Quinolones Analysis Service
Creative Proteomics offers high-sensitivity quinolones analysis services using advanced LC-MS/MS and Orbitrap platforms to accurately detect and quantify over 25 quinolones and 35 metabolites in biological, food, and environmental samples. Our services help clients monitor drug residues, study pharmacokinetics, and assess environmental impact with fast turnaround, low detection limits (as low as 0.1 ng/mL), and customizable analysis panels—ensuring reliable data for safety, compliance, and research needs.
Submit Your Request Now
×- What We Provide
- Advantage
- Workflow
- Technology Platforms
- Sample Requirements
- FAQ
- Publication
What are Quinolones?
Quinolones are synthetic, broad-spectrum antimicrobial agents that belong to the class of drugs known as fluoroquinolones. These agents are commonly used to treat bacterial infections by inhibiting bacterial DNA gyrase and topoisomerase IV, enzymes critical to bacterial DNA replication. The chemical structure of quinolones allows them to penetrate bacterial cells, thereby halting bacterial reproduction.
Quinolones are highly effective against a range of pathogens, including those responsible for respiratory, urinary tract, and gastrointestinal infections. Some of the most commonly used quinolones include ciprofloxacin, levofloxacin, and ofloxacin. These drugs are utilized in both human medicine and veterinary applications, as well as in agriculture to prevent bacterial infections in livestock.
However, the widespread use of quinolones has raised concerns over antibiotic resistance and their persistence in the environment, which has prompted rigorous analysis and monitoring. As a result, understanding the pharmacokinetics and degradation pathways of quinolones is essential for mitigating potential risks associated with their use.
Quinolones Analysis Service Offered by Creative Proteomics
- Quantitative Determination of Quinolones: Accurate quantification of multiple quinolone compounds in complex matrices such as plasma, urine, tissue, food, feed, aquaculture products, and environmental samples, using advanced LC-MS/MS or Orbitrap-based platforms.
- Multi-Residue Screening for Veterinary and Environmental Samples: Simultaneous detection of multiple quinolones and their metabolites in livestock, poultry, fish, and environmental matrices including soil, sediment, and surface water.
- Metabolite Profiling and Identification: Comprehensive analysis of both parent drugs and their key phase I (e.g. hydroxylation, demethylation) and phase II (e.g. glucuronidation, sulfation) metabolites to support biotransformation and residue studies.
- Degradation and Stability Studies: Assessment of quinolone stability under varying conditions such as light, pH, temperature, and storage, suitable for formulation, residue dissipation, and environmental persistence evaluations.
- Pharmacokinetic and Exposure Studies: Support for time-course studies by quantifying quinolone concentrations in biological matrices at different intervals, ideal for exposure modeling or residue withdrawal evaluation.
List of Detected Quinolones and Related Metabolites
| Parent Quinolone | Analyzable Metabolites |
|---|---|
| Ciprofloxacin | Desmethylciprofloxacin, Ciprofloxacin glucuronide, Ciprofloxacin N-oxide |
| Levofloxacin | Desmethyllevofloxacin, Levofloxacin glucuronide, Levofloxacin N-oxide |
| Ofloxacin | Desmethylofloxacin, Ofloxacin glucuronide, Ofloxacin N-oxide |
| Norfloxacin | Norfloxacin glucuronide, Norfloxacin N-oxide, Hydroxynorfloxacin |
| Moxifloxacin | Moxifloxacin N-sulfate, Moxifloxacin glucuronide, Moxifloxacin N-oxide |
| Enrofloxacin | Ciprofloxacin (active metabolite), Enrofloxacin glucuronide |
| Danofloxacin | Hydroxydanofloxacin, Desmethyldanofloxacin |
| Lomefloxacin | Desmethyllomefloxacin, Lomefloxacin glucuronide |
| Marbofloxacin | Desmethylmarbofloxacin, Hydroxymarbofloxacin |
| Sarafloxacin | Desmethylsarafloxacin, Hydroxysarafloxacin |
| Gatifloxacin | Gatifloxacin glucuronide, Hydroxygatifloxacin |
| Trovafloxacin | Trovafloxacin glucuronide, Trovafloxacin carboxylic acid |
| Sparfloxacin | Desmethylsparfloxacin, Sparfloxacin glucuronide |
| Prulifloxacin | Ulifloxacin (active metabolite), Prulifloxacin glucuronide |
| Balofloxacin | Balofloxacin N-oxide, Balofloxacin glucuronide |
| Gemifloxacin | Gemifloxacin N-oxide, Hydroxygemifloxacin |
| Fleroxacin | Desmethylfleroxacin, Fleroxacin glucuronide |
Advantages of Quinolone Assay
- Ultra-Low Detection Limits: Leveraging advanced LC-MS/MS platforms (Agilent 6495, Thermo Q Exactive), we achieve detection limits as low as 0.1 ng/mL, ensuring reliable quantification of trace-level residues in complex matrices.
