Meta Intent: A mechanism-first technical resource on how ATP is generated, partitioned, sensed, and consumed across plant organelles, with emphasis on chloroplast–mitochondrial coordination, compartment-specific measurement, and ATP-dependent control of carbon fixation.
Plant ATP is easy to oversimplify. The standard description says chloroplasts make ATP in the light, mitochondria make ATP in the dark, and the cell spends that ATP on growth, transport, and biosynthesis. That summary is useful for teaching, but it is not good enough for interpreting real plant physiology. In leaves, ATP is not a single pool with a single meaning. It is a rapidly turning set of compartment-specific fluxes shaped by light, organelle identity, cell type, and stress state.
That distinction matters because plant metabolism is organized around spatial constraints. ATP made in the chloroplast does not automatically solve a cytosolic ATP problem. Mitochondrial ATP support does not necessarily buffer stromal limitations. A whole-tissue ATP value can therefore be chemically correct while still being biologically incomplete. What researchers often need is not just ATP abundance, but ATP location, ATP timing, and ATP accessibility to the process that is becoming limiting first.
This is why plant bioenergetics is increasingly discussed in terms of spatiotemporal energy dynamics. The key issue is not simply how much ATP a tissue contains. The key issue is how ATP generation and consumption are coordinated across chloroplasts, mitochondria, and the cytosol during light transitions, diel cycling, and environmental stress. Once ATP is viewed that way, many classic plant physiology questions become easier to interpret. Why can electron transport remain active while carbon fixation drops? Why does one tissue tolerate heat better than another despite similar bulk ATP values? Why do some assays detect only weak ATP changes while live sensors show strong compartment-specific shifts? The answer in each case is that ATP behaves more like a regulated flux network than a stored reserve.
The dual-engine logic of ATP generation in plant cells
Photosynthetic cells rely on two energy-generating systems that work under different rules but continuously influence one another. The chloroplast is the primary site of light-driven ATP production. The mitochondrion is the primary site of respiratory ATP generation and metabolic flexibility. Neither operates in isolation. During active photosynthesis, the chloroplast supplies stromal ATP through photophosphorylation, while mitochondria help maintain broader cellular energy balance and redox continuity. During darkness, the hierarchy changes, but the dependence on coordination does not disappear.
A useful way to think about this arrangement is as a dual-engine system. One engine is optimized for harvesting light and converting it into a trans-thylakoid proton gradient. The other is optimized for respiratory buffering, redox balance, and ATP support under variable demand. In a stable environment these engines complement one another smoothly. Under fluctuating light, oxygen limitation, heat stress, or altered carbon flux, the coordination becomes much more visible. The plant is not simply maximizing ATP yield. It is continuously deciding where ATP must be protected, where flexibility matters more than efficiency, and which pathway can absorb the next imbalance.
Photophosphorylation: the Q-cycle, the Z-scheme, and ATP synthase
Chloroplast ATP synthesis begins with the light reactions embedded in the thylakoid membrane. Water is oxidized at photosystem II, electrons enter the photosynthetic electron transport chain, and the Z-scheme describes their energetic progression toward photosystem I and eventual reduction of NADP+ to NADPH. The pathway is familiar, but the ATP logic is often flattened in summary diagrams.
The missing point is that ATP production depends on how efficiently electron flow is converted into proton motive force, not merely on whether electrons move. As electrons pass through the chain, protons accumulate in the thylakoid lumen. The Q-cycle at the cytochrome b6f complex is especially important because it amplifies proton translocation efficiency and helps build the electrochemical gradient that powers ATP synthesis. That gradient, usually described as Δp, contains both a chemical component and an electrical component. Together they create the driving force for the chloroplast CFoCF1-ATP synthase.
ATP synthase is often presented as the final step of the light reactions. Functionally, it is much more than a terminal machine. It determines how quickly the chloroplast can convert stored electrochemical energy into stromal ATP. If proton release through ATP synthase slows, lumen acidification increases. That can strengthen photosynthetic control and alter upstream electron-transfer behavior. If proton release accelerates, ATP production improves, but the balance of photoprotection and energy conversion also shifts. Chloroplast ATP synthase therefore operates at the center of a regulatory tradeoff. It is not just harvesting energy. It is helping define how the photosynthetic apparatus behaves under changing load.
This matters especially under non-steady-state conditions. Real leaves rarely receive constant, uniform light. Sunflecks, canopy movement, stomatal behavior, and changing assimilation demand all reshape chloroplast energy handling within seconds to minutes. Under those conditions, ATP output is best understood as the product of dynamic pressure management across the thylakoid membrane. A measured ATP change may reflect altered electron delivery, altered proton retention, altered ATP synthase conductivity, altered stromal demand, or several of these at once. That is one reason ATP interpretation becomes much stronger when measurement is paired with organelle context rather than treated as an isolated metabolite value.
Mitochondrial oxidative phosphorylation in illuminated tissues
The common classroom narrative makes mitochondria sound secondary during the light period. That is misleading. Mitochondria remain metabolically important in illuminated tissues because chloroplast photochemistry does not automatically satisfy the full ATP and redox requirements of the cell. Carbon fixation, photorespiration, nitrogen metabolism, transport, and cellular maintenance all impose energetic demands that are not perfectly matched by linear electron flow in chloroplasts.
