Ion Mobility Mass Spectrometry (IM-MS) Service
4D separation for isobars, conformers, and native complexes.
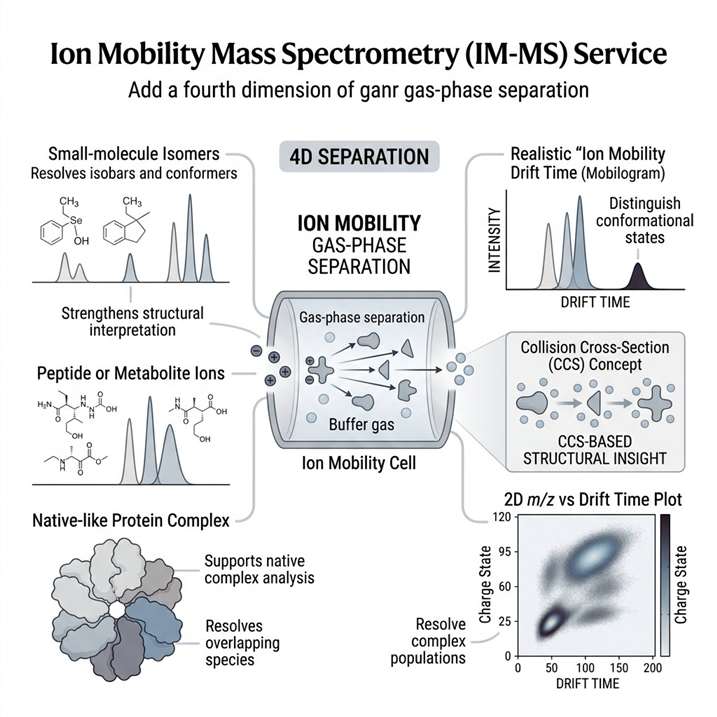
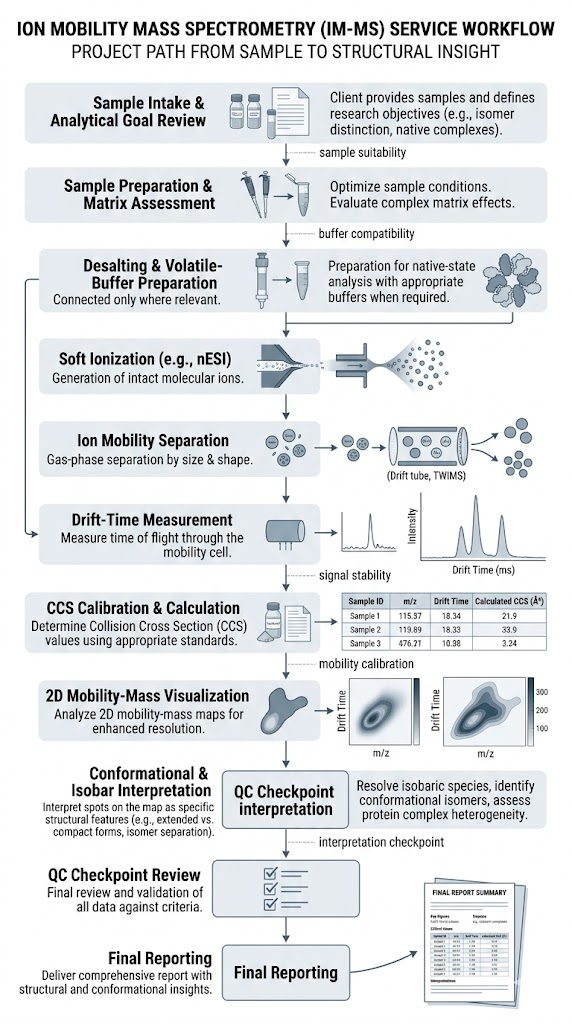
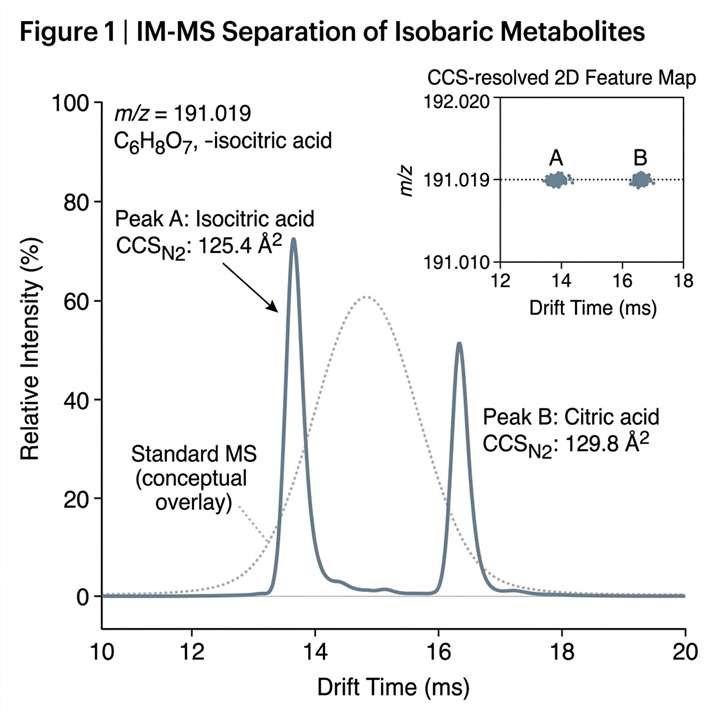
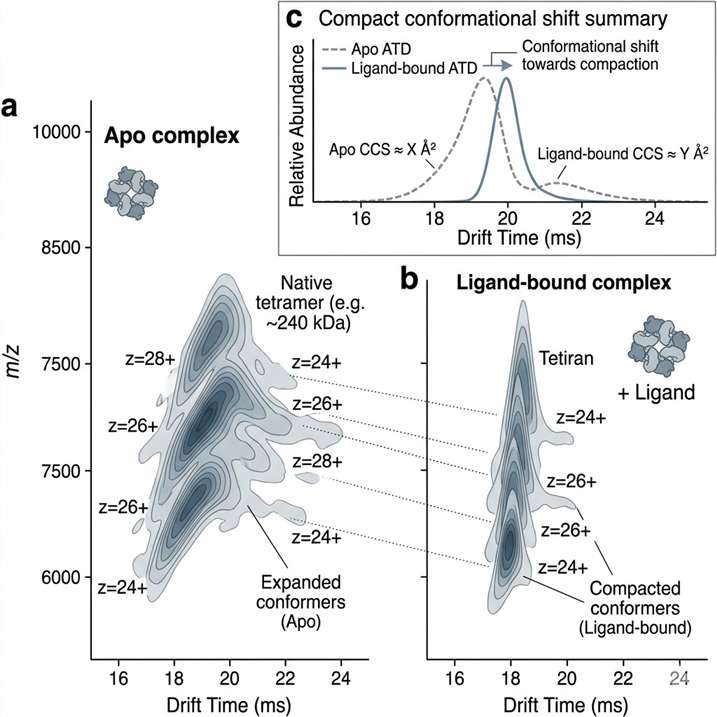
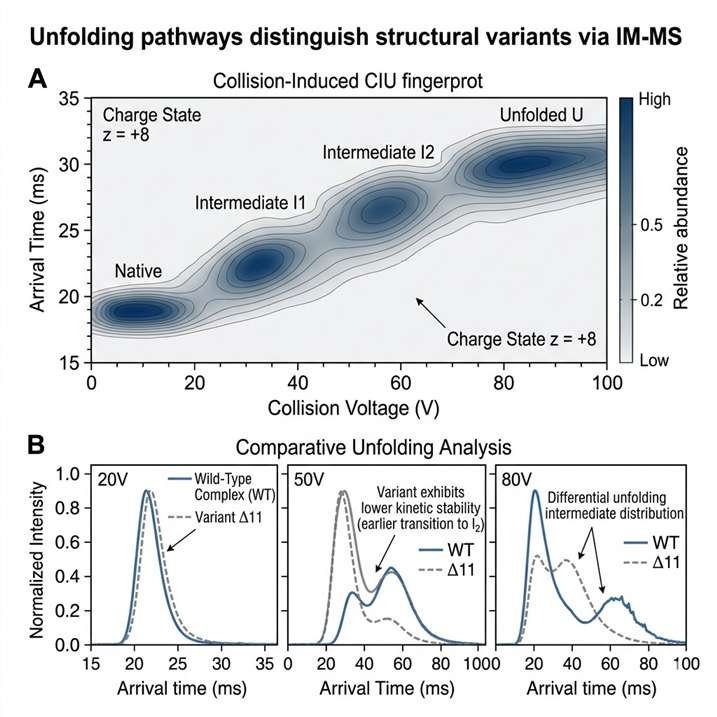
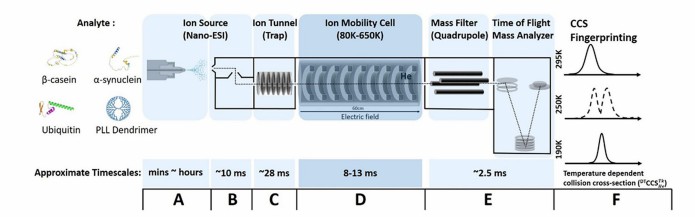
Ion Mobility Mass Spectrometry (IM-MS) Service adds shape-based gas-phase separation to mass spectrometry, helping us resolve isobars, distinguish conformers, and characterize intact complexes with greater structural confidence. It is especially useful when conventional LC-MS cannot clearly separate overlapping species or explain conformation-dependent behavior.
Key Advantages:
- 4D separation for complex samples.
- CCS-based structural discrimination.
- Useful for intact complexes.
- Supports conformational analysis.
- Built for follow-up decisions.