Carnitine Metabolism Analysis Service
Carnitine is critical for fatty acid oxidation and ATP production—understanding its metabolism is key to solving challenges in energy homeostasis, mitochondrial function, and metabolic disorders. Our cutting-edge LC-MS/MS platform delivers precise, pathway-level insights to power your research or development goals.
Why Choose Creative Proteomics?
- Quantify 70+ carnitines & intermediates with high sensitivity
- Resolve isobaric species via UHPLC-MS/MS precision
- Multi-omics integration for complete metabolic profiling
- Isotope tracing for dynamic flux and turnover studies
- Fast, reliable reporting with visualized pathway maps
Submit Your Request Now
×
- What We Provide
- Advantages
- Technology Platform
- Sample Requirement
- Demo
- FAQ
What Is Carnitine and Why It Matters in Energy Metabolism
Carnitine is an essential molecule that enables the transport of long-chain fatty acids across the mitochondrial membrane, fueling β-oxidation and cellular ATP production. Beyond fatty acid transport, carnitine regulates acyl-CoA/CoA balance, influences amino acid metabolism, and supports energy homeostasis under stress conditions such as fasting, exercise, or drug exposure.
Accurate analysis of carnitine metabolism—including its precursors and related metabolites—offers critical insights into metabolic disorders, nutritional interventions, mitochondrial function, and drug safety evaluations.
Why Perform Carnitine Metabolism Analysis?
Research and industry clients typically require carnitine metabolism analysis to answer questions such as:
- How does carnitine availability impact energy production?
- What is the effect of diet or supplementation on carnitine homeostasis?
- Do candidate compounds interfere with mitochondrial fatty acid oxidation?
- How does genetic regulation of carnitine biosynthetic enzymes influence metabolism?
- Can multi-omics integration reveal biomarkers for metabolic health?
These questions demand precise quantification and pathway-level interpretation—capabilities that Creative Proteomics delivers through advanced analytical workflows.
Creative Proteomics's Carnitine Analysis Solutions
Quantitative Carnitine Profiling
Absolute quantification of L-carnitine and short-, medium-, and long-chain acylcarnitines across diverse biological matrices. High-sensitivity LC-MS/MS detection with LOD down to femtomole level and CV ≤10%.
Acylcarnitine Panel Analysis
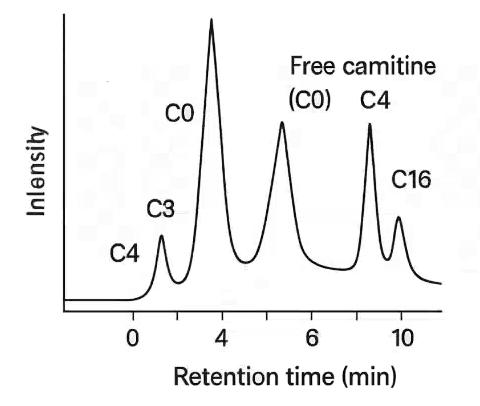
Comprehensive coverage of free carnitine (C0) and acylcarnitines from C2 to C18, including saturated, unsaturated, and hydroxylated species. Suitable for mitochondrial function assessment and β-oxidation studies.
Targeted Pathway Analysis
Evaluation of carnitine biosynthesis pathway intermediates such as trimethyllysine (TML), hydroxytrimethyllysine (HTML), and γ-butyrobetaine (GBB). Integration with fatty acid oxidation markers for systemic energy metabolism profiling.
Isotope Tracer Studies (Optional)
13C- or 15N-labeled carnitine or precursors for flux analysis and dynamic turnover rate determination.
Enzyme Activity and Regulation Assessment (Customizable)
Activity profiling for key enzymes involved in carnitine biosynthesis and transport, such as TMLHE, BBD, and OCTN transporters.
Data Interpretation and Bioinformatics
Delivery of absolute concentrations, pathway-level visualization, and optional multi-omics integration with lipidomics and metabolomics datasets.
