Thiols Analysis Service
Get reliable thiol quantification without oxidation artifacts.
Creative Proteomics stabilizes, derivatizes, and quantifies biothiols and volatile thiols across complex matrices—so you can track redox status, process consistency, and product quality with confidence.
- We solve: unstable thiols, matrix interferences, low-abundance detection
- You get: verified GSH/GSSG ratios, comprehensive biothiol panels, volatile thiol fingerprints
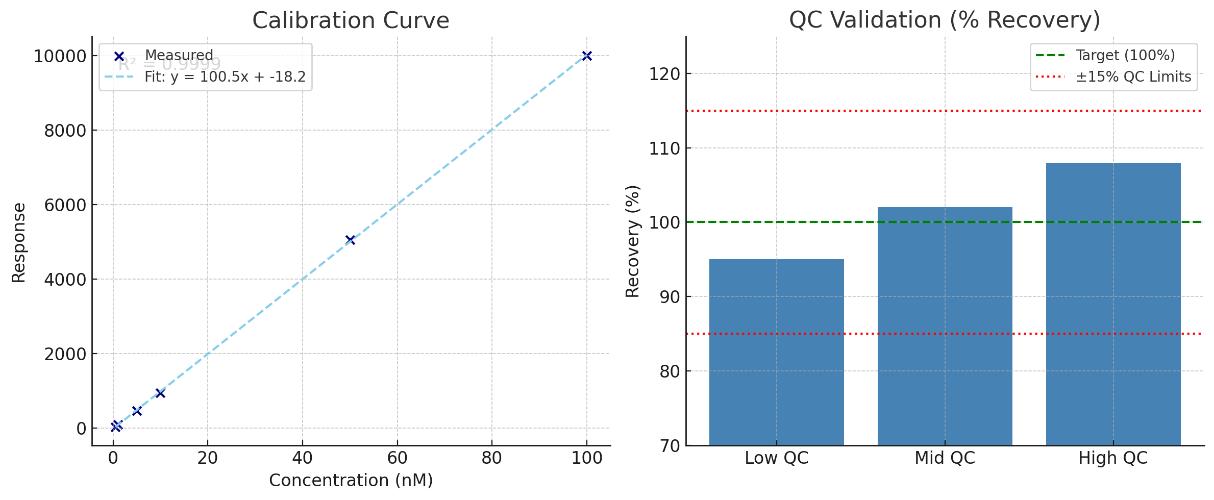
- Proof in numbers: LLOQ down to 0.5–10 nM (biothiols) and sub-ng/L (volatile thiols); linearity r² ≥ 0.995 over ≥4 orders; precision CV ≤ 10%
Submit Your Request Now
×
What You Will Receive
- Quantified Thiol Levels: Concentrations of GSH, Cys, 3MH, and others, reported in nM or ng/L.
- Redox Ratios & Metrics: GSH/GSSG, Cys/Cystine, Hcy ratios, and optional redox potential (Eₕ).
- QC-Verified Results: Includes calibration curves, recovery, CVs, blanks, and QC plots.
- Raw & Processed Data: Delivered in .raw / .wiff, plus .mzML and .csv summary files.
- What We Provide
- Advantages
- Technology Platform
- Sample Requirement
- Demo
- FAQs
What Are Thiols and Why Analyze Them?
Thiols (–SH compounds) are reactive sulfur-containing molecules critical for redox balance, antioxidant defense, and aroma profiles in biological and industrial systems. Key examples include glutathione (GSH) and cysteine in cellular redox buffering, as well as 3MH and 2-furfurylthiol in fermented beverages.
However, their high reactivity also makes them analytically unstable. Without rapid stabilization, thiols oxidize or degrade, leading to inaccurate quantification and misleading data. Conventional workflows often capture artifacts rather than true in-sample concentrations.
Targeted thiols analysis—with appropriate derivatization, internal standards, and matrix-specific methods—is essential for reliable results. Whether assessing GSH/GSSG ratios, profiling volatile sulfur compounds, or tracking low-abundance thiols in bioprocesses, accurate thiol data provides actionable insights into product quality, biological state, and process performance.
