- Service Details
- FAQ
- Demo
- Case Study
It is generally believed that any process capable of inducing changes in the native conformation of a protein without involving peptide bond cleavage is called denaturation. The denaturation of proteins caused by heating is referred to as thermal denaturation. Denaturation leads to changes in physical properties, chemical characteristics, and biological functions of proteins. The diverse biological functions exhibited by proteins in life activities entirely depend on their specific conformations. Once these specific conformations are disrupted, their biological functions will be lost. Therefore, studying the thermal denaturation patterns of proteins under different conditions is of significant importance. Differential Scanning Calorimetry (DSC) can directly provide temperature and energy changes during the thermal denaturation process of proteins, making it a highly effective method for studying conformational changes and structural stability of proteins. This technique achieves this by measuring the heat changes associated with molecularly controlled heating.
DSC is a classic application used to study the interaction between drugs and excipients. To determine whether there is mutual influence, the key factor is to observe whether the characteristic endothermic or exothermic peaks of the drug disappear or change in shape and whether there is a shift in the observed temperature. Additionally, the appearance of new peaks can be considered evidence of interaction between the drug and excipients.
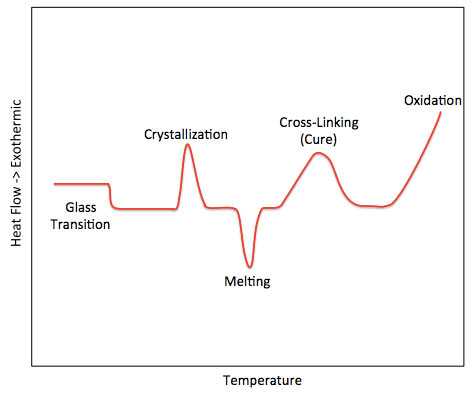
Our DSC Service Offerings
★ Identification and selection of the most stable proteins or potential drug candidates during the development of biopharmaceuticals.
★ Study of ligand interactions.
★ Rapid optimization of purification and production conditions.
★ Easy and fast determination of optimal conditions for liquid formulations.
★ Quick stability indication analysis of target proteins used in screening.
DSC Applications In Biology
Differential Scanning Calorimetry is a versatile technique with applications ranging from studying protein stability and nucleic acid folding to analyzing lipids, carbohydrates, and monoclonal antibodies. It enables researchers to investigate thermal properties, interactions, and structural changes in biological macromolecules, contributing to advancements in various fields, including pharmaceuticals and food science.
| Application | Description |
|---|---|
| Protein Analysis | - Investigate thermal stability of proteins and measure thermodynamic parameters of thermal unfolding. |
| - Determine ideal conditions for stabilizing liquid formulations of proteins. | |
| - DSC can also detect protein aggregation, a common culprit of protein instability leading to various diseases | |
| - Detect fine-tuning of interactions between protein domains, providing insights into protein flexibility and stabilization. | |
| Nucleic Acid Analysis | - Study thermodynamics of nucleic acid-folding transitions by measuring excess heat capacity in solution. |
| - Provide information about melting points, enthalpies, and physical-chemical interactions in nucleic acid structures. | |
| Lipid Analysis | - Analyze oils and lipids, particularly studying lipid oxidation and thermal effects. |
| - Offer a simple and direct way to monitor autoxidation processes and oxidative stability, relevant to food components. | |
| Carbohydrate Analysis | - Study gelatinization processes of starch granules in the presence of water or solvents. |
| - Reveal physicochemical changes during gelatinization, such as swelling, exudation, and granule disruption. | |
| - Valuable for investigating the thermoxidative behaviors of simple and complex carbohydrates. | |
| mAb Analysis | - Understand conformational changes, stability, and aggregation of monoclonal antibodies (mAbs) at different pH values. |
| - Provide insights into how acidic environments affect mAb domains and overall stability. | |
| - Aid in developing valuable approaches for diagnosing and treating autoimmune diseases. |
Experimental Instruments
Malvern VP-capillary DSC
Sample Requirement
Sample Concentration and Purity: Total mass of 500μg or more, concentration of 1mg/ml or higher, purity of 95% or higher.
