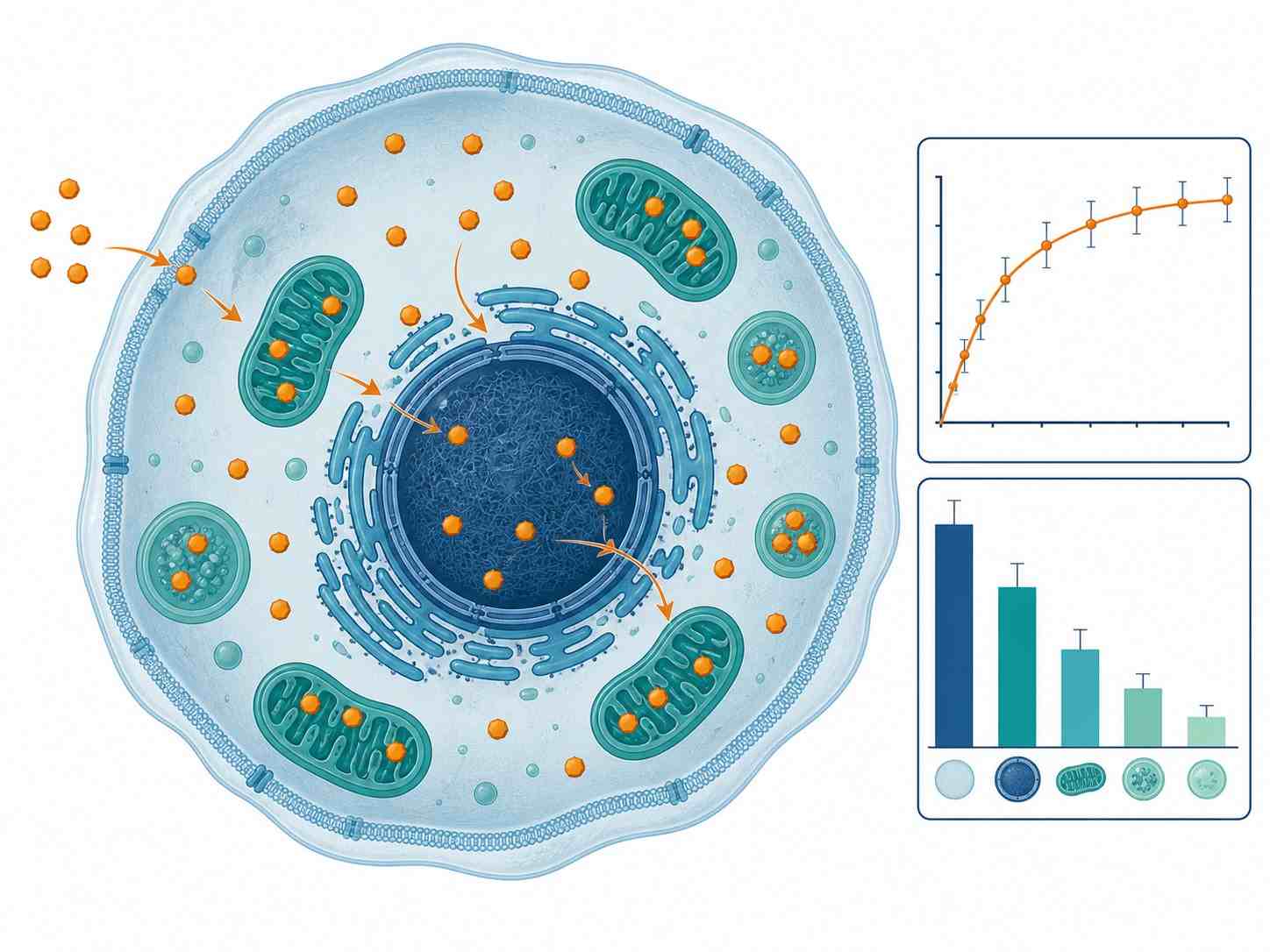
- Time-course uptake kinetics (0-120 min), steady-state accumulation, and efflux/retention profiling
- Subcellular fractionation: cytosolic, nuclear, mitochondrial, and membrane-bound distribution
- Multiple validated cell models: suspension cells, adherent cells, primary hepatocytes, 3D spheroids, and drug-resistant lines
- PROTAC, macrocycle, and beyond-Rule-of-5 compound compatibility
- Integrated with our cell-based MS drug screening platform