Breast cancer is one of the most common cancers in women, and both men and women are at risk for developing breast cancer, but the incidence is much higher in women than in men. Despite advances in genomic classification of breast cancer, which have significantly improved diagnosis and treatment, current clinical tests and treatment decisions are still usually based on protein-level information.
Proteomics research can provide data to support breast cancer pathogenesis studies, staging, and therapeutic target discovery.
Case 1 Study on Mechanism of Breast Cancer Development
The heterogeneity of breast cancer poses a great challenge for personalized treatment. Precision therapy is based on targeting DNA repair defects, activated protein kinases, estrogen receptors and immune microenvironment. However, effective treatment relies on accurate tumor analysis and target identification. Research teams from the Boulder Institute, Baylor College of Medicine, and the Clinical Proteomic Tumor Analysis Consortium (CPTAC) have taken advantage of the proteogenomic combination of DNA, RNA, and proteins to comprehensively characterize the mechanisms of breast carcinogenesis and its targeted therapeutic molecules.
Tandem mass tag (TMT)-based isobaric labeling provided precise relative quantification of proteins and phosphorylation and acetylation sites following published CPTAC protocols. 10,107 proteins, 38,968 phosphorylation sites and 9,869 acetylation sites were found.
Through proteomic characterization and genome sequencing of the breast cancer population, the phosphorylated protein characteristics, metabolic characteristics and immune microenvironment characteristics of each subtype can be elaborated, which can provide a new perspective and a more comprehensive analysis for personalized treatment.
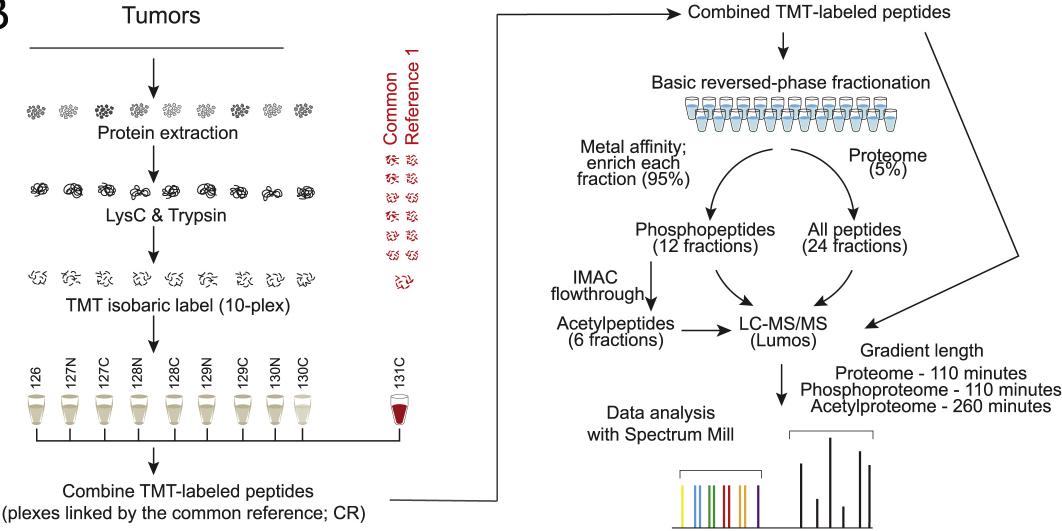
Schematic representation of the workflow used for proteome, phosphoproteome and acetylproteome analyses (Krug et al., 2020)
Case 2 Study of Breast Cancer Staging
Triple negative breast cancer (TNBC) is a highly aggressive cancer, and the most lethal subtype of TNBC is metaplastic breast carcinomas (MBC). This is a uniquely heterogeneous subtype that accounts for 0.2-5% of all breast cancers. MBCs are more metastatic and chemoresistant than non-chemotropic TNBCs, and the sideroblastic subtype has the poorest prognosis. Therefore, differentiation of MBC subtypes can lead to diagnosis and precise treatment.
