Glucosylceramide Analysis Service
Creative Proteomics offers a specialized glucosylceramide metabolomics analysis service that enables:
- Absolute quantification of GlcCer molecular species
- Isomer-specific resolution of GlcCer vs GalCer
- Pathway-level profiling of glycosphingolipid metabolism
- Cross-sample compatibility for cells, tissues, plasma, and exosomes
We support mechanistic studies, metabolic flux analysis, and lipid biomarker validation with high data precision and bioinformatics-ready outputs.
Submit Your Request Now
×
What You Will Receive
- Quantitative GlcCer profiles (C16–C26 chain variants)
- Raw + normalized data tables (absolute or relative units)
- Pathway heatmaps and metabolic summaries
- Isomer differentiation reports (GlcCer vs GalCer)
- Custom result interpretation + visual analytics (on request)
- What We Provide
- Advantages
- Technology Platform
- Sample Requirements
- Demo
- FAQs
- Case Study
What Is Glucosylceramide?
Glucosylceramide (GlcCer) is a fundamental glycosphingolipid consisting of a ceramide backbone linked to a glucose molecule. It contributes to membrane stability, supports lipid raft assembly, and acts as a biosynthetic precursor for more complex glycosphingolipids such as lactosylceramides and gangliosides. As a core component of the sphingolipid pathway, GlcCer plays structural and regulatory roles in a variety of cellular environments.
Its dynamic profile—determined by acyl chain length, saturation, and subcellular localization—offers valuable information for studying membrane composition, lipid-mediated signaling, and glycosylation patterns.
Why Analyze Glucosylceramide?
Analyzing GlcCer helps reveal the biochemical state of glycosphingolipid metabolism and membrane-related lipid processes. Its quantification and structural characterization can support:
- Tracking changes in sphingolipid biosynthetic or salvage pathways
- Investigating membrane composition and lipid microdomain behavior
- Monitoring cellular responses to environmental or experimental stimuli
- Differentiating isomeric species such as GlcCer and GalCer with biological relevance
- Supporting lipid flux studies and metabolic profiling in engineered systems
Glucosylceramide Analysis Service Offered by Creative Proteomics
- Quantitative Profiling of Glucosylceramide Species: Absolute and relative quantification of GlcCer with varying acyl chain lengths (e.g., C16:0, C18:0, C20:0, C24:1) using isotopically labeled internal standards.
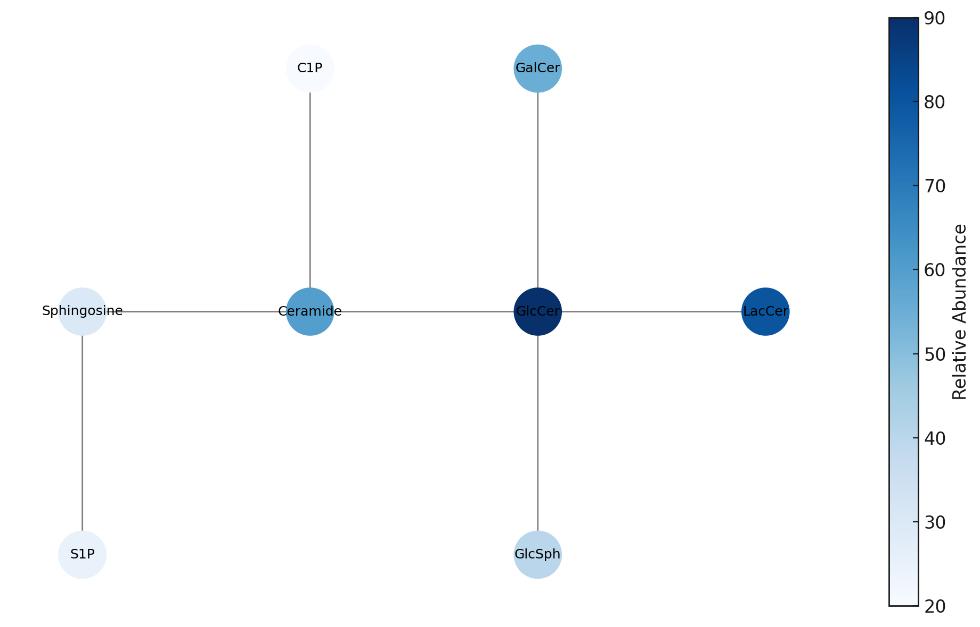
- Comprehensive Mapping of Related Metabolites: Measurement of upstream and downstream metabolites including ceramide, lactosylceramide, glucosylsphingosine, and sphingosine to characterize flux through the glycosphingolipid pathway.
