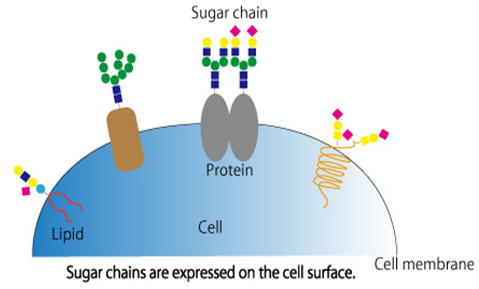
 Polysaccharides are made by polymerizing more than 10 monosaccharide molecules through glycosidic bonds. The molecular weight of polysaccharides is relatively large, usually consisting of hundreds or even tens of thousands of monosaccharide molecules. Polysaccharides, together with nucleic acids, proteins, and lipids, are called the four basic substances of life that play important roles in many life activities. Biological activities of polysaccharides are continued to be discovered. Some known activities include immune regulation, anti-tumor, lowering blood sugar, lowering blood lipids, anti-virus, eliminating oxidative free radicals, and delaying aging. Polysaccharides are found in higher plants, animals, algae and bacteria in nature and are widely distributed. Most of the polysaccharides are derived from tissue cells, which are less toxic and have side effects on cells and body, making them ideal drug sources. In recent years, with the rapid development of related disciplines such as biology and chemistry, polysaccharide compounds have also been increasingly studied. The international scientific community regards the research on polysaccharides as the frontier of life sciences, and has even proposes that the 21st century is the century of polysaccharides.
Polysaccharides are made by polymerizing more than 10 monosaccharide molecules through glycosidic bonds. The molecular weight of polysaccharides is relatively large, usually consisting of hundreds or even tens of thousands of monosaccharide molecules. Polysaccharides, together with nucleic acids, proteins, and lipids, are called the four basic substances of life that play important roles in many life activities. Biological activities of polysaccharides are continued to be discovered. Some known activities include immune regulation, anti-tumor, lowering blood sugar, lowering blood lipids, anti-virus, eliminating oxidative free radicals, and delaying aging. Polysaccharides are found in higher plants, animals, algae and bacteria in nature and are widely distributed. Most of the polysaccharides are derived from tissue cells, which are less toxic and have side effects on cells and body, making them ideal drug sources. In recent years, with the rapid development of related disciplines such as biology and chemistry, polysaccharide compounds have also been increasingly studied. The international scientific community regards the research on polysaccharides as the frontier of life sciences, and has even proposes that the 21st century is the century of polysaccharides.
Select Service
The Process of Polysaccharides Analysis
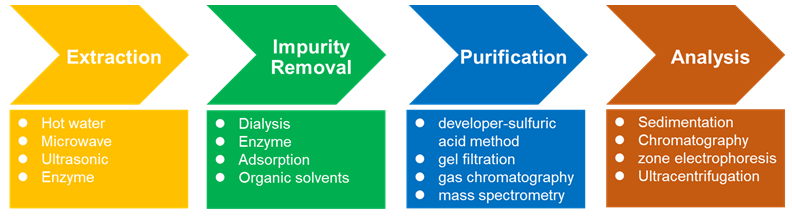
Extraction of polysaccharides
Polysaccharides are linked to the cell wall or interstitial substance by hydrogen or ionic bonds. Different extraction methods are usually used according to the existence of polysaccharides and different extraction sites. Hot water, acid, alkali, ethanol, etc. are often used as solvents, and crude extraction is assisted by microwave or ultrasound. The commonly used methods in recent years include supercritical fluid extraction technology and complex enzyme-assisted extraction technology. Using these methods, complex enzyme can specifically degrade the cell wall and the barrier for the dissolution of intracellular macromolecules in milder conditions to accelerate the release of polysaccharides. At the same time, the reaction can be controlled by changing the system conditions according to the characteristics of the enzyme.
Impurity removal of polysaccharides
Crude polysaccharide extracts usually contain impurities such as inorganic salts, lipids, proteins, and low-molecular non-polar substances. For low molecular weight impurities, dialysis can be used to remove them. Protein is usually removed by protease method, Sevag method, TCA method, and trifluorotrichloroethane method. Fat can be removed with organic solvents such as ethanol, ether, and petroleum ether. Adsorption and oxidation are common methods to remove pigment impurities.
Isolation and purification of polysaccharides
After separation and removal of impurities, a mixed polysaccharide solution can be obtained, and the process to separate the mixed polysaccharide solution into various single polysaccharides is the purification of polysaccharides. More commonly used methods include sedimentation, chromatography, zone electrophoresis, ultracentrifugation and other biochemical analysis methods. In general, polysaccharides purification requires the combination of two or more methods to optimize the results.
Analysis of polysaccharides
Polysaccharides have a wide variety of components, complex structures and large molecular weights, so they are usually analyzed from the following four aspects.
1) Sugar content determination
In order to determine the sugar content in the sample, a developer-sulfuric acid method is often used. Monosaccharides, polysaccharides, and their derivatives are hydrolyzed to monosaccharides under the action of sulfuric acid and quickly dehydrated to form aldehyde derivatives, which are condensed with phenols, aromatic amines into colored compounds. The content of polysaccharides is indirectly determined by colorimetric quantification. These methods are simple, fast and sensitive, and have good color stability of colored compounds.
2) Molecular weight determination
There is no absolute method for determining the molecular weight of polysaccharides, so the method of statistical average is generally used. Osmotic pressure method, end group method, viscosity method, and light scattering method are commonly used in the past, but the operations are complicated and causing errors. Currently, more commonly used methods are gel filtration and high-performance liquid chromatography. These two methods must use standard polysaccharides of known molecular weight as a reference. For polysaccharides with a molecular weight of less than 50,000, mass spectrometry can be used.
3) Component determination
Polysaccharide composition analysis methods can be generally divided into: traditional chemical analysis, physical analysis (instrument analysis) and biological analysis. Among them, chemical analysis includes partial or complete acid hydrolysis, neutralization and filtration. At the end, paper chromatography (PC), thin layer chromatography (TLC), gas chromatography (GC), liquid chromatography (HPLC) or ion chromatographic are implemented for analysis. Many widely used instrumental analysis include spectrophotometry, infrared spectroscopy, nuclear magnetic resonance, GC, and mass spectrometry (MS).
4) Structure determination
Polysaccharides have more complex macromolecular structures than proteins. The variety of monosaccharides, connection methods, and the complexity of branch chains make the structure determination difficult. At present, the primary structure is the target for polysaccharide structure determination, analyzing mainly the molecular weight range of polysaccharides, type, proportion, and connection order of monosaccharides, and configuration of glycosidic bonds. Common structural analysis methods are periodate oxidation, Smith degradation, and methylation reaction. Many advanced instruments used in recent years such as ultraviolet have greatly improved the analysis.
References
- Nie S, Xie M, Fu Z, et al. Study on the purification and chemical compositions of tea glycoprotein. Carbohydrate Polymers, 2008, 71(4): 626-633.
- Yang X B, Gao X D, Han F, et al. Purification, characterization and enzymatic degradation of YCP, a polysaccharide from marine filamentous fungus Phoma herbarum YS4108. Biochimie, 2005, 87(8): 747-754.