What Carnitine Can We Detect?
| Category | Detected Compounds |
|---|---|
| Free Carnitine | L-Carnitine (C0) |
| Short-Chain Acylcarnitines | Acetylcarnitine (C2), Propionylcarnitine (C3), Butyrylcarnitine (C4), Isobutyrylcarnitine (C4-i), Valerylcarnitine (C5), Isovalerylcarnitine (C5-i) |
| Medium-Chain Acylcarnitines | Hexanoylcarnitine (C6), Octanoylcarnitine (C8), Decanoylcarnitine (C10), Dodecanoylcarnitine (C12) |
| Long-Chain Acylcarnitines | Tetradecanoylcarnitine (C14), C14:1, C14-OH, Hexadecanoylcarnitine (C16), C16:1, C16-OH, Octadecanoylcarnitine (C18), C18:1, C18:2, Hydroxy derivatives (C18-OH) |
| Very Long-Chain Acylcarnitines | C20, C20:1, C22, C22:1, C24, C24:1 |
| Hydroxy and Dicarboxylic Acylcarnitines | 3-Hydroxybutyrylcarnitine (C4-OH), 3-Hydroxyhexanoylcarnitine (C6-OH), 3-Hydroxyoctanoylcarnitine (C8-OH), Glutarylcarnitine (C5-DC), Malonylcarnitine (C3-DC), Succinylcarnitine (C4-DC), Methylmalonylcarnitine (C4-DC-methyl) |
| Carnitine Biosynthesis Intermediates | Trimethyllysine (TML), 3-Hydroxytrimethyllysine (HTML), γ-Butyrobetaine (GBB), Betaine |
| Optional Related Analytes | Choline, Acetylcholine, Acyl-CoA derivatives (for extended pathway profiling) |
Advantages of Our Carnitine Analysis Services
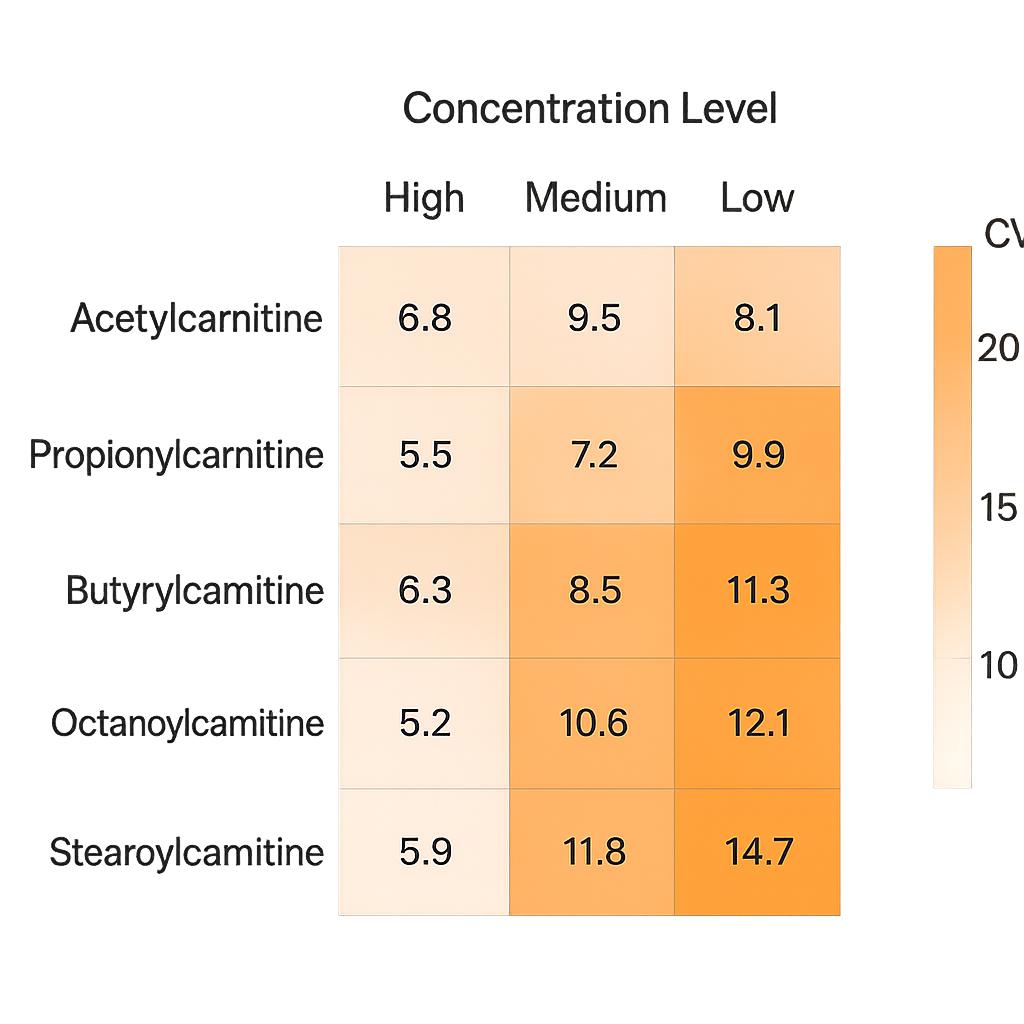
- High Sensitivity and Accuracy: LOD down to 0.1 ng/mL for carnitine and acylcarnitines, with precision CV ≤10%, supported by isotope-labeled internal standards.