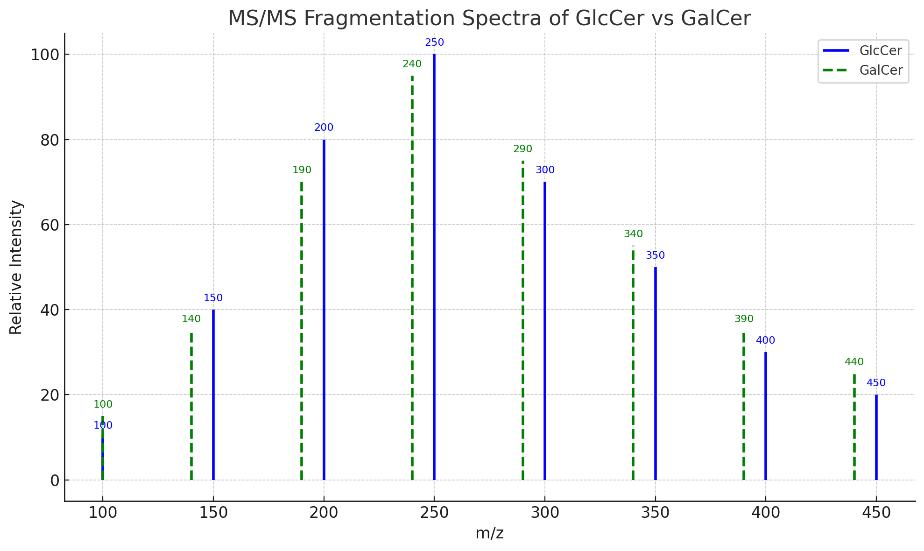
- Isomer-Specific Differentiation of Hexosylceramides: Separation and identification of GlcCer vs. GalCer using MS/MS-based fragmentation patterns and retention time indexing.
- Integrated Targeted and Untargeted Metabolomics: Simultaneous acquisition of targeted GlcCer data and broader sphingolipid profiles to support pathway enrichment and hypothesis generation.
- Bioinformatic Pathway Mapping and Flux Analysis: Annotation and integration of GlcCer data within curated metabolic networks (e.g., KEGG, HMDB) to support lipid pathway reconstruction and quantitative flux interpretation.
- Broad Sample Compatibility: Validated protocols for multiple sample types, including plasma, serum, cerebrospinal fluid (CSF), tissue homogenates, cell lysates, exosomes, and model organisms.
- Multi-Omics Integration Support: Cross-platform integration with proteomic, transcriptomic, and lipidomic datasets to enable systems-level interpretation of GlcCer-related biological processes.