This mismatch is a central feature of plant bioenergetics. Chloroplasts produce ATP and NADPH in a linked way, yet downstream metabolism does not always consume them in the same ratio or in the same compartment. The result is an energetic balancing problem. Part of that problem is handled by cyclic electron flow around PSI. Part is handled by shuttle systems and metabolite exchange. Part is handled by mitochondria, which continue to oxidize substrates, manage reductant pressure, and support ATP availability outside the chloroplast.
That role becomes especially important when photosynthetic metabolism is pushed away from an easy steady state. A leaf under fluctuating light, heat stress, or photorespiratory pressure may still show strong photochemical activity while becoming locally ATP-limited in a specific compartment or process. In these cases, mitochondrial function can determine whether the system remains metabolically balanced or tips into broader dysfunction. This is why plant mitochondria should not be described only as backup power units. They are active partners in maintaining energy continuity while photosynthesis is running.
The alternative oxidase pathway is an efficiency tradeoff, not a defect
A distinctive feature of plant mitochondrial bioenergetics is the alternative oxidase (AOX) pathway. AOX allows electrons to bypass part of the classical cytochrome pathway. Because this route is non-phosphorylating, it conserves less ATP per electron transferred. That is why AOX is often described as energetically inefficient. The description is true in a narrow accounting sense, but incomplete in physiological terms.
AOX is better understood as a flexibility valve. When the classical respiratory chain is over-reduced or when metabolic constraints make strict ATP maximization unstable, AOX allows continued electron flow with reduced ATP conservation. This rerouting lowers energetic efficiency while helping maintain redox continuity and broader metabolic function. In a stressed plant, that tradeoff can be beneficial. The system gives up some ATP yield in order to prevent deeper disruption in electron transport, reactive oxygen balance, and metabolic throughput.
This point is easy to miss if ATP is judged only by total yield. A plant using AOX more strongly may generate less ATP per unit electron flux, yet still preserve physiological performance better than a plant that attempts to conserve ATP more strictly but accumulates damaging redox pressure. For experimental interpretation, the lesson is clear: lower ATP efficiency is not always a sign of failure. Sometimes it is the signature of controlled adaptation.
ATP asymmetry across organelles is biologically meaningful
Another reason whole-cell ATP values can mislead is that ATP movement across organelles is selective rather than unrestricted. In mature mesophyll chloroplasts, stromal ATP is usually treated as a largely local product of photophosphorylation during the day. That means chloroplast ATP availability depends strongly on thylakoid coupling and chloroplast-specific demand. The cytosol cannot simply be assumed to buffer every stromal ATP fluctuation.
At the same time, plant cells are not uniform. Some cell types follow different energetic rules. Guard cells are a useful example because plastidial ATP handling there does not map neatly onto the classic mesophyll model. This is exactly why organelle-specific ATP interpretation matters. A statement that is broadly true for mesophyll chloroplasts may be misleading when extended to all plastids or all tissues. Plant ATP biology is compartment-specific, and often cell-type specific as well.
That asymmetry has direct experimental consequences. A genotype or treatment may show little change in whole-leaf ATP while still experiencing a severe stromal ATP constraint. Another may maintain chloroplast ATP reasonably well but lose cytosolic balance. Without compartment awareness, both cases can be compressed into the same misleading summary. This is one of the main reasons recent work emphasizes organelle-specific ATP/ADP sensing rather than relying entirely on pooled metabolite measurements.
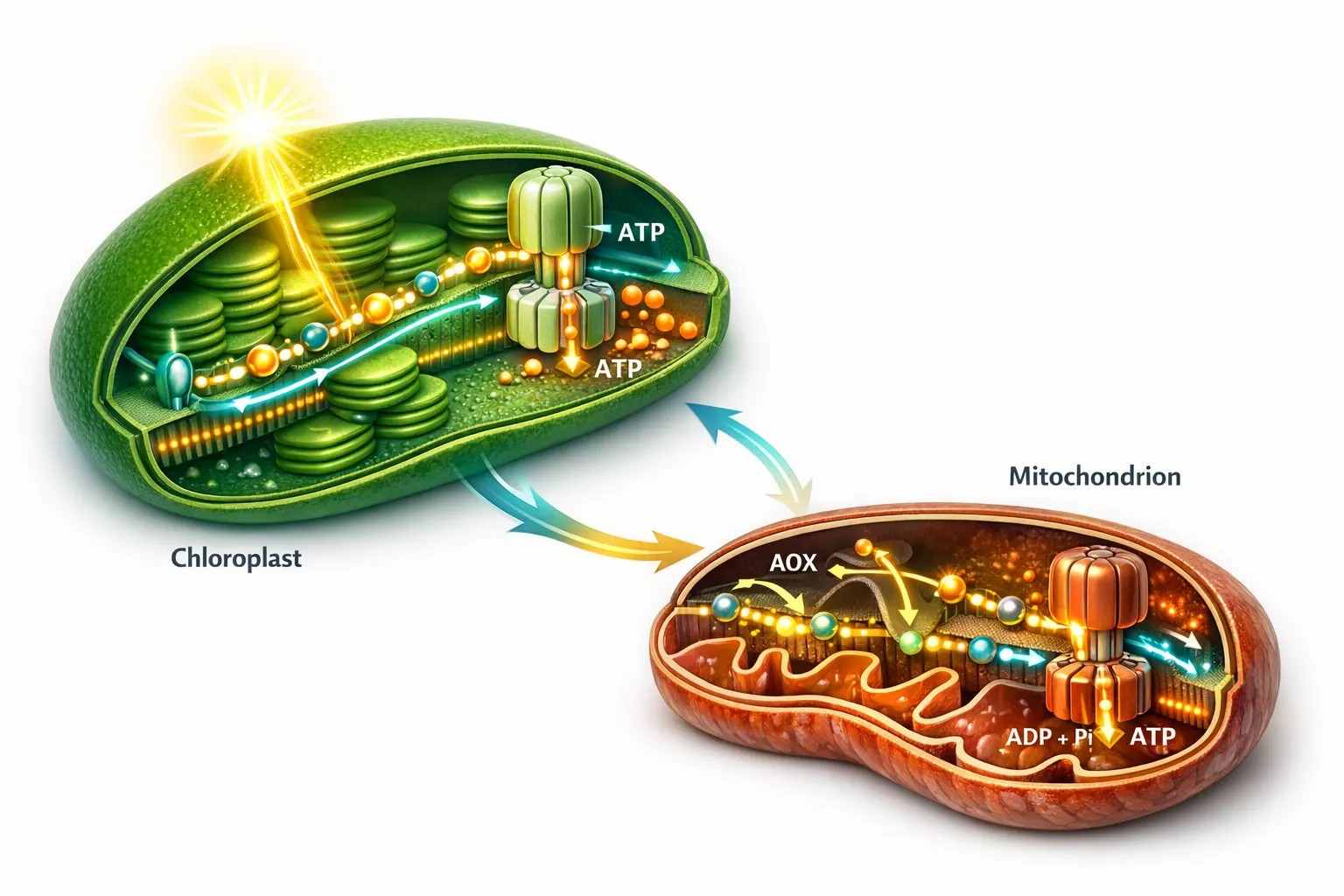