The team of Celina G. Kleer at the University of Michigan, Ann Arbor, USA, used quantitative proteomics techniques to analyze 27 cases of MBC, TNBC and normal breast tissues to systematically profile the full spectrum of proteins in MBC and TNBC. The different MBC isoforms showed different protein expression differences compared to TNBC. By comparing the human and mouse spindle cell MBC proteomes, 17 up-regulated proteins involved in the translation process and 19 down-regulated proteins involved in cellular metabolism were found to have the same spindle specificity. These data identified potential diagnostic and prognostic markers for MBC, as well as therapeutic targets for MBC subtypes.
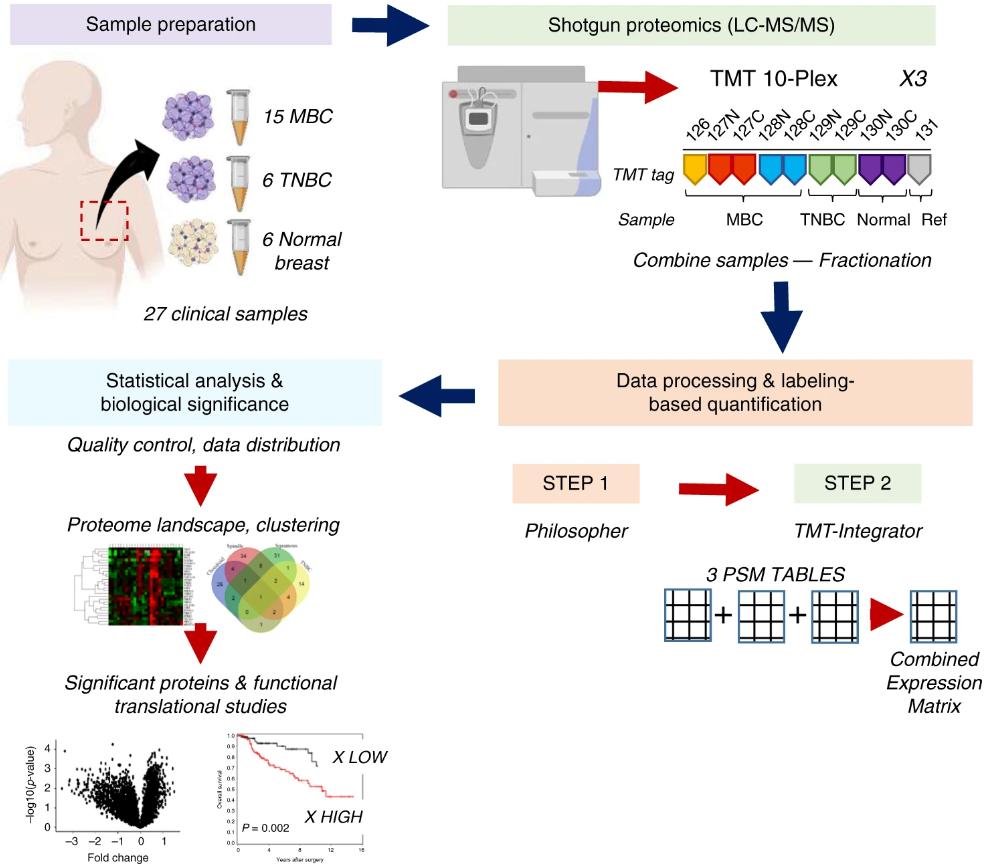
Workflow of quantitative mass spectrometry profiling (Djomehri et al., 2020)
Case 3 Identifying potential biomarkers and therapeutic targets for breast cancer
Immune differences, ECM and lipid metabolism pathways may have clinical relevance. This study used SP3-CTP multiplex MS proteomics to analyze FFPE samples. Digested peptides were labeled with stable isotope labeled tandem mass tags (TMT). The entire cohort was run in TMT-11-multiplex groups. Each group had two PAM50 subtypes, a normal sample, a standard SuperMix and a mixed sample. Proteomics analysis quantified a total of 9088 proteins (average ~6500 proteins/11-plex) and quantified 4214 proteins in all samples.