- Extensive Compound Coverage: Simultaneous detection of 25+ quinolones and over 35 key metabolites allows for comprehensive residue and metabolite profiling in a single run.
- Excellent Linearity and Precision: All compounds are quantified with R² ≥ 0.999, and both intra- and inter-day precision consistently fall below 10% RSD, ensuring accurate, reproducible results.
- High Recovery and Accuracy: Recovery rates range from 85% to 110% across biological and environmental matrices, supporting dependable quantification in real-world samples.
- Minimal Sample Volume: Efficient protocols require only 100 µL plasma, 0.5 g tissue, or 20 mL water, minimizing sample demands while maintaining analytical integrity.
- Customizable Analysis Panels: Targeted panels can be tailored for veterinary, aquaculture, or environmental applications, with built-in support for global MRL compliance.
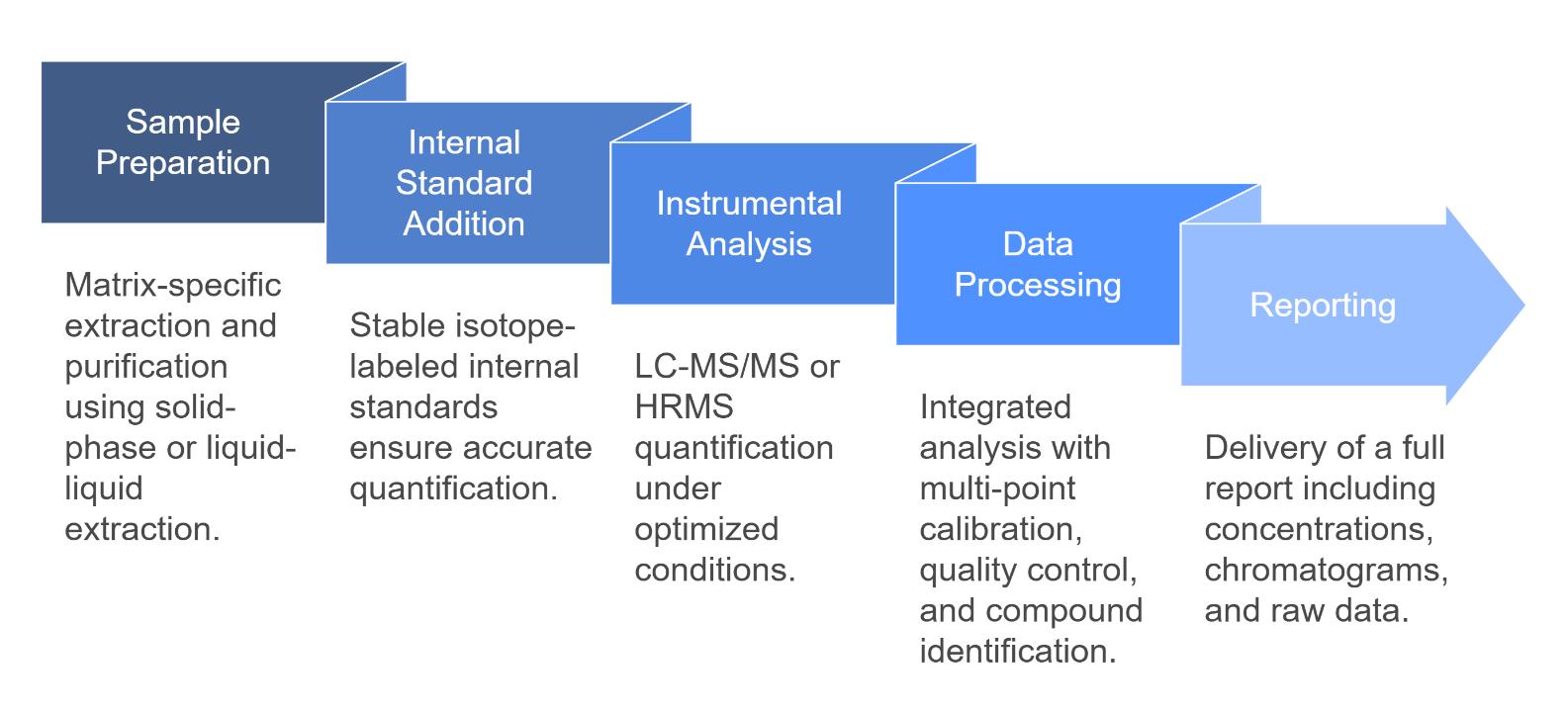
Workflow for Quinolone Analysis Service
Technology Platform for Quinolone Analysis Service
Agilent 1290 Infinity II UHPLC coupled with Agilent 6495C Triple Quadrupole LC-MS/MS – ideal for routine, high-throughput quantification.