Figure 1. Chloroplast and mitochondrial ATP generation form a dual-engine system in photosynthetic cells. The chloroplast builds proton motive force through the Z-scheme and Q-cycle, while the mitochondrion supports cellular energy continuity through oxidative phosphorylation and the AOX branch. The figure should also convey that ATP availability is asymmetric across stromal, cytosolic, and mitochondrial space rather than uniformly shared.
With that framework in place, the next question becomes methodological. If ATP is fast, compartmentalized, and physiologically selective, how can it be measured without destroying the logic that makes it meaningful?
Measuring the energy charge of plant tissues without flattening compartment biology
ATP is one of the most difficult metabolites to measure well in plants, not because the molecule is inherently elusive, but because the biological system surrounding it changes so quickly. Enzymes that consume and regenerate ATP continue operating during harvest. Mechanical handling triggers wound responses. Compartment boundaries can collapse during extraction. If sampling is slow or poorly controlled, the ATP value obtained from the instrument may partly reflect post-harvest redistribution rather than the living energetic state of the tissue.
That is why ATP measurement is never just an analytical question. It is also a quenching question, a timing question, and a spatial-fidelity question. A technically sensitive platform cannot rescue a sample whose biological state has already shifted before extraction. For plant ATP work, the first seconds after tissue excision often matter more than the last seconds of instrumental detection.
The rapid-turnover problem: why quenching strategy decides data quality
Destructive ATP workflows usually begin with one of two logic paths: aggressive quenching to arrest metabolism immediately, or solvent extraction protocols that aim to recover ATP efficiently while minimizing degradation. In practice, perchloric acid (PCA) quenching and boiling ethanol extraction are among the most discussed options.
PCA quenching is valued for speed. It halts enzymatic activity rapidly and can preserve adenylate relationships well if neutralization and cleanup are performed carefully. For ATP-focused experiments, that fast metabolic arrest is often the main attraction. The drawback is workflow severity. Harsh extraction conditions can complicate downstream handling, and small inconsistencies in neutralization or matrix cleanup can produce their own analytical noise.
Boiling ethanol represents a different compromise. It can integrate more naturally with some broader metabolite workflows and may appear operationally simpler. But operational simplicity is not the same as biological fidelity. In tissues with fast ATP turnover, the central question is whether the protocol arrests energy metabolism fast enough to preserve the state being studied. A method that works adequately for less labile metabolites may not be good enough for ATP. This is why ATP extraction cannot be treated as generic sample preparation. The extraction step is part of the biological interpretation.
For the same reason, sample handling should be viewed as an experimental variable, not a trivial pre-analytical detail. Leaf thickness, light history before sampling, harvest sequence, and delay between excision and quenching can all reshape the measured ATP state. When treatment effects are modest, these workflow differences can easily equal or exceed the biological signal.
Non-aqueous fractionation preserves spatial logic that bulk extraction destroys
Bulk extraction has a second limitation beyond timing. It merges compartments. Once the tissue is homogenized, chloroplast, mitochondrial, cytosolic, and vacuolar ATP-associated signals collapse into a single analytical pool. That may be acceptable if the only question is whether total tissue ATP changed. It is not sufficient if the real question concerns where the energetic imbalance began.
This is where non-aqueous fractionation (NAF) remains unusually valuable. NAF is designed to minimize metabolite redistribution during subcellular analysis by avoiding aqueous conditions that promote leakage and mixing after tissue disruption. In a typical NAF workflow, plant material is rapidly frozen, dried, powdered, and separated in non-aqueous density media such as heptane and tetrachloroethylene. The resulting fractions are then interpreted using compartment markers to reconstruct how metabolites were distributed across different subcellular spaces before homogenization erased that structure.
