8-Hydroxydeoxyguanosine Analysis Service
Creative Proteomics specializes in advanced 8-hydroxy-2'-deoxyguanosine (8-OHdG) analysis to assess oxidative DNA damage, a key biomarker for aging, cancer, and cardiovascular diseases. Using cutting-edge technologies like UHPLC-MS/MS, HPLC/EC, and ELISA, we provide precise quantification of 8-OHdG in biological samples such as urine, serum, and tissues. Our services support researchers and industries in studying genomic instability, evaluating therapeutic efficacy, and conducting safety evaluations, oxidative damage research, and environmental toxin monitoring.
Submit Your Request Now
×- What We Provide
- Advantage
- Workflow
- Sample Requirements
- FAQ
- Publication
What is 8-Hydroxydeoxyguanosine?
8-Hydroxydeoxyguanosine (8-OHdG) is a predominant marker of oxidative DNA damage and one of the most reliable indicators of oxidative stress in cells and tissues. Formed through the hydroxylation of the guanine base at the C8 position in DNA, 8-OHdG is released into biological fluids upon DNA repair and can be quantified in urine, plasma, serum, and tissue samples. It serves as a valuable biomarker in environmental studies, toxicology, aging research, and biomonitoring of oxidative damage resulting from radiation, chemical exposure, and lifestyle factors.
8-Hydroxydeoxyguanosine Analysis Service Offered by Creative Proteomics
Quantitative 8-OHdG Detection: Absolute quantification of 8-OHdG using UHPLC-MS/MS with isotopically labeled internal standards. Ultra-sensitive detection in urine, plasma, serum, tissue, and DNA extracts.
8-OHdG Profiling in Tissue and Cell Samples: Analysis of DNA oxidation levels in fresh, frozen, or cultured tissue and cell lines. Supports DNA repair and oxidative stress mechanism studies.
Multi-Matrix Compatibility Services: Validated protocols for urine, serum, plasma, cerebrospinal fluid (CSF), tissue homogenates, and cell lysates. Optional normalization using creatinine or DNA content (depending on matrix).
Targeted Oxidative Damage Panels: Simultaneous quantification of 8-OHdG and related markers such as 8-oxoguanine, 8-oxoGuanosine, and deoxyguanosine. Ideal for evaluating oxidative stress response and antioxidant efficacy.
Time-Course Oxidative Stress Monitoring: Longitudinal studies tracking 8-OHdG levels over time in biological systems. Supports pharmacodynamic and toxicokinetic modeling.
Custom Panel Design & Pathway Integration: Customizable metabolite panels to include nucleic acid oxidation and DNA repair intermediates. Integration with metabolomics and transcriptomics data for pathway-level insights.
Data Analysis & Bioinformatics Support: Raw data processing, statistical analysis, and biological interpretation. Optional pathway mapping for oxidative DNA damage and repair mechanisms.
List of Detected 8-Hydroxydeoxyguanosines and Related Metabolites
| Category | Analyte Name | Associated Pathway | Detection Method |
|---|---|---|---|
| DNA Oxidation Product | 8-Hydroxy-2'-deoxyguanosine (8-OHdG) | DNA oxidative damage, repair | UHPLC-MS/MS, HPLC-ECD |
| Oxidized Nucleotide Base | 8-Oxoguanine | Base excision repair (BER) | UHPLC-MS/MS |
| RNA Oxidation Product | 8-Oxoguanosine (8-oxoG) | RNA oxidation pathway | UHPLC-MS/MS |
| DNA Nucleoside | 2'-Deoxyguanosine | Purine metabolism | UHPLC-MS/MS |
| Guanine Oxidation Product | FapyGua (2,6-Diamino-4-hydroxy-5-formamidopyrimidine) | Ionizing radiation response | LC-MS/MS |
| Deamination Product | Xanthine | Purine catabolism | LC-MS/MS |
| Oxidized Nucleoside | 8-Nitroguanine | Nitrosative stress, inflammatory response | LC-MS/MS |
| DNA Repair Intermediate | 5-Hydroxycytosine | Oxidative demethylation | LC-MS/MS |
| Oxidized Adenine Derivative | 8-Hydroxyadenine | Oxidative DNA base damage | LC-MS/MS |
| Urinary Marker | 8-OxodGuo (urinary 8-OHdG) | Systemic oxidative stress marker | UHPLC-MS/MS |
Advantages of 8-Hydroxydeoxyguanosine Assay
- Precision & Reproducibility: All quantifications are performed with a coefficient of variation (CV) below 10%, ensuring high reliability and reproducibility of data across replicates and sample batches.
- Superior Sensitivity: Utilizing UHPLC-MS/MS technology, our platform achieves detection limits as low as 0.09 μg/L for 8-OHdG and 0.04 μg/L for related oxidative DNA damage metabolites—enabling detection even in trace-level samples.
- High Throughput Metabolomics Integration: Capable of simultaneous profiling of over 500 endogenous metabolites, including oxidized nucleotides, base excision repair intermediates, and purine metabolism products—ideal for systems biology and oxidative stress network analysis.
- Matrix Versatility: Our methods are validated across a comprehensive range of biological matrices.
- Internal Standard Normalization: All quantifications employ stable isotope-labeled internal standards, significantly improving data accuracy and compensating for matrix effects.
- End-to-End Data Transparency: All results are reported with raw chromatograms, standard curves, and quantification metrics for full scientific traceability.
Workflow for 8-OHdG Analysis Service
A. Sample Preparation: Snap-freezing in liquid nitrogen to preserve oxidative integrity.
B. Metabolite Extraction: Solid-phase extraction (SPE) or protein precipitation for clean analyte isolation.
C. Chromatographic Separation:
- UHPLC: Agilent 1290 Infinity II system with C18 columns.
- GC-MS: Thermo Scientific TRACE 1310 for volatile derivatives.
D. Detection:
- MS/MS: SCIEX Triple Quad 6500+ for MRM-based quantification.
- Electrochemical Detection: For low-abundance urinary 8-OHdG.
E. Data Analysis: Skyline or XCMS for metabolite identification and pathway mapping.

