The Basic Principles of Chromatography
When the components in the mixture of solutions are moved forward in the mobile phase, or the material moving the sample, these components pass through the stationary phase. The difference in size and strength of each component causes the difference in the residence time of the component to flow out from the stationary phase.
The Types of Chromatography
According to the different separation mechanisms, chromatography can be divided into five types: adsorption, distribution, exchange, exclusion and affinity. Chromatographic separation methods include adsorption chromatography, bonded-phase chromatography, ion exchange chromatography, affinity chromatography, and size exclusion chromatography.
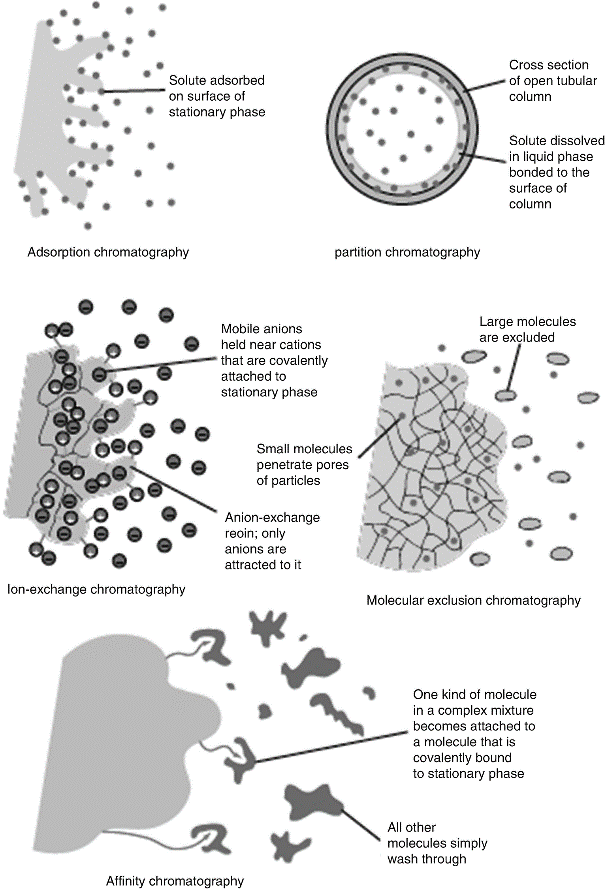
Physicochemical principles of chromatography (Broekaert J et al, 2015).
Comparison of Several Chromatographic Separation Techniques
- Affinity chromatography
Affinity chromatography is an important branch of chromatographic separation technology. It is a liquid chromatography method that uses the binding characteristics of stationary phases to adsorb target products and achieve separation and purification. For example, specific interactions between enzymes and matrices (or inhibitors), antigens and antibodies, hormones and receptors, exogenous lectins and base pairs of nucleic acids, etc. can be used to form one of the interacting substances with the insoluble support, which can be used as a stationary phase for chromatography. The other party can then be reversibly intercepted from a complex mixture to achieve the purpose of purification. Today, affinity chromatography has been widely used in the separation and purification of biomolecules. It is one of the most important methods for separating and purifying biological macromolecules, such as binding proteins, enzymes, inhibitors, antigens, antibodies, hormones, hormone receptors, glycoprotein, nucleic acid and polysaccharide.
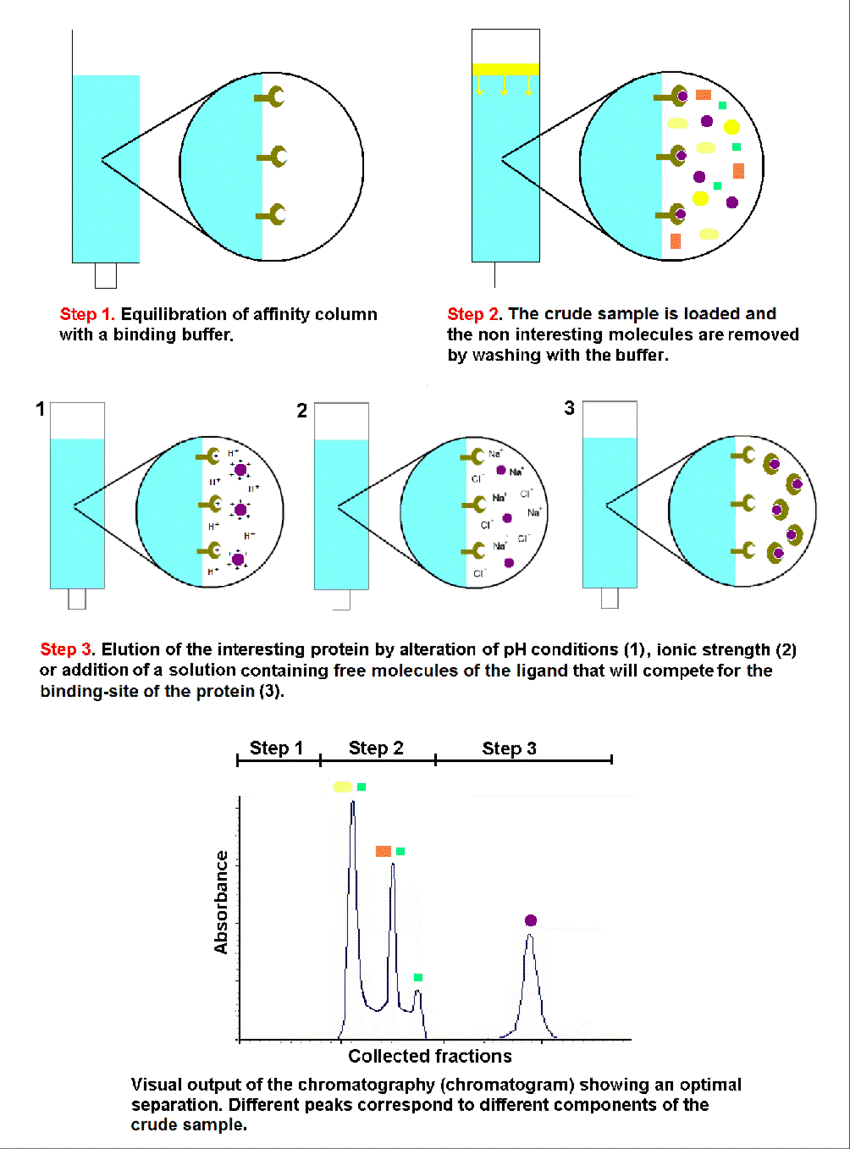
Schematic representation of the equilibration (1), adsorption/washing (2) and desorption (3) steps of an affinity chromatography for protein purification (Luana et al, 2012).
- Size exclusion chromatography
Size exclusion chromatography (SEC), also named gel chromatography, uses molecular sieves to make molecules different in size and quantity, and completes the separation of separated components according to the difference in the exclusion capability of each component. Small molecular weight compounds can enter the pores and have a long residence time; while large molecular weight compounds are prevented from entering the pores but continue flowing with mobile phase. When the eluent is an aqueous solution or buffer, it is called gel filtration chromatography (GFC), which has more applications in the biological world; when organic solvents are used as the eluent, known as gel permeation chromatography (GPC), is widely used in the polymer field. Size exclusion chromatography is often used to separate polymer compounds such as tissue extracts, peptides, proteins, and nucleic acids.