NAF is not a direct imaging method, and it does not deliver perfectly intact organelles in the classical cell-biology sense. Its power lies elsewhere. It provides a defensible bridge between chemistry and compartmentation. Instead of accepting total ATP as the only quantitative truth, NAF asks whether ATP-associated metabolites were preferentially enriched in plastidial, mitochondrial, cytosolic, or other fractions. That makes it especially useful when the biological question is about chloroplast–cytosol imbalance, respiratory compensation, or spatial redistribution under stress.
The method also forces better scientific discipline. Because NAF depends on fraction quality, marker fidelity, and statistical reconstruction, it requires the researcher to think clearly about compartment identity from the start. That is an advantage, not a burden. Too many ATP studies quietly assume that whole-tissue abundance explains mechanism. NAF makes that assumption harder to sustain.
This is exactly where targeted workflows become more informative than broad ATP screening alone. If the goal is robust quantification of adenylates and related metabolic state, Adenosine Triphosphate Analysis Solution can serve as a focused entry point. If the study expands into central carbon context or treatment-wide metabolite shifts, Targeted Metabolomics is often the better structural fit. The key is that NAF-type questions are not simply "How much ATP is present?" questions. They are "Which compartment is carrying the energetic strain?" questions.
Organelle-specific FRET biosensors changed what ATP experiments can ask
While NAF improves compartment inference after tissue disruption, live biosensors answer an entirely different class of question. Genetically encoded FRET-based sensors such as ATeam allow ATP-related dynamics to be followed inside living cells over time. This changes the purpose of the experiment. Instead of reconstructing energetic state after harvest, the researcher can monitor how ATP-associated conditions evolve in real time inside the cytosol, chloroplast, or mitochondrion.
That capability is transformative because ATP limitation is often transient before it becomes severe. Light induction, darkness transitions, oxygen shifts, heat pulses, and salt stress can all trigger fast organelle-specific ATP responses that bulk destructive workflows partly average away. A living ratio trace from a compartment-targeted sensor can therefore reveal the start of divergence long before total tissue ATP appears dramatically altered.
This does not mean FRET sensing makes extraction obsolete. Live sensors excel at kinetics, timing, and compartment contrast. They are less direct when the question is absolute concentration. Sensor behavior depends on targeting fidelity, calibration framework, local physicochemical conditions, and the relationship between fluorescence ratio and the underlying ATP or ATP/ADP state. A clean imaging dataset can still be biologically misread if the user assumes it is a direct substitute for absolute biochemical quantification in every context.
The stronger view is that destructive chemistry and live sensing answer complementary questions. Live imaging asks when ATP-related divergence appears and where it is localized. Extraction-based quantification asks how much the pool changed and whether the change holds up in orthogonal chemistry. NAF asks which compartment most plausibly carried the shift once tissue structure is analytically reconstructed. When these methods are combined, ATP becomes far more interpretable.
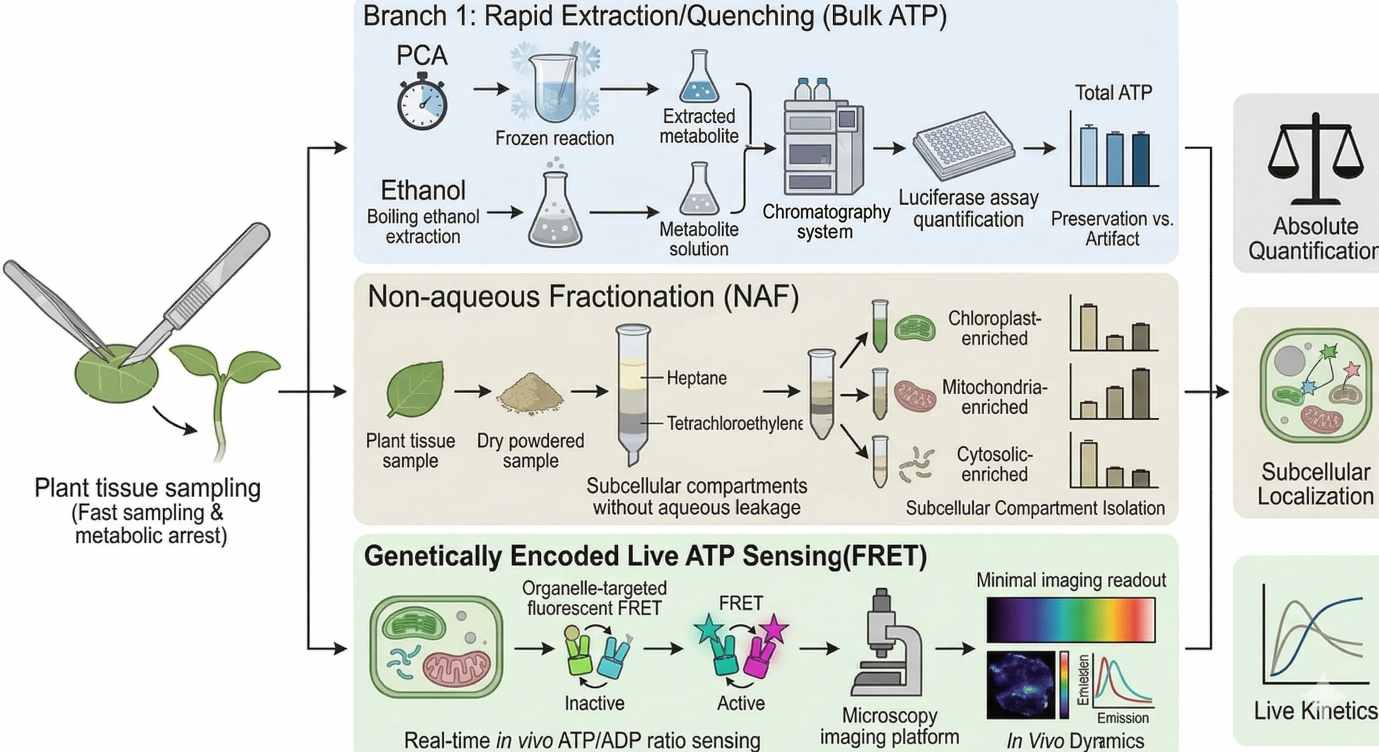 Figure 2. ATP measurement in plants requires explicit tradeoffs among quenching speed, spatial fidelity, live dynamics, and absolute quantification. Rapid chemical quenching preserves fast adenylate states, non-aqueous fractionation improves compartment inference, and FRET biosensors reveal organelle-specific kinetics that endpoint extraction cannot capture.