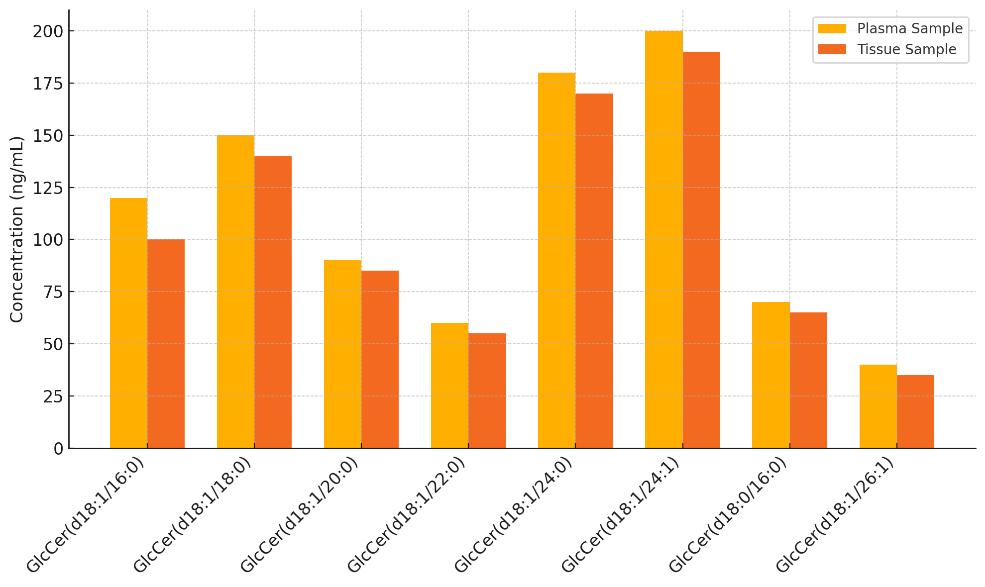
List of Detected Glucosylceramide and Related Metabolites
| Compound Name | Class | Description / Role |
|---|---|---|
| Glucosylceramides (GlcCer) | Glycosphingolipids | Primary targets with fatty acid chains such as: |
| GlcCer(d18:1/16:0) | GlcCer | Common saturated species |
| GlcCer(d18:1/18:0) | GlcCer | Abundant in mammalian tissues |
| GlcCer(d18:1/20:0) | GlcCer | Longer-chain variant |
| GlcCer(d18:1/22:0) | GlcCer | Tissue-enriched form |
| GlcCer(d18:1/24:0) | GlcCer | Major storage molecule |
| GlcCer(d18:1/24:1) | GlcCer | Unsaturated form linked to metabolic stress |
| GlcCer(d18:0/16:0) | GlcCer | Dihydro variant |
| GlcCer(d18:1/26:1) | GlcCer | Very long-chain unsaturated form |
| Glucosylsphingosine (Lyso-GlcCer) | Lyso-sphingolipid | Deacylated GlcCer; indicator of lysosomal storage dysregulation |
| Ceramides (precursors) | Sphingolipids | Core building blocks of GlcCer biosynthesis |
| Cer(d18:1/16:0) to Cer(d18:1/24:1) | Ceramides | Substrates for glucosylation |
| Cer(d18:0/24:0), Cer(d18:0/20:0) | Ceramides | Dihydroceramide forms |
| Lactosylceramides (LacCer) | Glycosphingolipids | Immediate downstream product of GlcCer |
| LacCer(d18:1/16:0) to LacCer(d18:1/24:1) | LacCer | Found in membranes and signaling vesicles |
| Hexosylceramides (total) | Glycosphingolipids | Includes both GlcCer and GalCer isomers |
| GalCer(d18:1/18:0), GalCer(d18:1/24:1) | Galactosylceramides | Differentiated by MS/MS fragmentation |
| Sphingoid Bases | Backbone lipids | Metabolites in degradation or recycling |
| Sphingosine (d18:1), Sphinganine (d18:0) | Sphingoid bases | Downstream of ceramide |
| Sphingomyelins (SM) | Phosphosphingolipids | Competing metabolic fates |
| SM(d18:1/16:0) to SM(d18:1/24:1) | Sphingomyelins | Related structural lipids |
| UDP-glucose | Nucleotide sugar | Donor for glucosylation of ceramide |
| Other Relevant Lipid Intermediates | ||
| GlcCer-phosphate | Rare intermediate | Possible under stress conditions |
| Ceramide-1-phosphate (C1P) | Signaling lipid | Inflammatory signaling molecule |
| Sphingosine-1-phosphate (S1P) | Signaling lipid | Angiogenesis and immune signaling |
Advantages of Glucosylceramide Assay
- High Sensitivity: Detection limit as low as 0.5 ng/mL for major GlcCer species using isotopically labeled standards and optimized MRM acquisition.