DSC Service Measurement Principles
In a solution, biological molecules maintain a balance between their native (folded) and denatured (unfolded) conformations. The higher the temperature at the midpoint of the thermal transition (Tm), the more stable the molecule. DSC measures the unfolding enthalpy (∆H) caused by thermal denaturation. It is also used to determine the changes in heat capacity (ΔCp) during the denaturation process. DSC can elucidate the factors contributing to the folding and stability of native biomolecules, including hydrophobic interactions, hydrogen bonding, conformational entropy, and the physical environment.
Accurate and high-quality data obtained from DSC provide crucial information about the stability of proteins during process development and potential candidate therapeutic drug formulation.
Large molecules and macromolecular complexes (>5000 Daltons), such as proteins, nucleic acids, and lipids, can undergo distinct conformational changes upon heating. The rearrangement of non-covalent bonds results in heat absorption. Differential scanning calorimetry can measure this heat absorption.
What Is Differential Scanning Calorimetry
Differential Scanning Calorimetry, or DSC for short, is a formidable analytical instrument that exhibits exceptional capability in measuring the heat energy that is essential for the heating or cooling of a sample in relation to temperature or time. This cutting-edge technology is known for its extensive applications in material science, pharmaceuticals, food, and biochemistry, where it serves the crucial role of meticulously analyzing the thermal behavior of a diverse range of samples, encompassing polymers, small molecules, and biomolecules, among others.
DSC is used to determine the difference of the heat flow rate between the tested sample and a reference, over a controlled temperature range. This analytical procedure is achieved in a closed system, which is isolated from the surroundings by some boundary, through which only heat and energy but not mass are able to flow. Calorimetry can be conducted at either constant pressure or constant volume, which allows the analysts to monitor the temperature changes induced by the investigated reaction.
Key Features of DSC
Highly sensitive: Small changes in heat flow can be detected, making it a powerful tool for analyzing thermal events in materials.
Non-destructive: Can be used to analyze a wide range of materials including solids, liquids and gases.
Ease of use: Most instruments offer automated data collection and analysis.
Versatility: Widely used in molecular analysis in pharmaceutical, material, food industries

| Sample Name | Batch Number | Tm(C) | ||
|---|---|---|---|---|
| Tm₁ | Tm₂ | Tm₃ | ||
| QC1 Reference | Q**** | 70.43 | 75.61 | 83.33 |
| QC1 Product | QC1-01 | 70.65 | 75.65 | 83.48 |
| QC1 Product | QC1-02 | 70.75 | 75.69 | 83.33 |
| QC1 Product | QC1-03 | 70.62 | 75.67 | 83.34 |
| QW | ***1 | 71.09 | 75.63 | 83.28 |
| QW | ***2 | 71.14 | 75.65 | 83.25 |
| QW | ***3 | 71.44 | 75.67 | 83.24 |
Table 1: DSC Test Results
Low-dose metformin targets the lysosomal AMPK pathway through PEN2
Journal: Nature
Published: 2020
Background:
SPR (Biacore), DSC, and ITC Experiments Facilitate Metformin Target Analysis
Metformin (chemical formula C4H11N5, molecular weight 129.164) is a derivative of a natural compound and is the most commonly used drug for treating type 2 diabetes. In recent years, researchers have discovered various remarkable effects of metformin. Besides lowering blood glucose levels, it also shows potential in resisting various cancers caused by diabetes and may have anti-aging and longevity effects for healthy individuals. However, the effects of metformin are limited; for example, it only targets a few tissues such as the liver and intestines and has no effect on adipose tissue. Therefore, designing drugs that specifically target metformin to adipose tissue could bring significant benefits to the treatment of metabolic diseases caused by excessive nutrition. This article reveals the mechanism behind metformin's blood glucose-lowering effect and identifies new clues related to its anticancer properties.
Methods and Results:
The research team first synthesized a chemical probe for metformin (Met-P1). They incubated biotinylated metformin with lysate from lysosomes and performed pull-down analysis using NeutrAvidin beads, followed by mass spectrometry analysis (Figure 1). Among 367 proteins identified in the lysosomes, 113 proteins were found to be pulled down by the Met-P1 probe. Subsequently, the researchers used shRNA silencing to knock down each of these proteins in cells, eventually identifying a protein named PEN2 that mediates metformin's activation of AMPK. Co-localization microscopy STORM and transmission electron microscopy data also showed that approximately 40% of PEN2 is located in lysosomes.