Immune differences, ECM and lipid metabolism pathways may have clinical relevance. The study used SP3-CTP multiplex MS proteomics to analyze FFPEsamples. digested peptides were labeled with stable isotope labeled tandem mass tags (TMT). The entire cohort was run in TMT-11-multiplex groups.
Each group had two PAM50 isoforms, a normal sample, a standard SuperMix and a mixed sample. Proteomics analysis quantified a total of 9088 proteins (average of ~6500 proteins/11-plex) and quantified 4214 proteins in all samples. Among the basal-like cases with aggressive PAM50 classification, proteomic analysis identified one group with a characteristic immune thermal expression profile and the best survival.
Her2-Enriched cases were classified into heterogeneous groups based on extracellular matrix, lipid metabolism and immune response characteristics. In 88 cases of triple-negative breast cancer, four proteomic groups showed basal immune heat, basal immune cold, mesenchymal and ductal features with different survival outcomes.
Proteomic analysis identified potential biomarkers and therapeutic targets and provided a resource for clinical breast cancer classification.
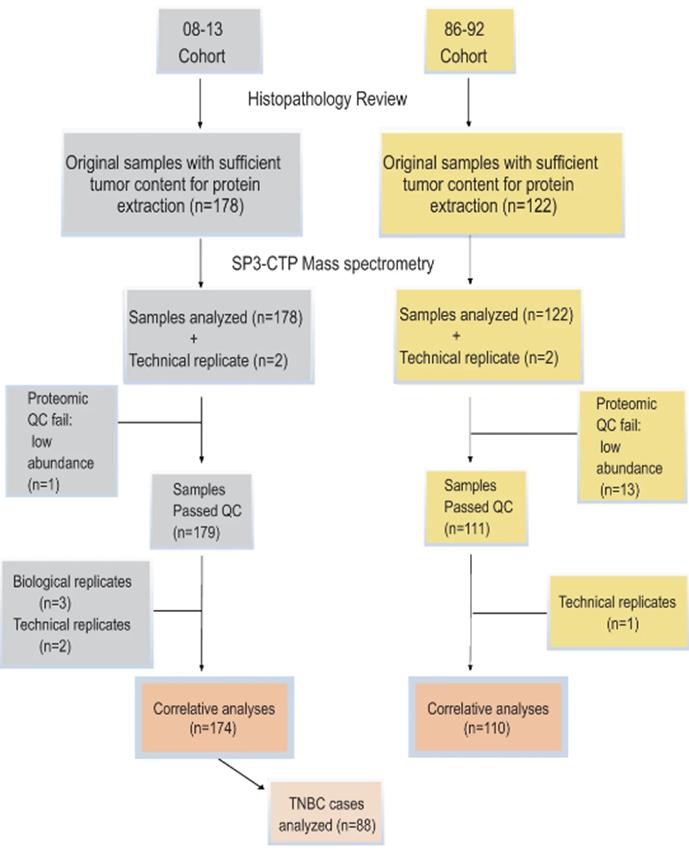
CONSORT flow diagram depicting the workflow numbers for the cases included in the study cohorts (Asleh et al., 2020)
Resources
- Krug, K., Jaehnig, E. J., et al. (2020). Proteogenomic landscape of breast cancer tumorigenesis and targeted therapy. Cell, 183(5), 1436-1456.
- Djomehri, S. I., Gonzalez, M. E., et al. (2020). Quantitative proteomic landscape of metaplastic breast carcinoma pathological subtypes and their relationship to triple-negative tumors. Nature communications, 11(1), 1-15.
- Asleh, K., Negri, G. L., et al. (2022). Proteomic analysis of archival breast cancer clinical specimens identifies biological subtypes with distinct survival outcomes. Nature communications, 13(1), 1-19.