Thiols Analysis Services Offered by Creative Proteomics
- Targeted Biothiols Panel (quantitative): Cys, Cys-Gly, Hcy, γ-Glu-Cys, GSH/GSSG, CoA-SH, NAC, Cystine, Homocystine
- Extended Sulfur Metabolites (semi-quant/quant): Cysteamine, 3-mercaptopropionic acid, 2-mercaptoethanol, penicillamine
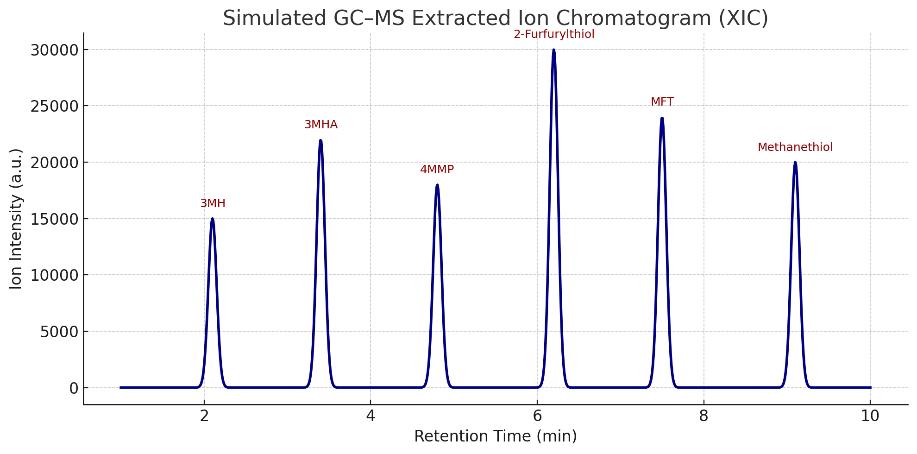
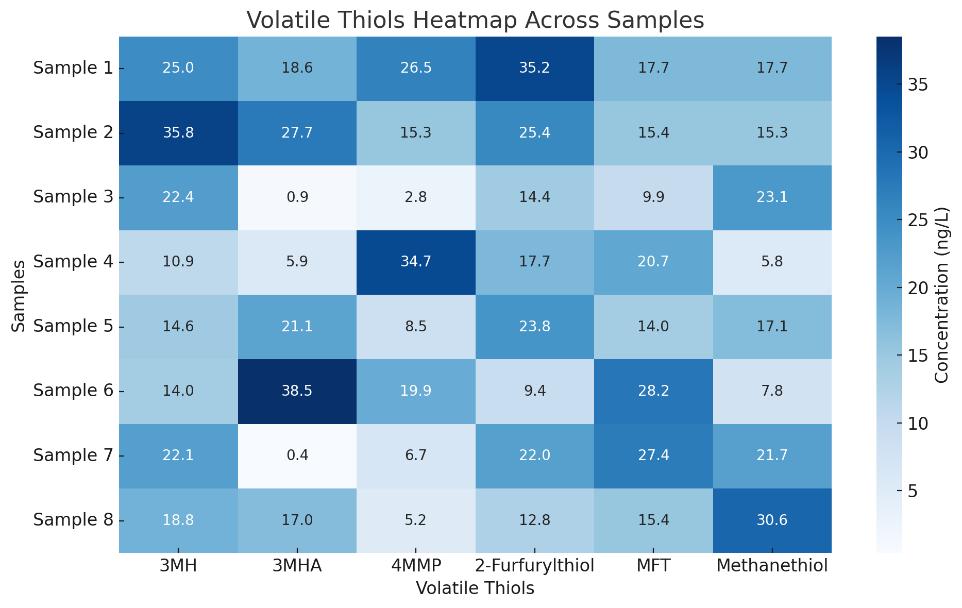
- Volatile Thiols Fingerprinting (targeted GC–MS/MS): 2-furfurylthiol, 4MMP, 3MH, 3MHA, MFT, benzyl mercaptan, ethanethiol
- Protein Thiol/Disulfide State (mapping): Differential alkylation and peptide-level LC–MS for relative thiol occupancy
- Redox Metrics: GSH/GSSG, Cys/Cystine, Hcy/Homocystine ratios and Nernst-based redox potential (optional)
- Stability & Stress Studies: Thiol loss kinetics under process conditions; antioxidant/additive screening
Thiols Analysis Targets & Compound Panels
| Panel | Representative Analytes | Typical Application | Typical LLOQ* |
|---|---|---|---|
| Core Biothiols (LC–MS/MS) | - Glutathione (GSH) - Oxidized Glutathione (GSSG) - Cysteine (Cys) - Cystine - Homocysteine (Hcy) - Homocystine | Redox balance, oxidative stress, metabolic studies | 0.5–10 nM |
| Extended Biothiols (LC–MS/MS) | - γ-Glutamylcysteine - Cysteinylglycine - N-acetylcysteine (NAC - Coenzyme A (CoA-SH) - Cysteamine - 3-Mercaptopropionic acid - 2-Mercaptoethanol - Penicillamine | Drug metabolism, cell signaling, thiol scavenger profiling | 1–20 nM |
| Volatile Thiols (GC–MS/MS) | - 3-Mercaptohexanol (3MH) - 3-Mercaptohexyl acetate (3MHA) - 4-Mercapto-4-methylpentan-2-one (4MMP) - 2-Furfurylthiol (FFT) - 2-Methyl-3-furanthiol (MFT) - Benzyl mercaptan - Methanethiol - Ethanethiol | Flavor/aroma profiling in wine, beer, fermented products | 0.2–2 ng/L |
| Thiol Ratios & Redox Metrics | - GSH/GSSG ratio - Cys/Cystine ratio - Hcy/Homocystine ratio - Redox potential (Eₕ, optional) | Redox state monitoring, stress response profiling | – |
Advantages of Our Thiols Analysis Services
- Sensitivity & Range: LLOQ 0.5–10 nM (biothiols) and 0.2–2 ng/L (volatiles); dynamic range ≥ 10⁴ with weighted calibration (r² ≥ 0.995).