Figure 2. ATP measurement in plants requires explicit tradeoffs among quenching speed, spatial fidelity, live dynamics, and absolute quantification. Rapid chemical quenching preserves fast adenylate states, non-aqueous fractionation improves compartment inference, and FRET biosensors reveal organelle-specific kinetics that endpoint extraction cannot capture.
ATP-dependent carbon fixation is limited by regulatory cost as well as substrate demand
Once ATP has been generated and measured properly, the next step is to ask what ATP is actually paying for in photosynthetic carbon metabolism. The obvious answer is Calvin-cycle stoichiometry. ATP is required for the cycle to proceed. But that answer is still too shallow for modern plant bioenergetics. ATP does not only fund carbon fixation chemistry. It also funds the regulatory machinery that keeps carbon fixation competent.
This becomes clear when Rubisco activase enters the picture. Rubisco is the central carboxylase of photosynthetic carbon assimilation, but it is not automatically maintained in a catalytically open state. It can enter inhibited states that prevent efficient carbon fixation, especially under changing metabolic and environmental conditions. Rubisco activase (Rca) uses ATP hydrolysis to remodel Rubisco and restore catalytic competence. That means ATP is not merely a downstream substrate source. It is a control input to the enzyme system that determines whether carbon fixation can proceed efficiently at all.
This is a critical shift in interpretation. If stromal ATP becomes limiting, the earliest physiologically important effect may not be a dramatic drop in every Calvin-cycle intermediate. Instead, the system may begin losing the ability to keep Rubisco sufficiently activated. In that situation, carbon fixation slows because the machinery of competence is compromised before the chemistry of complete substrate exhaustion appears. ATP limitation is therefore often first visible as a regulatory problem.
That is why ATP biology cannot be separated cleanly from photosynthetic control. A chloroplast may still show active electron transport while carbon assimilation drops. On paper, that can look paradoxical. In practice, the explanation may be straightforward: photochemistry is still running, but ATP-dependent regulation of catalytic readiness is under strain. This is especially relevant under conditions where chloroplast coupling remains partially intact but ATP delivery, ATP timing, or ATP allocation no longer matches demand.
Heat stress makes ATP-dependent regulatory bottlenecks easier to see
Heat stress is a classic example. Leaves exposed to elevated temperature do not usually lose all energetic function at once. Electron transport can continue. Proton gradients can still form. Some ATP synthesis can persist. Yet productive carbon fixation declines, often earlier than a simple bulk ATP narrative would predict. One reason is that ATP-dependent regulatory proteins, especially Rubisco activase, are highly sensitive to thermal disruption.
This does not mean every heat response is a case of total uncoupling. More often the leaf enters a state of partial functional decoupling. Electron transfer, proton retention, ATP synthesis, and carbon-assimilation competence begin to drift out of alignment. The system is still energized, but not in the right way, at the right place, or at the right time. That is a more realistic description of what many plants experience under transient or moderate heat stress.
For ATP interpretation, this is a major point. A plant can retain measurable ATP and still be functionally constrained because the ATP-sensitive control nodes are the first to fail. The decisive variable is not always total ATP abundance. A more informative variable is whether ATP remains available at the most heat-sensitive regulatory steps. Once phrased that way, organelle-specific sensing and mechanism-aware metabolite analysis become much more valuable than bulk end-point chemistry alone.
ATP/ADP translocators and the night-phase redistribution problem
The light period gives the chloroplast a clear energetic role. It generates stromal ATP through photophosphorylation and uses that ATP to support carbon fixation, regulatory turnover, and chloroplast-localized metabolism. At night, that arrangement changes abruptly. Photophosphorylation stops, stromal ATP production falls, and the cell must redistribute energetic responsibility across compartments without losing metabolic continuity.
This is where ATP/ADP translocators become important, but also easy to misinterpret. It is tempting to describe them as simple channels that equalize adenylates between compartments. That description is too loose. In plants, adenylate transport is selective, context-dependent, and constrained by membrane properties, transporter identity, local ATP/ADP ratios, and the metabolic state of the compartment receiving support. The presence of a transporter does not mean free equilibration. It means the cell has a regulated route through which adenylate exchange can become favorable under particular conditions.
That distinction matters most when discussing chloroplasts at night. In mature photosynthetic chloroplasts, stromal ATP is largely produced in situ during the day. Once the light reactions stop, the chloroplast cannot simply be assumed to remain energetically buffered by the cytosol in a universal way. Instead, night-phase ATP handling depends on how starch is mobilized, how mitochondria sustain cytosolic ATP supply, how plastidial metabolism is reprioritized, and which tissues are still drawing strongly on chloroplast-derived intermediates.
