Biomarkers are defined as a characteristic that is objectively quantified and evaluated as an indicator of physiological processes, pathological processes, or pharmacologic responses to a therapy. Biomarkers can implicate various health and disease characteristics, such as the level or type of exposure to an environmental factor, genetic susceptibility, markers of subclinical or clinical disease, or indicators of response to therapy. Biomarkers can be categorized as antecedent biomarkers, screening biomarkers, diagnostic biomarkers, staging biomarkers or prognostic biomarkers. Biomarkers exist in various forms, such as particular hormones (e.g., TSH in Hashimoto's Disease), metabolites (e.g., phenylalanine in urine of newborns with phenylketonuria), antibodies (e.g., anti-citrullinated protein antibodies for rheumatoid arthritis), proteins or peptides (e.g., PSA as an indicator of increased risk for prostate cancer), cell types (e.g., white blood cell counts in infection or cancer) and enzyme levels (e.g., various hepatic enzymes for liver cancer).
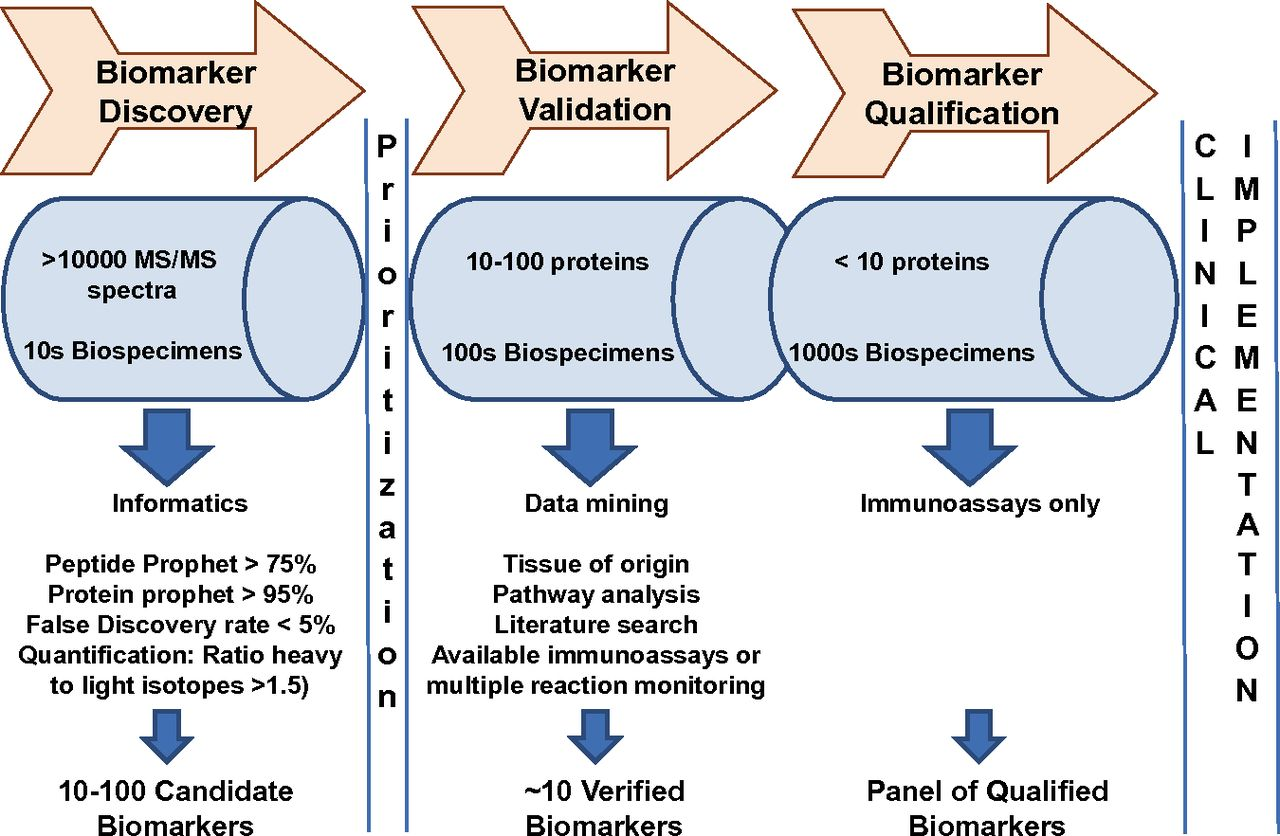
Biomarkers can also be used to evaluate the effectiveness of particular therapies in treating a disease. By measuring drug response rate or toxic effects associated with the drug, specific biomarkers can be used to monitor a patient's reaction to a particular drug, checking whether the treatment is effective and facilitating earlier detection of adverse drug response. Biomarkers also play an important role in the development of new drugs with the discovery of "druggable targets." Alterations in the progression markers can be used to understand whether and how a disease is slowed or reversed by a new therapy, which allows researchers to focus efforts and resources on the most effective ones. Finally, biomarkers can be used to evaluate the progression of a disease or the susceptibility of a disease to a given treatment and hold great promise for personalized medicine. The treatment will be tailored and the most efficient intervention for each individual will be used.
Quantification of biomarkers can be used in various fields, such as basic research and drug development. The quantification can provide a framework for development and regulatory acceptance of biomarkers used in drug development. A big challenge in biomarker quantification is the need to reduce sample complexity while minimizing errors during sample handling at the same time. A HPLC chromatographic separation is often used before mass spectrometric analysis. The improved derivatization techniques, improved sensitivity as well as enhanced speed of MS allowed for the higher sensitivity for the analysis of complex samples with accuracy. The recent advance of MS analyses have greatly contributed to the quantification of biomarkers, minimizing the time and costs for quantification of multiple samples.
Platform
- HPLC-MS
Summary
- Identification and quantification of High-Value Disease Biomarkers by HPLC-MS
Sample Requirement
- Normal Volume: 200ul plasma, 200 mg tissue, 2e7 cells
- Minimal Volume: 50ul plasma, 50 mg tissue, 5e6 cells
Report
- A detailed technical report will be provided at the end of the whole project, including the experiment procedure, MS/MS instrument parameters.
- Analytes are reported as uM or ug/mg (tissue), and CV's are generally<10%.
- The name of the analytes, abbreviation, formula, molecular weight and CAS# would also be included in the report.
| High-Value Disease Biomarkers Quantified in This Service | ||
|---|---|---|
| Aldosterone | Aminoadipic acid | Asymmetric Dimethylarginine (ADMA) |
| Beta-Hydroxybutyrate | Betaine | Billirubin |
| Carnosine | Choline | Creatinine |
| Estradiol | Folate | Formate |
| Glucose | Glutamate | Glutamine |
| Glycerol | Homocysteine | HPHPA |
| Indoxylsulfate | Lactate | Leucine |
| Neopterin | Phenylalanine | Pyruvate |
| Taurine | Testosterone | TMAO |
| Tyrosine | Uric Acid | Vitamin D |
With integrated set of separation, characterization, identification and quantification systems featured with excellent robustness & reproducibility, high and ultra-sensitivity, Creative Proteomics provides reliable, rapid and cost-effective High-Value Disease Biomarkers targeted metabolomics services.