- Wide Quantification Range:Dynamic linear range from 0.5 ng/mL to 10 µg/mL, enabling accurate quantification in both trace-level and high-abundance samples.
- Isomer-Specific Resolution:Differentiation of GlcCer vs GalCer and other hexosylceramide isomers based on MS/MS fragmentation patterns and retention time profiles.
- Comprehensive Pathway Coverage: Simultaneous profiling of 50+ glycosphingolipid-related metabolites, including precursors (ceramides), downstream products (lactosylceramides), and degradation intermediates.
- Cross-Matrix Compatibility: Validated for use with plasma, serum, CSF, tissues, cell lysates, and exosomes, ensuring versatility across research models.
- Reproducibility and Technical Precision: Intra-assay CVs typically below 10%, with external QC materials applied for batch consistency.
Workflow for Glucosylceramide Analysis Service

Technology Platform for Glucosylceramide Analysis Service
Mass Spectrometry
SCIEX QTRAP® 6500+ LC-MS/MS
Utilized for high-sensitivity targeted quantification of GlcCer species with multiple reaction monitoring (MRM) and isotope-labeled internal standards.
Thermo Scientific™ Q Exactive™ HF Orbitrap
Deployed for untargeted and semi-targeted profiling of GlcCer and related sphingolipids, offering high-resolution accurate mass detection (HRAM) and isomer differentiation.
Chromatographic Separation
Waters ACQUITY UPLC System
Equipped with BEH Amide and C18 columns for HILIC and reverse-phase separation, optimized for glycosphingolipid retention and resolution.
Sample Requirements for Glucosylceramide Analysis Service
| Sample Type | Minimum Volume/Weight | Storage Condition | Notes |
|---|---|---|---|
| Fruit Juice / Wine | ≥ 1.0 mL | 4°C (short-term), -20°C (long-term) | Filtered; avoid preservatives if possible |
| Fermentation Broth | ≥ 0.5 mL | -20°C | Centrifuge to remove cells/debris; provide fermentation stage info |
| Plant Tissue (Fresh) | ≥ 100 mg | Flash-frozen at -80°C | Indicate species, part (e.g., leaf, fruit), and growth condition |
| Plant Tissue (Dry) | ≥ 50 mg | Room temp (sealed, dry) | Grind to fine powder recommended |
| Plasma / Serum | ≥ 200 µL | -80°C | EDTA preferred; avoid hemolysis |
| Food Product (Solid) | ≥ 2.0 g | 4°C or -20°C | Homogenized sample preferred; indicate composition |
| Standard Solution | ≥ 0.5 mL (optional) | 4°C | If customer-prepared standards are supplied |
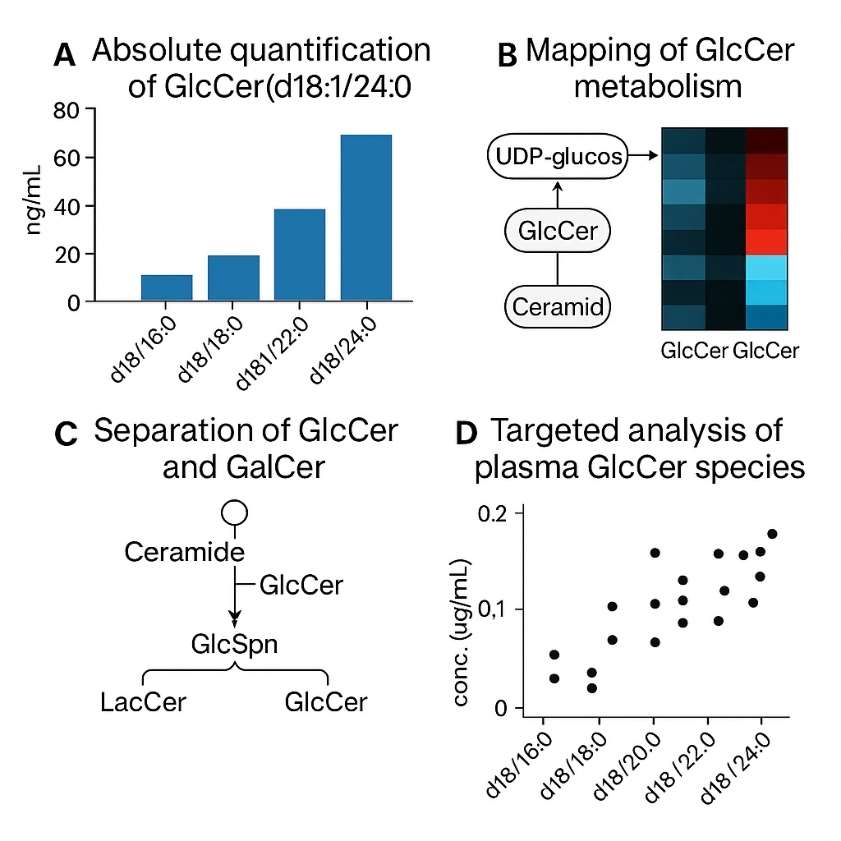
Demo Results
FAQ of Glucosylceramide Analysis Service
Can I submit different types of samples within the same project?
Yes. We accept mixed sample types (e.g., plasma, tissue, cells) within the same project. However, matrix effects and normalization strategies may differ—please indicate sample type clearly on submission.