This is why night-phase ATP biology should be read together with carbon allocation rather than as an isolated nucleotide problem. Starch degradation, organic acid turnover, respiratory substrate use, and adenylate exchange all become part of the same energetic negotiation. If ATP is measured without this context, the result can look deceptively simple. A nighttime ATP decline may represent pathological energy loss. It may also represent a normal redistribution pattern in which some compartments are temporarily de-emphasized while mitochondrial respiration preserves the functions that must remain continuous until dawn.
A stronger interpretation therefore asks three linked questions at night. First, which compartment is losing direct ATP production? Second, which compartment is compensating through transport or respiration? Third, which metabolic process becomes limiting before bulk ATP appears critically low? Those questions are much more informative than asking whether nighttime ATP is merely "high" or "low."
Heat stress and partial functional decoupling
Heat stress often exposes ATP problems that remain hidden under moderate conditions. The simplest description is that heat damages photosynthesis. The more accurate description is that heat gradually weakens the coordination among electron transport, proton retention, ATP synthesis, ATP-dependent regulation, and carbon-assimilation demand. In many experiments, the system does not fail at once. It drifts out of alignment.
That drift is why the phrase partial functional decoupling is useful. Complete uncoupling implies a near-total separation between electron flow and ATP production. In practice, many leaves under moderate or transient heat stress still sustain some electron transport and still produce some ATP. The deeper problem is that ATP production no longer matches the timing, amount, or location required for productive downstream metabolism. The plant remains energized, but no longer optimally synchronized.
Several mechanisms can drive this state. Thylakoid membrane properties can shift, changing proton leakage or proton retention. ATP synthase regulation can become less well matched to stromal demand. Heat can also increase the energetic cost of repair, protein turnover, and ion homeostasis, raising ATP demand even before ATP supply fully collapses. At the same time, ATP-sensitive control points such as Rubisco activase become increasingly vulnerable. The result is not one lesion but a layered mismatch.
This is why heat-stress datasets often look confusing when they rely on one endpoint measurement. A leaf may still show measurable ATP, active electron transport, and even partial pmf retention, yet display clear loss of carbon assimilation. That is not a contradiction. It is exactly what should be expected if ATP-dependent regulatory competence fails before total energy production disappears. A system can still be partially charged and still be functionally constrained.
From an experimental standpoint, heat should therefore be treated as a timing problem as much as a dose problem. Early transition points can reveal energetic divergence long before late-stage collapse is obvious. If sampling begins only after visible physiological decline, much of the decisive ATP biology has already been missed. This is where time-resolved design, compartment-aware sensing, and multi-layer metabolite analysis become especially important.
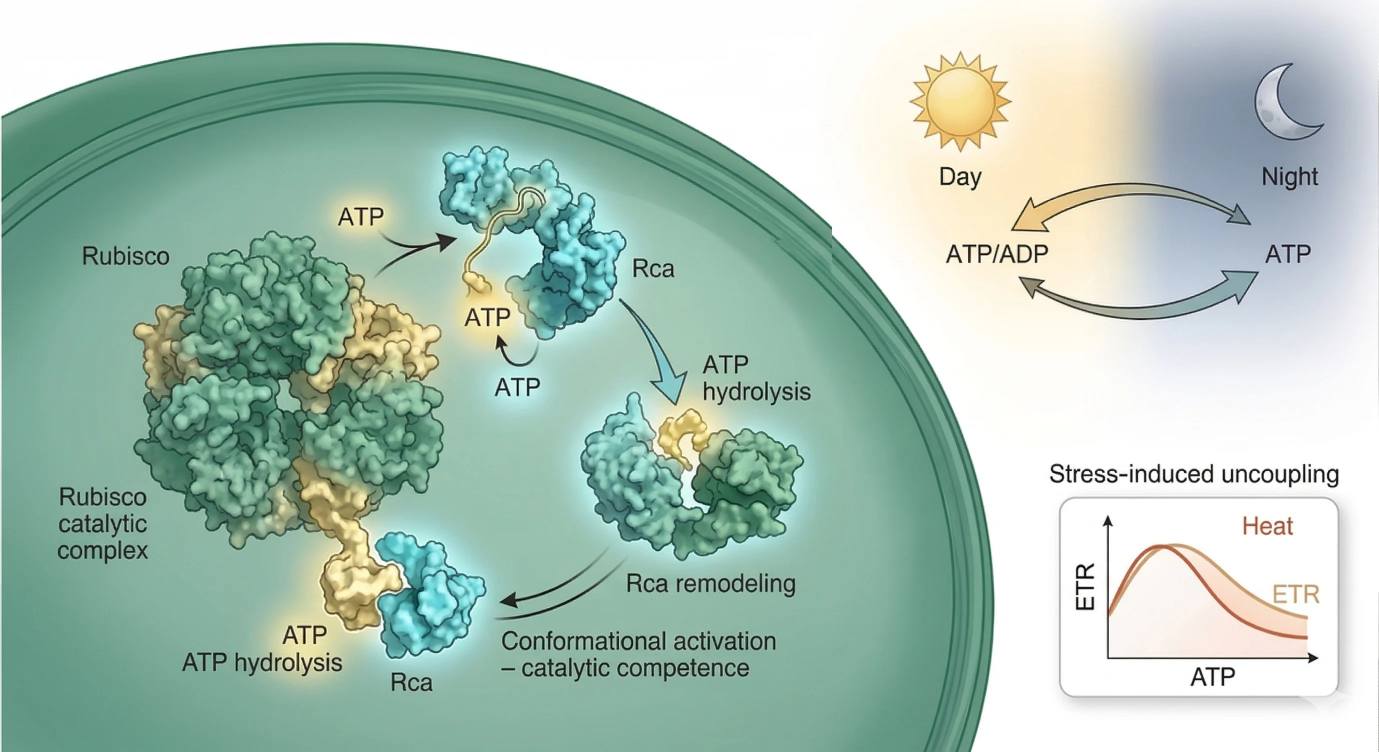 Figure 3. ATP-dependent regulatory bottlenecks in plant bioenergetics. The figure should integrate three layers: ATP hydrolysis by Rubisco activase as a control point for carbon-fixation competence, diel shifts in ATP logistics between chloroplast and extra-plastidial metabolism, and heat-induced partial functional decoupling in which electron transport, ATP production, and productive carbon assimilation drift out of alignment.