- Comprehensive Coverage: Detection of 70+ compounds, including free carnitine, C2–C24 acylcarnitines, hydroxylated/dicarboxylic derivatives, and biosynthetic intermediates.
- Advanced LC-MS/MS Platforms: Triple quadrupole and Orbitrap systems deliver signal-to-noise >100:1, while UHPLC separation ensures resolution of isobaric species (e.g., C4 vs. C4-i).
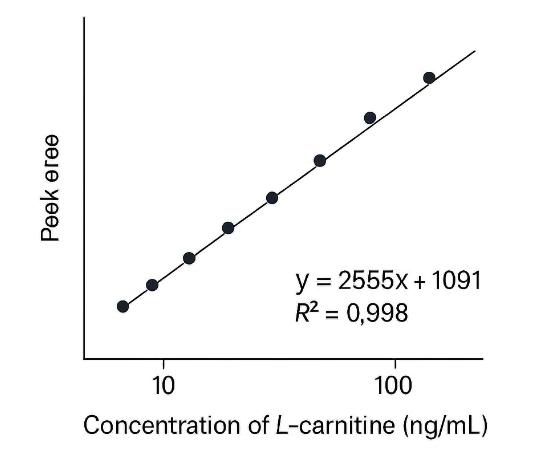
- High Recovery and Reproducibility: Extraction efficiency >85% across plasma, urine, and tissues, with calibration curve correlation R² >0.99 for reliable quantification.
- Flexible Customization: Options for 13C/15N isotope tracer studies and integration with fatty acid oxidation profiling for in-depth energy metabolism research.
Workflow for Carnitine Analysis Service
1. Sample Receipt and Quality Check
Verify sample integrity, matrix type, and volume compliance. Assess storage conditions to maintain metabolite stability.
2. Metabolite Extraction and Stabilization
Apply optimized protocols for high-yield extraction (>85% recovery).
Use antioxidant and pH-controlled buffers to prevent carnitine degradation.
3. Targeted LC-MS/MS Analysis
Perform separation on UHPLC for clear resolution of isobaric acylcarnitines.
Quantification with triple quadrupole or Orbitrap systems using isotope-labeled internal standards.
4. Data Processing and Normalization
Calibration curve validation (R² >0.99) and isotopic correction for absolute accuracy. Normalization by sample type and internal standards.
5. Reporting and Data Delivery
Provide absolute concentrations, QC metrics, and annotated pathway maps. Optional integration with multi-omics data interpretation for broader insights.

Technology Platform for Carnitine Analysis Service
Chromatographic System
- Instrument: Thermo Scientific™ UltiMate™ 3000 UHPLC or Waters ACQUITY UPLC
- Column: C18 reverse-phase (e.g., 2.1 × 100 mm, 1.7 μm)
- Mobile Phases:
- A: Water + 0.1% formic acid
- B: Acetonitrile + 0.1% formic acid
- Flow Rate: ~0.3 mL/min
- Injection Volume: 2–5 μL
- Gradient: Optimized for resolving short-, medium-, and long-chain acylcarnitines, including isobaric species.