Do I need to add internal standards before sending samples?
No. Internal standards are added during sample preparation at our facility. If you've already spiked standards, please inform us in advance to avoid overestimation or interference.
Does hemolysis or lipemia affect GlcCer quantification?
Yes. Hemolyzed or lipemic samples may introduce background noise or interfere with accurate GlcCer profiling. Please avoid hemolysis during collection and note any visible sample quality issues.
Can you work with low-abundance or precious samples?
Yes. Our platform supports high-sensitivity detection. If your sample is limited in volume or quantity, contact us—we can evaluate feasibility and adjust extraction protocols accordingly.
How should I prepare and ship tissue or cell pellet samples?
Flash-freeze immediately after collection, store at −80°C, and ship on dry ice. For cell pellets, wash with cold PBS before freezing. Include protein or wet weight data if available.
Will I receive information on related metabolites beyond GlcCer?
Yes. In addition to GlcCer species, our analysis provides data on ceramides, lactosylceramides, glucosylsphingosine, and sphingoid bases—enabling interpretation across the sphingolipid pathway.
How do you ensure separation of isobaric species like GlcCer(d18:1/18:1) vs GalCer(d18:1/18:1)?
We use retention time indexing with validated reference standards and MS/MS fragmentation patterns to distinguish GlcCer from GalCer and other isobars.
What kind of normalization or quantification approach do you use?
We apply isotope-labeled internal standards for absolute quantification when available, and normalize across sample input or protein content to ensure data comparability.
Can I integrate this lipidomics data with my proteomics or transcriptomics dataset?
Yes. We offer exportable, normalized metabolite matrices suitable for multi-omics integration. Bioinformatics support is also available if needed.
Will the result file include both normalized and raw ion intensity data?
Yes. Our reports include both raw peak areas and normalized concentrations, along with calibration curve performance and QC metrics.
Learn about other Q&A.
Glucosylceramide Analysis Service Case Study

Title: Glucosylceramide is essential for Heartland and Dabie bandavirus glycoprotein-induced membrane fusion
Journal: PLOS Pathogens
Published: 2023
- Background
- Methods
- Results
- Reference
Heartland virus (HRTV) and Dabie bandavirus (DBV) are emerging tick-borne pathogens associated with hemorrhagic fever and high mortality. These viruses rely on Gn/Gc glycoproteins for entry into host cells, yet the precise host lipid components enabling this fusion process have remained unclear.