Figure 3. ATP-dependent regulatory bottlenecks in plant bioenergetics. The figure should integrate three layers: ATP hydrolysis by Rubisco activase as a control point for carbon-fixation competence, diel shifts in ATP logistics between chloroplast and extra-plastidial metabolism, and heat-induced partial functional decoupling in which electron transport, ATP production, and productive carbon assimilation drift out of alignment.
Choosing the right ATP detection method
A frequent mistake in plant ATP research is to compare detection platforms as if they are all competing to answer the same biological question. They are not. Each method preserves one layer of ATP behavior better than another. The right workflow depends on whether the experiment is asking about total abundance, adenylate balance, compartment distribution, or live kinetics.
Comparison Table: ATP Detection Methods in Plant Research
| Method | Main Readout | Strengths | Main Limitations | Best Use Case | Tissue Imaging | Absolute Quantification | Live Kinetics |
|---|---|---|---|---|---|---|---|
| Luciferase assay | Bulk ATP signal | High sensitivity, good throughput, practical for treatment screening | Strongly dependent on quenching quality, limited compartment information | Rapid ATP screening across many samples | Low | High with careful calibration | Low |
| HPLC-based adenylate profiling | ATP, ADP, and AMP separation | Strong chemical specificity, supports ATP/ADP/AMP interpretation | More demanding extraction and cleanup, destructive | Precise adenylate quantification and orthogonal validation | Low | High | Low |
| ^31P-NMR | Phosphate-containing metabolite pools in vivo | Non-destructive systems-level view, physiological context | Lower sensitivity and limited compartment resolution | Whole-tissue energetic context and phosphorus-linked metabolism | Medium | Moderate | Moderate |
| FRET-based ATP biosensors | Relative ATP/ADP-related dynamics in living compartments | Real-time measurement, organelle specificity, strong temporal resolution | Calibration and interpretation depend on sensor behavior and targeting | Light transitions, stress kinetics, compartment divergence | High | Relative rather than direct absolute | High |
| Non-aqueous fractionation with downstream quantification | Compartment-enriched metabolite distribution inference | Preserves spatial logic better than bulk extraction, useful for compartment reconstruction | Indirect, method-intensive, not live | Chloroplast–cytosol–mitochondrial partition analysis | Medium | Moderate to high with validated workflow | Low |
Which ATP question are you actually asking?
This is the decision point that improves most ATP studies. If the question is "Did total ATP change across treatments?", bulk chemical assays may be sufficient. If the question is "Did ATP/ADP/AMP balance shift?", chromatographic adenylate profiling is more informative. If the question is "Which compartment changed first?", non-aqueous fractionation or organelle-targeted biosensors become much more defensible than bulk extraction alone. If the question is "How fast did the energy state diverge during light, darkness, or heat?", live FRET sensing is usually the stronger tool.
In other words, method choice should follow the biological uncertainty. Timing problems favor live sensors. Compartment problems favor NAF-supported workflows or compartment-targeted imaging. Absolute pool questions favor carefully controlled extraction and quantitative chemistry. The most informative plant ATP studies often combine these layers instead of forcing one method to answer every question.
Why plant ATP is better understood as a flux problem
The most useful conceptual shift in current plant bioenergetics is the move away from ATP as a static stockpile. Plants do not maintain large, slowly changing ATP reserves that can be interpreted like stored fuel. They maintain ATP through rapid, ongoing cycles of production and consumption that respond continuously to environment and compartment demand. ATP is therefore better understood as a regulated flux currency.
This view changes how experimental results should be read. A late endpoint may suggest that ATP homeostasis was preserved, even though the tissue passed through a sharp transient ATP bottleneck earlier in the treatment. A strong whole-leaf ATP value may conceal a local plastidial shortage. A modest ATP decrease may prove more physiologically important than a larger one if it occurs at the wrong regulatory node or at the wrong time in the diel cycle.
Once ATP is treated as flux rather than storage, organelle-aware design becomes much easier to justify. It also becomes easier to explain why plant bioenergetics now places so much emphasis on spatiotemporal sensing, compartment-specific inference, and ATP-dependent regulatory control. These are not niche refinements. They are required if ATP is to be interpreted as a physiological driver rather than a background metabolite.
Conclusion
Plant ATP biology is not defined by one pathway or one organelle. It is defined by coordination. Chloroplasts generate ATP through photophosphorylation, but that ATP is meaningful only in relation to the proton motive force that created it, the stromal processes that consume it, and the mitochondrial functions that stabilize the rest of the cell. Mitochondria generate ATP through oxidative phosphorylation, but their importance in leaves extends beyond yield. They supply flexibility, redox continuity, and energetic buffering when chloroplast output and metabolic demand stop matching cleanly.