Thermo Scientific Vanquish UHPLC paired with Q Exactive Plus Orbitrap HRMS – for high-resolution, full-scan screening and metabolite identification.
Optimized ESI (Electrospray Ionization) in positive mode, MRM or full scan/dd-MS² modes depending on project needs.

Agilent 1260 Infinity II HPLC (Figure from Agilent)

Q Exactive™ UHMR Hybrid Quadrupole Orbitrap™ Mass Spectrometer (Figure from Thermo Fisher)
Sample Requirements for Quinolones Analysis Service
| Sample Type | Required Volume | Preparation Guidelines |
|---|---|---|
| Blood/Plasma | 1 mL | Collect in EDTA or heparin tubes, refrigerate if not analyzed immediately |
| Urine | 5 mL | Store at -20°C for long-term storage |
| Tissue (muscle, liver) | 0.5 g | Flash freeze immediately after collection |
| Soil/Sediment | 10 g | Dry and grind to a fine powder before analysis |
| Water | 50 mL | Filter and store at 4°C |
Applications of Quinolone Assay Service
Pharmaceutical Industry
Quantification of quinolone residues in drugs for regulatory compliance and safety studies.
Agricultural Sector
Monitoring quinolone levels in crops, livestock, and veterinary products to ensure compliance with pesticide residue limits.
Environmental Studies
Tracking quinolone contamination in water, soil, and sediment to evaluate environmental pollution and sustainability.
Research and Development
Studying the metabolism, pharmacokinetics, and ecological impact of quinolones in various biological systems.
Demo

Chromatogram and mass spectra of fluoroquinolones in sewage hospital water using SPE-MD-LC-MS = MS procedure (Montesdeoca-Esponda et al., 2012).
FAQ of Quinolone Analysis Service
How many quinolones can be detected in one analysis?
Our platform supports simultaneous detection of 25+ quinolones and over 35 validated metabolites in a single run, depending on the selected panel.
Do you provide raw data and chromatograms along with the report?
Yes. Upon completion, clients receive a full analytical report, including quantitative results, calibration curves, chromatograms, and raw data files in your preferred format.
What analytical instruments do you use for quinolone testing?
We primarily use Agilent 6495 Triple Quadrupole LC-MS/MS and Thermo Scientific Q Exactive Orbitrap systems for high-sensitivity, high-resolution analysis.
Can your method detect metabolites of quinolones?
Yes. We offer comprehensive metabolite profiling, including common phase I and phase II quinolone metabolites, using validated reference standards and high-resolution MS.
Are stability or storage condition studies available?
Absolutely. We can design stability and degradation studies under various conditions (e.g., pH, temperature, light) to support formulation research or environmental persistence studies.
What concentration range can you accurately quantify?
Our methods support quantification from 0.1 ng/mL up to 1000 ng/mL, depending on the compound and matrix. Calibration ranges are adjusted to fit your sample's expected levels.
Learn about other Q&A.
Publications
Here are some publications in Metabolomics research from our clients:

- Bacterial–fungal interactions revealed by genome-wide analysis of bacterial mutant fitness. 2021. https://doi.org/10.1038/s41564-020-00800-z
- Disruption of CYCLOPHILIN 38 function reveals a photosynthesis-dependent systemic signal controlling lateral root emergence. 2020. https://doi.org/10.1101/2020.03.11.985820
- Sex modifies the impact of type 2 diabetes mellitus on the murine whole brain metabolome. 2023. https://doi.org/10.3390/metabo13091012
- Enhance trial: effects of NAD3® on hallmarks of aging and clinical endpoints of health in middle aged adults: a subset analysis focused on blood cell NAD+ concentrations and lipid metabolism. 2022. https://doi.org/10.3390/physiologia2010002
- NUDT22 promotes cancer growth through pyrimidine salvage and the TCA cycle. 2022. https://doi.org/10.21203/rs.3.rs-1491465/v1
Reference
- Montesdeoca-Esponda, Sarah, Zoraida Sosa-Ferrera, and José Juan Santana-Rodríguez. "Comparison of solid phase extraction using micellar desorption combined with LC-FD and LC-MS/MS in the determination of antibiotics fluoroquinolone residues in sewage samples." Journal of liquid chromatography & related technologies 35.14 (2012): 2081-2096. https://doi.org/10.1080/10826076.2011.627622







