Exosomes
Exosomes are extracellular vesicles (EVs) with a diameter of 30-140 nm. They are carriers of functional substances such as proteins, non-coding RNA, mRNA, and lipids. They act as messengers in cells, and It has the function of changing the biological activity of recipient cells. In 1980, exosomes were defined by Johnstone et al. As small vesicles that retain similar enzymatic activity as the parental cells. They undergo budding and formation of multiple vesicles (MVBs), and merge with the plasma membrane (PM), and eventually released outside the cell, at the same time, the exosome can also be internalized by endocytosis within the cell, and its contents are delivered to the target cell, thereby changing the biological function of the cell.
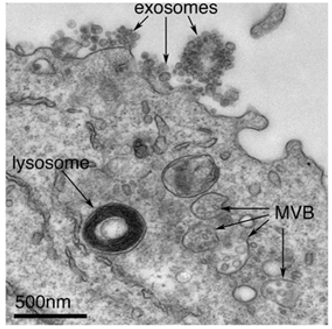
Exosomes correspond to intraluminal vesicles of multivesicular bodies (Edgar J.R, 2016).
Exosomes and Cancer
Compared with normal cells, cancer cells produce and secrete more proteins, nucleic acids, and lipids. These substances need to rely on exosomes as carriers for intercellular transport, which results in generating a large number of tumor-related exosomes. The diameter of tumor-associated exosomes is generally 30-100nm, and tumor exosomes are important mediators of the tumor microenvironment. They play an important role in the occurrence and development of cancer. It guides the interaction between different types of cells in the tumor microenvironment. Therefore, it is an important intercellular communication signaling messenger in the process of cancer cell metastasis and invasion.
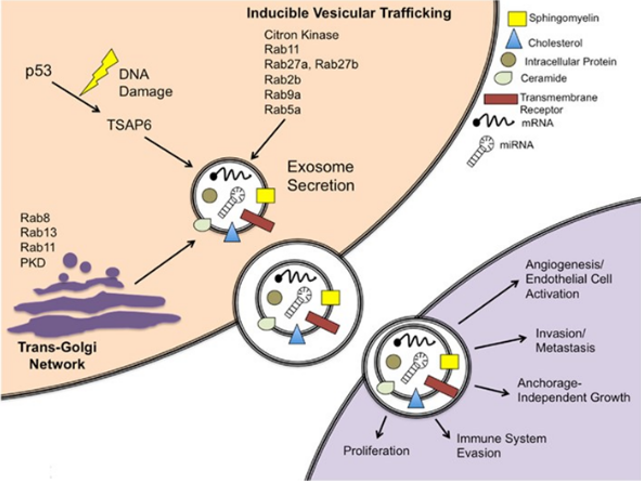
Schematic of exosome secretion in a cancer cell model (Henderson Meredith et al, 2015).
Exosomes and Tumor Microenvironment
Exosomes play a role in promoting tumor proliferation and changing the microenvironment in the tumor microenvironment during the development of the tumor. Tumor-related exosomes in the process of promoting tumor proliferation, the biological macromolecular substances contained in the exosomes are protected by the lipid bilayer. For this reason, they can be protected from enzymatic hydrolysis or other substances. After the tumor exosomes bind to the receptor, it can change the function and phenotype of the receptor cells, which in turn affects the non-tumor cells and creates a favorable tumor microenvironment for the tumor cells. In addition to promoting tumor cell proliferation, tumor-associated exosomes can change the tumor microenvironment, mainly in that they can induce the formation of blood vessels, provide nutrients and waste removal for the growth of tumor cells, and promote the transfer and invasion of cancer.
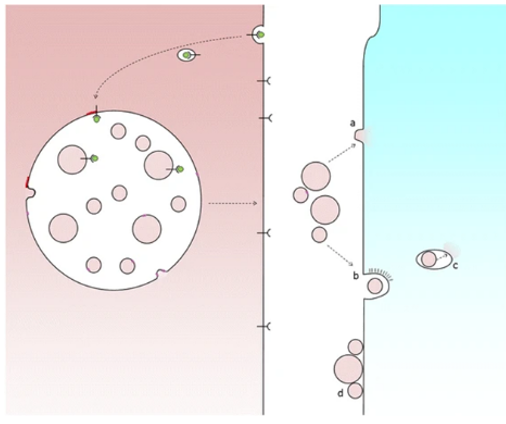
Exosome uptake by recipient cells (Edgar J.R, 2016).
Exosomes and Cancer Metastasis
The transfer of tumor cells involves multiple steps, including the detachment of tumor cells from the primary organ, invasion and migration of the basement membrane, the spread of blood flow to the adaptation to colonize the secondary organs and other processes. During the migration of tumor cells, exosomes play an active role in mediating, which is mainly reflected in the fact that exosomes can transfer key nucleic acid macromolecules in tumor cells to normal cells, thereby inducing their invasion ability. At the same time, when the derived exosomes are taken up by the receptor, they can change the phenotype of the receptor, thereby creating a good pre-metastatic niche for the invasion and metastasis of tumor cells, and completing the invasion, and after infiltrating into blood vessels, exosomes can regulate the external environment of tumor cells and promote tumor cells to reach the destination of colonization.
Exosome-based Cancer Treatment Strategies
Exosomes can be used as an intermediary for cancer treatment interventions, mainly reflecting the release of tumor-derived exosomes to control the progress of cancer, and the natural molecular structure and special biological functions of exosomes can be used as natural delivery of drug carriers. In the process of inhibiting the release of tumor-derived exosomes, it was found that targeting Rab protein can effectively inhibit the release of exosomes, and at the same time, it can use clathrin-mediated endocytosis and phagocytosis to block colleges and universities. The uptake pathway of exosomes, which in turn blocks tumor-associated exosomes, alters the tumor microenvironment and changes in receptor phenotypes, thereby inhibiting cancer progression. In terms of using exosomes as a natural carrier for cancer treatment, the natural structure of exosomes such as its unique membrane proteins and lipids can assist exosomes to bind to specific receptors, thereby improving the efficiency of drug delivery. Studies have shown that electroporation of doxorubicin (Dox) in exosomes and its application in breast cancer cell lines can find that engineered exosomes can specifically deliver Dox to tumor tissues, thereby inhibiting tumor growth, and finally achieve the goal of cancer treatment.
The Challenge of Exosomes in Cancer Treatment
Exosomes do have great advantages in cancer treatment, mainly because they can increase the targeting of drug delivery and have a higher delivery efficiency. However, its isolation and purification requirements for exosomes are relatively high, and the presence of non-exosome EVs will greatly reduce the efficiency of cancer treatment. Therefore, the establishment of standard and rapid exosome isolation technology is now faced in the field of cancer treatment challenge. At the same time, exosomes should pay attention to the source of cells in the treatment of cancer, to avoid affecting the delivery characteristics of tumor exosomes that may promote cancer progression. For this reason, accurate characterization of exosomes is also a challenge in the field of cancer treatment.
With years' experience and state-of-the-art instrumentation, Creative Proteomics can provide you with Exosome Proteomics services to support your research projects on exosomes.
References
- Edgar, J.R. Q&A: What are exosomes, exactly? BMC Biology, 2016.
- Henderson Meredith, Azorsa David. The Genomic and Proteomic Content of Cancer Cell-Derived Exosomes. Frontiers in Oncology, 2012.





