- Service Details
- Case Study
Metabolic Pathways of Tryptophan
Tryptophan is an essential amino acid and a constituent of proteins. It serves as a precursor for various biologically active compounds within living organisms. Tryptophan metabolites refer to a series of compounds produced through the metabolic processes of tryptophan in organisms, many of which play crucial physiological roles.
Tryptophan plays a significant role in growth, development, and health of animals and humans. In the past decades, tryptophan is widely studied as an essential amino acid of protein synthesis in mammalian. However, there are increasing evidence show that besides its primary role serving as a building block in protein biosynthesis, tryptophan and its metabolites is also the key nutrient essential for regulating nervous system, reproduction system, immune responses and anti-stress responses. What's more, tryptophan may affect whole-body homeostasis in organisms by modulating gene expression and nutrient metabolism.
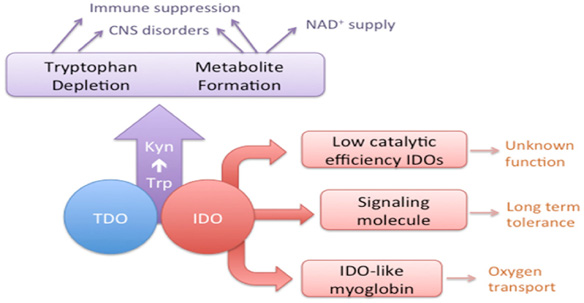
(1) Kynurenine Pathway-Mediated Tryptophan Metabolism
The Kynurenine pathway primarily occurs in the liver, where over 95% of tryptophan is degraded into various active compounds. In humans, Indoleamine 2,3-dioxygenase (IDO) is expressed in organs such as the brain, gastrointestinal tract, and liver, while Tryptophan 2,3-dioxygenase (TDO) is predominantly expressed in the liver. Tryptophan is first converted into N-formylkynurenine (NFK) by three rate-limiting enzymes: IDO1, IDO2, and TDO. NFK is further demethylated to form Kynurenine (Kyn). Kynurenine is also a precursor for NAD+ biosynthesis. TDO is primarily found in the liver and brain and is regulated by tryptophan levels and glucocorticoids. Most human tissues do not express IDO. It is induced by various pro-inflammatory mediators such as interferon-gamma, interleukin-2, and tumor necrosis factor-alpha. The expression of IDO correlates positively with Kynurenine levels, and the Kynurenine/Tryptophan ratio serves as a valuable biomarker for IDO activity.
(2) 5-Hydroxytryptamine (5-HT) Pathway-Mediated Tryptophan Metabolism
Tryptophan hydroxylase (TPH) catalyzes the conversion of a small amount of tryptophan into 5-hydroxytryptophan (5-HTP). Aromatic L-amino acid decarboxylase, in conjunction with the cofactor pyridoxal phosphate, converts 5-HTP into 5-hydroxytryptamine (5-HT), commonly known as serotonin. 5-HT is a monoamine neurotransmitter in the central nervous system of humans, where it regulates various physiological functions, including vascular smooth muscle contraction, hence its alternative name, serotonin. Serotonin synthesis occurs in the intestines and the brain. Peripheral serotonin is primarily synthesized by TPH1 in enterochromaffin cells of the gut. It is then stored in platelets and facilitates various physiological functions, including vascular tone regulation and smooth muscle cell proliferation.
(3) Indole Pathway-Mediated Tryptophan Metabolism
Gut microbiota convert small amounts of tryptophan into indole and its derivatives, including indolepropionic acid, indole-3-acetic acid (IAA), indole-3-propionic acid (IPA), indole-3-acetylaldehyde (3-IAld), and skatole. Bacteria involved in breaking down tryptophan into indole derivatives include Anaerostipes, Bacteroides, Clostridium, Bifidobacterium, and Lactobacillus. Clostridium sporogenes and Ruminococci convert tryptophan into skatole. Lactobacillus catalyzes the formation of indole-3-carboxaldehyde (I3a) from tryptophan. Certain bacteria like Pseudomonas, Staphylococcus, and Providencia facilitate the conversion of tryptophan into Kynurenine. Indole and its derivatives contribute to gastrointestinal function, inflammation, antioxidant activities, and immune system regulation.