Gel filtration chromatography is based on the differences in molecular weight or molecular shape of proteins. When the sample moves down from the top of the column, large protein molecules cannot enter the gel particles and are quickly eluted; while smaller protein molecules can enter the gel particles, and the retention time of the smaller proteins that enter the gel are different. The larger the molecular weight, the earlier the elution time. Proteins with different molecular sizes are separated.
- Bonded-phase chromatography
Based on the different solubility of the separated components in the mobile and stationary phases, components are separated. This method uses a specific liquid substance to coat the surface of the support, or chemically bond to the surface of the support to form a stationary phase. Coated fixing (the first method of operation) is now rarely used. Nowadays, chemically bonded stationary phases, such as C18, C8, amino columns, cyano columns and phenyl columns, are mostly used in the industry. Liquid-liquid chromatography can be divided into normal-phase chromatography (NPC) and reverse-phase chromatography (RPC) according to the types of stationary and mobile phases. With the rapid development of column packing, the scope of application of reversed phase chromatography has gradually expanded, and it has now been applied to the analysis of some non-polar samples or easily dissociated samples.
- Adsorption chromatography
Adsorption chromatography uses a solid adsorbent to separate the components in the mixture. The commonly used adsorbent is silica gel or alumina. This method is based on the size of the fixed relative component adsorption force to separate each component. The whole process depends on the equilibrium of adsorption-desorption. This separation method is suitable for separating isomers with molecular weights of 200-1000, and separating non-ionic compounds. The limitation is that the separation of ionic compounds is prone to tailing.
Creative Proteomics has a chromatographic separation platform, which can provide you with corresponding technical services and accelerate the progress of the project.
References
- Kecskemeti A, Gaspar A. Particle-based liquid chromatographic separations in microfluidic devices - A review. Analytica Chimica Acta, 2018.
- Broekaert, J.A.C. Daniel C. Harris. Quantitative chemical analysis, 9th ed. Analytical and Bioanalytical Chemistry, 2015(407): 8943–8944.
- Luana Cassandra Breitenbach Barroso Coelho, Andréa Santos, Maria T.S. Correia, et al. Protein Purification by Affinity Chromatography, Intech Open, 2012.
- Wong, Min-Liang. Shirts off to boost separation methods. Nature, 2017, 543(7646):491-491.