 Vanquish™ UHPLC (Figure from Thermo Scientific)
Vanquish™ UHPLC (Figure from Thermo Scientific)
Mass Spectrometry Platform
Triple Quadrupole MS (Targeted Quantification):
- Thermo Scientific™ TSQ Altis or AB Sciex QTRAP® 6500+
- Ionization: ESI positive
- Scan Mode: Multiple Reaction Monitoring (MRM)
- Typical MRM Transitions:
- L-Carnitine: m/z 162 → 103
- Acetylcarnitine (C2): m/z 204 → 85
- Capillary Voltage: ~3.5 kV
- Source Temp: ~350 °C
 Q Exactive™ HF-X (Figure from Thermo Scientifi)
Q Exactive™ HF-X (Figure from Thermo Scientifi)
High-Resolution MS (Confirmation/Discovery):
- Thermo Q Exactive™ Orbitrap
- Resolving Power: Up to 120,000 FWHM @ m/z 200
- Mass Accuracy: <2 ppm
 SCIEX Triple Quad™ 6500+ (Figure from Sciex)
SCIEX Triple Quad™ 6500+ (Figure from Sciex)
Sample Requirements for Carnitine Analysis Service
| Sample Type | Recommended Volume | Storage Condition | Notes |
|---|---|---|---|
| Plasma / Serum | ≥ 100 µL | -80 °C | Collect in EDTA or heparin tubes; avoid repeated freeze–thaw cycles. |
| Urine | ≥ 500 µL | -80 °C | Store immediately after collection; adjust pH if required for stability. |
| Tissue (e.g., liver, muscle) | ≥ 50 mg | Snap-freeze in liquid N₂; store at -80 °C | Homogenization in extraction buffer required prior to analysis. |
| Cell Pellets | ≥ 1 × 10⁶ cells | -80 °C | Wash with cold PBS; remove media to minimize matrix effects. |
| Cell Culture Media | ≥ 500 µL | -80 °C | Filter to remove debris; store immediately after collection. |
Notes:
- Avoid hemolysis during plasma or serum collection to prevent interference.
- Do not use preservatives that may react with carnitine or acylcarnitines.
- Ship samples on dry ice to maintain integrity.
Demo Results
FAQ of Carnitine Analysis Service
What is the typical sensitivity of your method for detecting carnitine and acylcarnitines?
Our LC-MS/MS platform achieves an LOD as low as 0.1 ng/mL for free carnitine and major acylcarnitines, ensuring trace-level detection in complex biological matrices.
Can you differentiate isobaric acylcarnitines such as C4 and C4-i?
Yes. Using UHPLC separation and optimized gradient conditions, we can resolve isobaric species like butyrylcarnitine (C4) and isobutyrylcarnitine (C4-i) before MS detection.
Do you provide analysis for hydroxylated and dicarboxylic acylcarnitines?
Yes. Our method covers hydroxyacylcarnitines (e.g., C4-OH, C8-OH) and dicarboxylic derivatives (e.g., malonylcarnitine, glutarylcarnitine), which are important for fatty acid oxidation disorder studies.
How do you ensure quantitative accuracy across different sample types?
We use isotope-labeled internal standards and validated calibration curves for all target analytes, with R² ≥ 0.99 and inter-batch QC monitoring (CV ≤10%).
What types of data interpretation support can you provide?
Beyond raw and processed data, we offer pathway-level visualization, statistical comparisons, and optional multi-omics integration with lipidomics and metabolomics datasets.
Can the analysis be used for metabolic flux or tracer studies?
Yes. We support 13C/15N isotope tracer experiments, enabling quantification of carnitine turnover rates and pathway flux.
How do you handle matrix effects for tissues and cell cultures?
Our extraction protocols include matrix-specific optimization (e.g., deproteinization, cleanup) and normalization strategies to minimize ion suppression and variability.
Do you offer expanded profiling beyond carnitine and acylcarnitines?
Yes. On request, we can include related metabolites such as trimethyllysine (TML), γ-butyrobetaine, and choline derivatives, supporting carnitine biosynthesis studies.
What quality control measures are included?
We perform blank runs, pooled QC samples, and spiked recovery tests in each batch, and provide CV% and recovery data in the report.
Learn about other Q&A about proteomics technology.
Publications
Here are some of the metabolomics-related papers published by our clients:

- Methyl donor supplementation reduces phospho‐Tau, Fyn and demethylated protein phosphatase 2A levels and mitigates learning and motor deficits in a mouse model of tauopathy. 2023.
- A human iPSC-derived hepatocyte screen identifies compounds that inhibit production of Apolipoprotein B. 2023.
- The activity of the aryl hydrocarbon receptor in T cells tunes the gut microenvironment to sustain autoimmunity and neuroinflammation. 2023.
- Lipid droplet-associated lncRNA LIPTER preserves cardiac lipid metabolism. 2023.
- Inflammation primes the kidney for recovery by activating AZIN1 A-to-I editing. 2023.
- Anti-inflammatory activity of black soldier fly oil associated with modulation of TLR signaling: A metabolomic approach. 2023.
- Plant Growth Promotion, Phytohormone Production and Genomics of the Rhizosphere-Associated Microalga, Micractinium rhizosphaerae sp. 2023.