Significance of Tryptophan Metabolism Analysis
Tryptophan metabolism primarily involves the conversion of tryptophan into 5-hydroxytryptophan by the action of tryptophan hydroxylase, followed by the transformation of 5-hydroxytryptophan into 5-hydroxytryptamine (5-HT), commonly known as serotonin, through the activity of aromatic L-amino acid decarboxylase. Serotonin is a crucial neurotransmitter involved in various physiological processes, including mood regulation, sleep modulation, and appetite control. Therefore, serotonin is widely utilized in clinical settings for the treatment of psychiatric disorders such as depression and anxiety.
Apart from serotonin, tryptophan metabolism yields other essential metabolites. For instance, tryptophan can be converted into indole-3-acetic acid through the tryptophan-indoleacetic acid pathway. Indole-3-acetic acid serves as a vital antioxidant capable of protecting cells against oxidative damage. Additionally, tryptophan can be transformed into quinolinic acid via the tryptophan-quinolinate pathway. Quinolinic acid is a significant alkaloid with various biological activities, including anti-inflammatory, antibacterial, and anti-tumor properties.
Tryptophan Metabolism Analysis Service
Liquid chromatography-tandem mass spectrometry (LC-MS/MS) technology is a commonly employed analytical method. Due to its robust specificity, high sensitivity, and rapid analysis capabilities, it has become increasingly integrated with clinical diagnosis and monitoring. In recent years, this approach has gained substantial recognition and favor among mass spectrometry experts and medical researchers, contributing to the rapid development of clinical mass spectrometry.
In order to provide improved services, Creative Proteomics has developed a high-throughput, highly comprehensive, and highly sensitive targeted quantification method for tryptophan metabolites. This method enables precise quantitative analysis of dozens of amino acids and their derivatives in a short period. Through a single analysis, multiple tryptophan metabolites can be simultaneously quantified, comprehensively covering the metabolic pathways associated with tryptophan. This approach not only saves time and resources but also meets the requirements for the detection of most tryptophan metabolites.
| No. | Full Name | Abbreviation |
|---|---|---|
| 1 | Tryptophan (L-Tryptophan) | L-TRP |
| 2 | Kynurenine (L-kynurenine) | L-KYN |
| 3 | Picolinic acid | PA |
| 4 | Tryptamine | TRM |
| 5 | Melatonin | MLT |
| 6 | Xanthurenate/Xanthurenic acid | XA |
| ... | ... | ... |
| 26 | 3-Indoleacrylic acid | IA |
| 27 | Indole-3-acetamide | IAM |
| 28 | 5-Methoxytryptamine | 5 MeO TA/5 MeOT |
| 29 | N-methyltryptamine | N-Me-TA |
Service Advantages
Comprehensive Substance Detection: Comprehensive coverage of all three tryptophan metabolic pathways.
Customized Experimental Design: Optimal metabolite pre-processing methods, chromatography, and mass spectrometry method selection based on sample types to enhance detection sensitivity and accuracy.
Accurate Qualitative and Quantitative Results: Calibration with standard curves combined with isotope internal standards ensures precise qualitative and quantitative outcomes.
Premium Detection Platform: Utilization of ultra-sensitive detection platforms with MRM mode combined with internal standard calibration to guarantee quantitative accuracy.
Method Validation: Rigorous methodological validation to ensure detection accuracy.
Excellent Post-Service Support: Fast turnaround time for quicker results. A robust research and development team ensures comprehensive support for data utilization.
Sample Requirement
Animal and Clinical Tissue Samples: 200 mg/sample
Serum and Plasma: 200 μL/sample
Cells: 1 x 10^7 cells/sample
Feces: 200 mg/sample
Impaired Aryl Hydrocarbon Receptor Ligand Production by the Gut Microbiota Is a Key Factor in Metabolic Syndrome
Journal: Cell Metabolism
Published: 2018
Abstract
The role of microbiota alterations in defining or influencing metabolic diseases remains unclear, but microbiota metabolism byproducts play a crucial role in host-microbiota interactions. In both pre-clinical and clinical contexts, the author have identified an association between metabolic syndrome and reduced microbiota capacity to metabolize tryptophan into derivatives that activate the aryl hydrocarbon receptor (AhR). This alteration isn't merely a consequence of the disease; supplementation with AhR agonists or a high AhR ligand-producing Lactobacillus strain improves dietary and genetically induced metabolic impairments, particularly addressing glucose dysmetabolism and liver steatosis. These improvements result from enhanced intestinal barrier function and the secretion of the incretin hormone GLP-1. The author utilized microbiota 16S sequencing coupled with tryptophan-targeted metabolism analysis to unveil the pivotal role of impaired generation of aryl hydrocarbon receptor (AhR) ligands by the gut microbiota in metabolic syndrome.
Technical methods
Microbiota 16S Sequencing
Tryptophan-Targeted Metabolism Analysis
1. Fecal AhR activity and related metabolite statistics of patients with metabolic syndrome
Through fecal testing of diabetic and healthy patients with metabolic syndrome classified according to BMI index and relatively healthy people, it was found that the intestinal flora of metabolic syndrome have a significant ability to metabolize tryptophan to generate aryl hydrocarbon receptor (AhR) ligands. weakened; and impaired AhR activity was significantly positively correlated with metabolic syndrome.