- Accuracy & Precision: Isotope dilution recovery 85–115%; intra-run precision CV ≤ 10%, inter-run ≤ 15% across matrices.
- True-state Capture: Immediate NEM/IAM capping prevents >90% of artifactual GSH oxidation observed without stabilization.
- Carryover Control: Post-run and needle wash validated to <0.1% for high-abundance thiols.
- Trace Volatiles: SPME enrichment + MS/MS achieves signal-to-noise ≥ 10 at sub-ng/L with matrix-matched standards.
- QC Transparency: Blanks, duplicates, matrix spikes, and low/medium/high QCs bracket every batch; full QA plots provided.
Workflow for Thiols Analysis Service
1. Consult & Panel Design: Define targets, matrices, expected ranges, stabilization strategy (NEM/IAM/mBBr).
2. Method Setup: Choose LC–MS/MS, GC–MS/MS, or hybrid; assign internal standards and calibration range.
3. Sample Stabilization: Provide pre-loaded tubes or on-receipt alkylation; confirm pH/temperature controls.
4. Extraction & Cleanup: Protein precipitation or SPE; HILIC/RP selection to minimize matrix effects.
5. Acquisition: MRM-based quant with bracketed QCs, blanks, and matrix spikes; volatility-optimized SPME for aroma thiols.
6. Quantification & QA: Weighted regression, back-calculation checks, recovery and precision acceptance, carryover review.
7. Reporting: Concentrations (with units), key ratios (e.g., GSH/GSSG), method metadata, calibration/QC plots, and raw files.
Technology Platform for Thiols Analysis Service
LC–MS/MS for Biothiols
Widely used for quantifying GSH, GSSG, cysteine, homocysteine and related thiols in biological matrices (plasma, tissues, cell lysates).
Instrumentation:
UHPLC: Agilent 1290 Infinity II or Waters ACQUITY UPLC
MS/MS: AB Sciex Triple Quad 6500+ or Waters Xevo TQ-S micro
Chromatography:
Column: 2.1 × 100 mm, 1.7 μm C18 or HILIC
Derivatization & Stabilization:
N-ethylmaleimide (NEM) or iodoacetamide (IAM) to prevent oxidation prior to extraction
MS Parameters:
Ion source: ESI positive mode
Quantification:
Internal standards: ¹³C₂-Gly, d₅-GSH, d₄-Cys, etc.
Calibration: 1/x or 1/x² weighting
LOQ: Typically 0.5–10 nM
GC–MS/MS for Volatile Thiols
Used to detect ultra-trace levels of aroma-active thiols in beverages, fermentation broths, and flavor samples.