Agilent 1260 Infinity II HPLC (Figure from Agilent)

RACE 1310 (Figure from Thermo)

SCIEX Triple Quad™ 6500+ (Figure from Sciex)
Sample Requirements for 8-Hydroxydeoxyguanosine Analysis Service
| Sample Type | Required Volume/Weight | Storage Condition |
|---|---|---|
| Urine | ≥ 1 mL | -80°C |
| Plasma/Serum | ≥ 500 µL | -80°C |
| Tissue | ≥ 100 mg | Flash frozen in liquid nitrogen, -80°C |
| Cultured Cells | ≥ 1 × 10⁶ cells | Cell pellets, -80°C |
| DNA Extracts | ≥ 1 µg | TE buffer, -20°C |
Applications of 8-Hydroxydeoxyguanosine Assay Service
Environmental Toxicology Research
Evaluate oxidative DNA damage caused by exposure to environmental pollutants, heavy metals, and airborne particulates in ecological and human biomonitoring studies.
Pharmacological and Chemical Safety Testing
Assess genotoxic and oxidative stress-inducing potential of pharmaceutical compounds, industrial chemicals, or novel materials during preclinical safety evaluation.
Nutritional and Antioxidant Research
Quantify changes in oxidative stress biomarkers, such as 8-OHdG, in response to dietary interventions, functional foods, and antioxidant supplementation studies.
Aging and Longevity Research
Monitor the accumulation of DNA oxidative damage as a biomarker of cellular aging and mitochondrial dysfunction across tissue or organism models.
Occupational Exposure Monitoring
Track oxidative DNA lesions in individuals exposed to industrial hazards (e.g., solvents, radiation, heavy metals) to support occupational health risk assessment.
Oxidative Stress Mechanism Studies
Support mechanistic research into ROS-mediated cellular damage and DNA repair pathways in cell-based systems, in vitro models, or animal studies.
Demo
FAQ of 8-Hydroxydeoxyguanosine Analysis Service
What types of samples can be submitted for 8-OHdG analysis?
We accept a wide range of biological samples including urine, serum, plasma, cerebrospinal fluid (CSF), tissue homogenates, cultured cells, and purified DNA extracts. Please refer to our sample requirements table for specific volume and storage conditions.
Can I submit frozen samples?
Yes, frozen samples are preferred for preserving analyte stability. We recommend storing and shipping samples at -80°C with dry ice to minimize degradation.
What is the recommended sample volume for accurate quantification?
For optimal results, we recommend at least 1 mL of urine, 500 µL of serum or plasma, or 100 mg of tissue. DNA extracts should contain at least 1 µg of DNA.
What is the typical turnaround time for analysis?
Standard turnaround time is 10–15 business days from the date of sample receipt. For large-scale studies or custom projects, we provide estimated timelines upon consultation.
How should samples be labeled and packaged for shipping?
All samples should be clearly labeled with unique identifiers. Use leak-proof, sterile containers and ship in dry ice with appropriate cushioning. A completed sample submission form must be included.
Is creatinine normalization provided for urinary 8-OHdG analysis?
Yes, we offer optional creatinine quantification to normalize urinary 8-OHdG levels, allowing for more accurate comparisons between samples.
Do you provide data in a publication-ready format?
Absolutely. All reports include raw data, quantitative results, calibration curves, internal standard details, and figures suitable for publication or presentations.
Can you analyze 8-OHdG in species other than human?
Yes. Our methods are suitable for multiple species including rodent, primate, and model organisms. Species-specific validation can be provided upon request.
Is it possible to analyze 8-OHdG in DNA isolated from FFPE samples?
Yes, but FFPE-derived DNA often requires additional preparation due to cross-linking. We recommend contacting us in advance for FFPE-specific protocols.
Can I request only data acquisition without full analysis?
Yes. We offer flexible service packages including raw data-only options, data acquisition with basic quantification, or full bioinformatics interpretation.
Are replicate measurements included in the service?
Technical replicates can be included upon request. Biological replicates should be prepared and submitted by the client as separate samples.
What QC measures are included in the analysis?
Each batch includes blanks, quality control standards, internal standards, and pooled matrix controls to ensure analytical reliability.
Learn about other Q&A.
8-Hydroxydeoxyguanosine Analysis Service Case Study
Publications
Here are some publications in Metabolomics research from our clients:

- Disruption of CYCLOPHILIN 38 function reveals a photosynthesis-dependent systemic signal controlling lateral root emergence. 2020. https://doi.org/10.1101/2020.03.11.985820
- Methyl donor supplementation reduces phospho‐Tau, Fyn and demethylated protein phosphatase 2A levels and mitigates learning and motor deficits in a mouse model of tauopathy. 2023. https://doi.org/10.1111/nan.12931
- Pan-lysyl oxidase inhibition disrupts fibroinflammatory tumor stroma, rendering cholangiocarcinoma susceptible to chemotherapy. 2024. https://doi.org/10.1097/HC9.0000000000000502
- UDP-Glucose/P2Y14 Receptor Signaling Exacerbates Neuronal Apoptosis After Subarachnoid Hemorrhage in Rats. 2024. https://doi.org/10.1161/STROKEAHA.123.044422
- Untargeted metabolomics reveal sex-specific and non-specific redox-modulating metabolites in kidneys following binge drinking. 2023. https://doi.org/10.1530/REM-23-0005
References
- Guo, Cheng, et al. "Association between oxidative DNA damage and risk of colorectal cancer: sensitive determination of urinary 8-hydroxy-2′-deoxyguanosine by UPLC-MS/MS analysis." Scientific reports 6.1 (2016): 1-9. https://doi.org/10.1038/srep32581
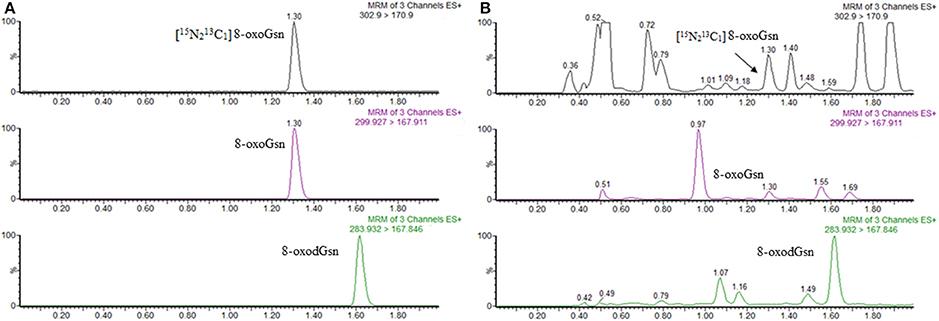
- Gan, Wei, et al. "Urinary 8-oxo-7, 8-dihydroguanosine as a potential biomarker of aging." Frontiers in aging neuroscience 10 (2018): 34. https://doi.org/10.3389/fnagi.2018.00034

![(a) Representative chromatograms from human urine sample displaying internal standard [15N5]8-OHdG (m/z 289.1 > 173.0), 8-OHdG (m/z 284.1 > 168.0) and corresponding qualifier ion (m/z 284.1 > 117.0). (b) Representative chromatograms of internal standard and 8-OHdG standard.](/upload/image/pic-8-hydroxydeoxyguanosine-analysis-service-4.jpg)