2. The intestinal flora and AhR ligands of mice with metabolic syndrome are significantly changed.
In order to identify the causal link between the weakening of AHR ligands caused by changes in intestinal flora and metabolic syndrome, the researchers constructed a mouse model of metabolic syndrome through HFD diet and found that, similar to clinical patient indicators, HFD mice were unable to produce glucose. Resistance and steatosis, and the weakened ability of intestinal flora to produce AhR ligands leads to a decrease in AhR agonist activity, accompanied by a significant decrease in the expression of AhR target genes Il22, Reg3g and Reg3b. On the contrary, the concentration of kynurenine is significantly increased. . 16S sequencing showed that the bacterial flora structure of HFD mice changed, and the intestinal flora that produced AhR ligands changed significantly.

3. Supplementing AhR agonist or AhR ligand-producing bacterial flora L. reuteri can slow down HFD/gene-induced metabolic damage
In order to further determine the function of intestinal flora on metabolic damage, the researchers injected the AhR agonist Ficz into metabolic syndrome mice or supplemented Lactobacillus reuteri CNCM I-5022, which can produce a large amount of AhR ligands. The results found that AhR agonist Although the drug Ficz or Lactobacillus reuteri did not reduce obesity, it improved glucose metabolism, fatty liver and blood lipids. The above results prove that the AhR agonist Ficz has a good therapeutic effect on HFD or gene-induced metabolic syndrome.

4. Supplementing AhR agonists improves HFD-induced intestinal barrier disorder and promotes the production of incretin GLP-1
The researchers then studied the molecular mechanism by which the restoration of AhR activity improves metabolic syndrome. The results showed that the activation of AhR signaling can promote the recovery of intestinal barrier function, reduce systemic inflammation, and stimulate enteroendocrine cells to produce incretin GLP-1 through AhR. Improve metabolic syndrome disorders.

Summary
This study started with fecal 16S microbial sequencing of patients with metabolic syndrome and found that the intestinal flora changed significantly, and the ability of the flora to produce aryl hydrocarbon receptor (AhR) ligands decreased, thereby causing intestinal barrier damage and reducing incretin. GLP-1 secretion ultimately aggravates metabolic syndrome. Subsequent functional verification experiments can improve these problems by supplementing AhR agonists or probiotics that can produce large amounts of AhR ligands, which is a new strategy for preventing and treating metabolic disorders.
References
- Hu S,Luo L,Bian X, et al. Pu-erh Tea Restored Circadian Rhythm Disruption by Regulating Tryptophan Metabolism.Journal of Agricultural and Food Chemistry.2022.
- Paul Petrus, Marlene Cervantes,et al.Tryptophan metabolism is a physiological integrator regulating circadian rhythms.Molecular Metabolism.2022.
- Bingnan Liu,Dongming Yu, et al.Characterizing the influence of gut microbiota on host tryptophan metabolism with germ-free pigs.Animal Nutrition.2022.
- Chen Xue et al. Tryptophan metabolism in health and disease. Cell Metabolism. 2023
- Natividad JM et al. Impaired Aryl Hydrocarbon Receptor Ligand Production by the Gut Microbiota Is a Key Factor in Metabolic Syndrome. Cell Metab. 2018