This is why plant ATP should be interpreted as a compartmentalized, rapidly turning regulatory flux. Its biological importance depends on where it is generated, where it is consumed, how quickly it is redistributed, and which ATP-dependent control step becomes limiting first. That is also why modern plant ATP research increasingly converges on three high-value areas: non-aqueous fractionation for compartment-aware metabolite inference, FRET-based sensing for real-time ATP/ADP dynamics, and Rubisco activase-centered regulation for understanding how ATP controls carbon-fixation competence under stress and diel change.
A strong ATP study in plants therefore does more than quantify adenylates. It matches measurement strategy to the real biological question. It distinguishes total abundance from compartmentation. It distinguishes endpoint chemistry from live kinetics. And it treats ATP not as a passive currency, but as an actively regulated constraint on photosynthetic performance.
FAQ
What is the main daytime source of ATP in photosynthetic plant cells?
During the light period, the main stromal ATP source is chloroplast photophosphorylation. However, this does not make mitochondria irrelevant. Respiratory activity remains important for extra-plastidial energy balance and for buffering metabolic mismatch.
Why is ATP in plants considered compartment-specific?
Because ATP is generated and consumed under different transport and regulatory constraints in chloroplasts, mitochondria, and the cytosol. A whole-tissue ATP value can therefore hide biologically important local limitations.
Can chloroplast ATP be freely buffered by the cytosol?
Not in a simple universal sense. Mature photosynthetic chloroplasts do not behave as if stromal ATP is freely equilibrated with the cytosol under all conditions. ATP exchange is selective and context-dependent.
What is the difference between ATP abundance and ATP compartmentation?
ATP abundance describes how much ATP is present in a pooled sample. ATP compartmentation describes how ATP-related pools are distributed across organelles or subcellular spaces. The second can be more mechanistically informative than the first.
Why is ATP difficult to measure accurately in plant tissue?
Because ATP turnover is rapid, tissue handling changes metabolism quickly, and extraction often destroys compartment information. Good ATP data depends heavily on sampling speed and quenching quality.
Why use non-aqueous fractionation in plant ATP studies?
Because it helps preserve subcellular distribution logic better than many aqueous workflows. This makes it useful when the key question is where the energetic imbalance is localized.
What do FRET-based ATP biosensors add to plant bioenergetics studies?
They provide real-time, compartment-specific insight into ATP-related dynamics in living tissue. That makes them especially useful for light transitions, stress kinetics, and organelle divergence.
Why is Rubisco activase so important in ATP biology?
Because it uses ATP hydrolysis to maintain Rubisco in a catalytically competent state. ATP therefore supports carbon fixation not only as a substrate input, but also as a regulatory requirement.
How does heat stress affect ATP use in plants?
Heat often causes partial functional decoupling. Electron transport may persist, yet ATP delivery, ATP timing, and ATP-dependent regulation become progressively less well matched to productive carbon fixation.
References
- De Col V, Fuchs P, Nietzel T, et al. ATP sensing in living plant cells reveals tissue gradients and stress dynamics of energy physiology. eLife. 2017. DOI: 10.7554/eLife.26770
- Arrivault S, Guenther M, Ivakov A, et al. Dissecting the subcellular compartmentation of proteins and metabolites in Arabidopsis leaves using non-aqueous fractionation. Molecular & Cellular Proteomics. 2014. DOI: 10.1074/mcp.M114.038190
- Shameer S, Ratcliffe RG, Sweetlove LJ. Leaf energy balance requires mitochondrial respiration and export of chloroplast NADPH in the light. Plant Physiology. 2019. DOI: 10.1104/pp.19.00624
- Voon CP, Guan X, Sun Y, et al. ATP compartmentation in plastids and the cytosol of Arabidopsis thaliana revealed by fluorescent protein sensing. Proceedings of the National Academy of Sciences of the United States of America. 2018. DOI: 10.1073/pnas.1711497115
- Yamori W, Shikanai T. Physiological functions of cyclic electron transport around photosystem I in sustaining photosynthesis and plant growth. Annual Review of Plant Biology. 2016. DOI: 10.1146/annurev-arplant-043015-112002
- Voon CP, Guan X, Sun Y, et al. ATP uptake into Arabidopsis guard cell chloroplasts drives stomatal opening. Nature Communications. 2022. DOI: 10.1038/s41467-022-28263-2
- Wang Q, Guan C, Wang P, et al. Rubisco activase: a key thermal tolerance factor in photosynthesis and a promising target for crop improvement. Journal of Experimental Botany. 2023. DOI: 10.1093/jxb/erac394
- Schwenkert S, Fernie AR, Geigenberger P, et al. Chloroplast ATP synthase: from structure to efficiency optimization in photosynthesis and growth. The Plant Cell. 2024. DOI: 10.1093/plcell/koae164
- Igamberdiev AU, Bykova NV. Role of alternative oxidase in redox and ROS signaling under stress conditions. Antioxidants. 2022. DOI: 10.3390/antiox11050884
- Gout E, Bligny R, Douce R. Regulation of intracellular pH values in higher plant cells. Carbon-13 and phosphorus-31 nuclear magnetic resonance studies. Journal of Biological Chemistry. 1988. DOI: 10.1016/S0021-9258(19)49654-3