Sphingolipids—especially glycosphingolipids such as glucosylceramide (GlcCer)—are known to play regulatory roles in membrane structure and viral lifecycle processes. This study investigates whether GlcCer is specifically required for viral membrane fusion and entry.
The authors used genome-wide CRISPR-Cas9 screening in human 293T cells to identify host dependency factors for HRTV infection. Hits from the screen were validated through:
- Targeted gene knockouts (UGCG, SPTLC2, UGT8, B4GALT5/6)
- Chemical inhibition of GlcCer synthesis (e.g., NB-DNJ)
- Quantitative lipidomics analysis using supercritical fluid chromatography–tandem mass spectrometry (SFC-MS/MS)
- Exogenous lipid rescue experiments with GlcCer, GalCer, and LacCer
- Membrane fusion assays and protein-lipid binding studies (SPR, BLI)
Creative Proteomics' Contribution:
The study's reliance on quantitative glycosphingolipid profiling, especially absolute GlcCer quantification and metabolic flux interpretation, demonstrates the critical role of:
- Targeted Glucosylceramide LC-MS/MS analysis
- Differentiation of GlcCer vs GalCer species
- Pathway-level lipidomics of ceramide–GlcCer–LacCer axis
Creative Proteomics can support such research through:
- High-sensitivity detection of GlcCer and related metabolites
- Sample compatibility with cell lysates, knockout models, and viral systems
- Quantification of metabolic changes upon gene editing or drug treatment
- The CRISPR screen identified multiple genes in the sphingolipid biosynthesis pathway (e.g., UGCG, SPTLC2) as essential for HRTV infection.
- Genetic or chemical inhibition of UGCG (GlcCer synthase) significantly reduced viral infectivity.
- Supplementation with GlcCer, but not GalCer or LacCer, rescued viral entry in UGCG-deficient cells.
- SFC-MS/MS lipidomics showed GlcCer depletion upon NB-DNJ treatment or gene knockout.
- Biophysical assays confirmed direct binding of viral Gc protein to GlcCer, enabling membrane insertion and fusion.
- Mutation of D841 in the Gc lipid-binding pocket abolished GlcCer interaction and viral entry.
- Downstream glycosphingolipids (e.g., LacCer) were not required for infection; instead, GlcCer accumulation correlated with enhanced infectivity.
 Glucosylceramide (GlcCer) is essential for HRTV infection efficiency.
Glucosylceramide (GlcCer) is essential for HRTV infection efficiency.
(A–B) Knockout of SPTLC2 or UGCG significantly reduces HRTV replication in 293T cells, as shown by qPCR. (C) Heatmap displaying intracellular GlcCer and GalCer levels across knockout lines. (D–E) Infectivity assays (TCID₅₀) show marked reduction in viral titer in UGCG- and SPTLC2-deficient cells compared to controls.
Reference
- Xia, Tian, et al. "Glucosylceramide is essential for Heartland and Dabie bandavirus glycoprotein-induced membrane fusion." PLoS pathogens 19.3 (2023): e1011232. https://doi.org/10.1371/journal.ppat.1011232
Publications
Here are some of the lipidomics-related papers published by our clients:

- White matter lipid alterations during aging in the rhesus monkey brain. 2024. https://doi.org/10.1007/s11357-024-01353-3
- Characterization of Dnajc12 knockout mice, a model of hypodopaminergia. 2024. https://doi.org/10.1101/2024.07.06.602343
- Annexin A2 modulates phospholipid membrane composition upstream of Arp2 to control angiogenic sprout initiation. 2023. https://doi.org/10.1096/fj.202201088R
- Lipid Membrane Engineering for Biotechnology (Doctoral dissertation, Aston University). 2023. https://doi.org/10.48780/publications.aston.ac.uk.00046663
- Summative and ultimate analysis of live leaves from southern US forest plants for use in fire modeling. 2020. https://doi.org/10.1152/ajpgi.00184.2023