Instrumentation:
GC: Agilent 7890B
MS/MS: Agilent 7010B Triple Quadrupole
Ionization: EI or NCI (depending on derivatization)
Sample Preparation:
Preconcentration via SPME (DVB/CAR/PDMS fiber)
Salt and pH adjustment to optimize recovery
Derivatization (if applicable):
Pentafluorobenzyl bromide (PFB-Br) for enhanced volatility and MS response
Quantification:
Stable-isotope dilution (e.g., d₄-3MH, d₂-4MMP)
Method-specific MRM transitions
LOQ: Typically 0.2–2 ng/L, matrix-dependent

SCIEX Triple Quad™ 6500+

Agilent 5977B GC/MSD

Waters ACQUITY UPLC System

Thermo Fisher Q Exactive
Sample Requirements for Thiols Analysis Service
| Sample Type | Minimum Amount | Container & Preservative | Handling & Storage | Notes |
|---|---|---|---|---|
| Plasma / Serum | ≥100 μL | EDTA or heparin tube; transfer to amber microtube pre-loaded with NEM (final ~10 mM) | Chill on ice immediately; snap-freeze; ship on dry ice | Avoid hemolysis; ≤1 freeze-thaw |
| Whole Blood (for rapid GSH/GSSG) | 200 μL | Tube pre-loaded with NEM; gentle inversion | Process or freeze promptly; dry ice shipping | Record anticoagulant type |
| Urine | ≥500 μL | Amber tube; optional NEM stabilization | Freeze at −80 °C; dry ice shipping | Provide creatinine for normalization (optional) |
| Cell Pellets | ≥1×10⁶ cells or ≥10 mg wet mass | Pre-cooled tube; add alkylating PCA buffer on receipt | Keep cold; snap-freeze | Supply cell count/protein if available |
| Tissue | ≥20 mg | Cryovial; no buffer; dry ice | Snap-freeze post-collection; store −80 °C | Do not thaw before shipment |
| Fermentation / Media | ≥1 mL | Polypropylene; optional filtration 0.22 μm; NEM if biothiols targeted | Freeze; dry ice shipping | Record medium composition |
| Beverages (volatile thiols) | ≥10 mL | Amber headspace vials filled to minimize headspace | Chill; ship cold or on ice packs; avoid light | Provide pH and ethanol (% v/v) |
| Formulations | ≥1 mL or ≥100 mg | Original container plus aliquot in amber tube | Protect from light; ship cold | Include excipient list for matrix match |
Demo Results
FAQ of Thiols Analysis Service
How is thiol stability maintained during sample transport and processing?
Alkylating agents such as NEM or IAM are applied immediately upon sample collection or receipt. This prevents disulfide formation and ensures true-state quantification of reduced thiols.
What controls are included to ensure data quality and comparability across batches?
All runs are bracketed with internal standards, matrix-matched calibration curves, and multi-level QCs (low/mid/high). Recovery, CVs, and back-calculation are reported per analyte.
Can the method distinguish between endogenous thiols and exogenous thiol-containing additives?
Yes. Targeted MRM transitions and isotope dilution quantification enable compound-specific resolution, even in the presence of formulation excipients or derivatized analogs.
Is the method validated for complex matrices like fermentation broth or high-protein samples?
Yes. Matrix-specific cleanup (e.g., SPE or precipitation) and method adjustments are implemented to handle high protein, sugar, or salt content without compromising sensitivity or selectivity.
How are thiol-related redox metrics (e.g., GSH/GSSG) calculated and interpreted?
Molar concentrations are directly measured and used to compute relevant ratios. Optional redox potential (Eₕ) calculations can be provided using the Nernst equation.
Are volatile thiols quantified in absolute or relative terms?
Absolute quantification (ng/L) is performed using matrix-matched standards and deuterated internal references. This enables batch comparability and aroma intensity ranking.
Can this platform support thiol fingerprinting for batch release or raw material QC?
Yes. Volatile thiol profiles or total biothiol content can be used as stability or identity markers for QC applications in manufacturing pipelines.
Is differential alkylation for thiol/disulfide mapping supported at the peptide level?
Yes. Peptide-level LC–MS/MS analysis with differential labeling can identify site-specific thiol modifications or disulfide scrambling in proteins under redox stress.
What's the typical carryover risk in high-abundance thiol samples?
Carryover is routinely tested and controlled below 0.1% using post-run solvent blanks and dedicated needle wash cycles.
Can custom thiol panels be aligned with previous in-house or published workflows?
Yes. Custom panels can replicate legacy methods or published MRM transitions. Cross-platform method harmonization and comparison studies can be provided on request.
Learn about other Q&A about proteomics technology.
Publications
Here are some of the metabolomics-related papers published by our clients:

- A human iPSC-derived hepatocyte screen identifies compounds that inhibit production of Apolipoprotein B. 2023.
- The activity of the aryl hydrocarbon receptor in T cells tunes the gut microenvironment to sustain autoimmunity and neuroinflammation. 2023.
- Lipid droplet-associated lncRNA LIPTER preserves cardiac lipid metabolism. 2023.
- Inflammation primes the kidney for recovery by activating AZIN1 A-to-I editing. 2023.
- Anti-inflammatory activity of black soldier fly oil associated with modulation of TLR signaling: A metabolomic approach. 2023.
- Non-invasive elevation of circulating corticosterone increases the rejection of foreign eggs in female American robins (Turdus migratorius). 2022.
- Untargeted metabolomics reveal sex-specific and non-specific redox-modulating metabolites in kidneys following binge drinking. 2023.
- Nicotine exposure during rodent pregnancy alters the composition of maternal gut microbiota and abundance of maternal and amniotic short chain fatty acids. 2022.
- Sex hormones, sex chromosomes, and microbiota: identification of Akkermansia muciniphila as an estrogen-responsive bacterium. 2023.