Subsequently, the researchers further investigated the binding between the PEN2 protein and metformin at the molecular level. Firstly, they confirmed the binding of metformin to the PEN2 protein through DSC (Differential Scanning Calorimetry) and ITC (Isothermal Titration Calorimetry) experiments. Then, they quantitatively studied the interaction between metformin and the PEN2 protein using SPR (Surface Plasmon Resonance) (Figure 2f).
Figure 2. Interactions between metformin and PEN2 protein detected by DSC, ITC, and SPR.Computational model of metformin binding to PEN2 and SPR detection of the interaction between PEN2-2A and metformin.
The SPR experiments were conducted using Biacore T200. PEN2 or double-mutant PEN2 protein was diluted in pH 4.0 sodium acetate solution and immobilized on two channels of a CM5 chip, with adjacent channels serving as reference. Metformin was diluted into concentrations of 0.05, 0.78, 1.56, 3.125, and 6.25 μM and injected for binding, with chip regeneration performed using 10mM Glycine pH 2.0. The Biacore results revealed an affinity KD of 0.15 μM (ka=2.8E+3 M-1S-1, kd=4.2E-4 S-1) between metformin and the PEN2 protein, a value within the range of intracellular metformin concentrations in animals or patients under normal medication. The computational model showed that metformin interacts directly with the F35 and E40 residues of PEN2 (Figure 3e), while mutation of F35 and E40 to alanine (PEN-2A) blocked the binding with metformin, and Biacore results demonstrated that PEN-2A does not bind to metformin (Figure 3f).
The authors continued their investigation into the downstream pathway of PEN2. After incubating PEN2 with lysate proteins, immunoprecipitation was performed followed by mass spectrometry analysis. A total of 1881 proteins were found to interact with PEN2, among which 889 showed significant changes after treatment with metformin. Out of these 889 proteins, 123 were intracellular lysosomal proteins. Through a series of in-cell and in-vitro co-immunoprecipitation experiments, ATP6AP1 (a v-ATPase auxiliary factor) was identified as a protein that relies on the interaction between metformin and PEN2. Subsequent experiments further indicated that PEN2 is a prerequisite for metformin to activate the lysosomal pathway and activate AMPK. Deletion of PEN2 not only prevented AMPK activation by metformin but also abolished its effects on reducing fatty liver, alleviating hyperglycemia, and extending lifespan.
 Figure 3. AMPK activation signaling pathways based on metformin-PEN2-ATP6AP1 and other pathways.
Figure 3. AMPK activation signaling pathways based on metformin-PEN2-ATP6AP1 and other pathways.
The authors identified PEN2 as the target of metformin. High concentrations of metformin can activate AMPK by increasing AMP levels. However, after stimulation with low concentrations of metformin, PEN2 binds to ATP6AP1, inhibiting the activity of v-ATPase without increasing AMP and ADP levels, and subsequently activating lysosomal AMPK. Thus, the PEN2-ATP6AP1 pathway constitutes a signal diversion that intersects with the lysosomal v-ATPase-AXIN-AMPK pathway, allowing low concentrations of metformin to utilize an AMP-independent AMPK activation signaling pathway, which can also be activated by glucose starvation. The authors confirmed that the PEN2-ATP6AP1 pathway does not participate in low-glucose-induced AMPK activation, indicating that the PEN2-ATP6AP1 pathway is a parallel route to the v-ATPase complex. In this way, the two pathways, PEN2-ATP6AP1 and aldolase-TRPV, respectively sense the presence of metformin and glucose starvation and influence v-ATPase to control AMPK activation (Figure 4).
Our Capabilities
Creative Proteomics can provide the following experiments used in this study: SPR protein-small molecule affinity analysis, ITC affinity measurement, and DSC experiments. Additionally, we also offers nanoDSF experiments to detect protein-small molecule interactions with reduced sample consumption compared to DSC.




