The Biochemistry of the Sphingoid Backbone
Technical Sovereignty: Membrane Physics and the Liquid-Ordered Phase
Technical Sovereignty: Bioactive Signaling — The SM–Ceramide Rheostat
Technical Sovereignty: Advanced Sphingolipidomics via LC-MS/MS
Sphingomyelin vs. Phosphatidylcholine
A Systems View: From Sphingoid Flux to Membrane Signaling Output
FAQ
Related Services
Meta Intent
An advanced research-facing guide to sphingomyelin that moves beyond basic definition and explains how sphingoid backbone chemistry, membrane asymmetry, liquid-ordered phase behavior, and species-resolved lipid analysis shape experimental interpretation in modern sphingolipidomics.
Sphingomyelin is usually introduced as a major membrane sphingolipid. That description is accurate, but it is not enough for modern lipid biology. In real membranes, sphingomyelin is not just a passive structural component. It is a membrane-organizing variable. Its sphingoid backbone changes packing geometry. Its amide linkage changes interfacial behavior. Its acyl-chain composition changes membrane thickness, nanodomain stability, and cholesterol compatibility. And when sphingomyelin is converted to ceramide, the result is not simply a metabolic exchange. It is a rapid physical rewrite of the membrane surface.
That is why sphingomyelin sits at the center of three connected fields. The first is structural biophysics, because its chemistry directly affects bilayer order. The second is signaling biology, because sphingomyelin-rich regions can stabilize or destabilize membrane platforms used by receptors, adaptors, and stress-response machinery. The third is analytical lipidomics, because class-level abundance often hides the species-level remodeling that actually drives phenotype.
A better 2026 framing is not “What is sphingomyelin?” but “How do sphingomyelin species shape membrane order, sterol distribution, and signal-permissive topology, and how can those changes be measured without analytical oversimplification?” That shift matters because two samples may show similar total sphingomyelin while behaving very differently in membranes. One may be enriched in long-chain, tightly ordering species. Another may be dominated by shorter or more dynamically turned-over species. At the class level they look similar. At the membrane level they are not.
This is also why sphingomyelin projects often expand beyond a single assay. A membrane study that starts with targeted lipidomics often grows into sphingolipid metabolism analysis, cholesterol LC-MS analysis, and sometimes integrated proteomics and lipidomics analysis when lipid remodeling is expected to affect receptor organization or membrane-proximal signaling. That workflow logic is especially important for sphingomyelin because its biological meaning comes from flux, location, and physical context rather than abundance alone.
The Biochemistry of the Sphingoid Backbone
The first principle is simple: sphingomyelin is not built on glycerol. It is built on a sphingoid base. That difference is foundational. Glycerophospholipids such as phosphatidylcholine use a glycerol backbone with two ester-linked fatty chains. Sphingomyelin uses a long-chain base, usually sphingosine, that already contributes one hydrophobic segment and a distinct polar architecture before an N-acyl fatty acid is even added. As a result, sphingomyelin enters the membrane with a different structural logic from the start.
This matters because membrane behavior is not determined by the headgroup alone. Backbone geometry, bond type, chain ordering, and interfacial hydration all matter. Sphingomyelin may share a phosphocholine headgroup with phosphatidylcholine, but the rest of the molecule is different enough to generate different packing behavior, different sterol interactions, and different domain tendencies.
Sphingosine versus ceramide: the chemical threshold that changes membrane logic
Sphingosine is the backbone platform. Ceramide is the structural threshold. Once a fatty acid is attached to the amino group of the sphingoid base through an amide bond, the molecule becomes ceramide. Addition of a phosphocholine headgroup then yields sphingomyelin.
This sequence is not just a biochemical formality. Each step changes what the molecule can do inside a membrane. The sphingoid base contributes a long aliphatic chain and a 1,3-diol motif. The ceramide step adds an amide linkage, which increases hydrogen-bonding potential and changes the local polarity of the interfacial region. The phosphocholine addition makes the lipid compatible with the outer membrane surface while preserving the structural discipline imposed by the ceramide core.
That is one reason sphingomyelin behaves differently from many glycerophospholipids. It is not simply more hydrophobic or more abundant in certain membranes. It is more structurally constrained. Its interfacial region can support stronger lateral organization. Its hydrophobic region can align efficiently with cholesterol. Its species-level chain variation has outsized effects on membrane order.
Another practical implication appears in mass spectrometry. Sphingomyelin and phosphatidylcholine both carry a phosphocholine headgroup, so both can generate the classic phosphocholine fragment at m/z 184 in positive-ion tandem MS. That shared fragment is useful at the class-diagnostic level, but it can also create false confidence. A signal that looks “phosphocholine-positive” is not automatically sphingomyelin. The backbone still matters, both structurally and analytically.
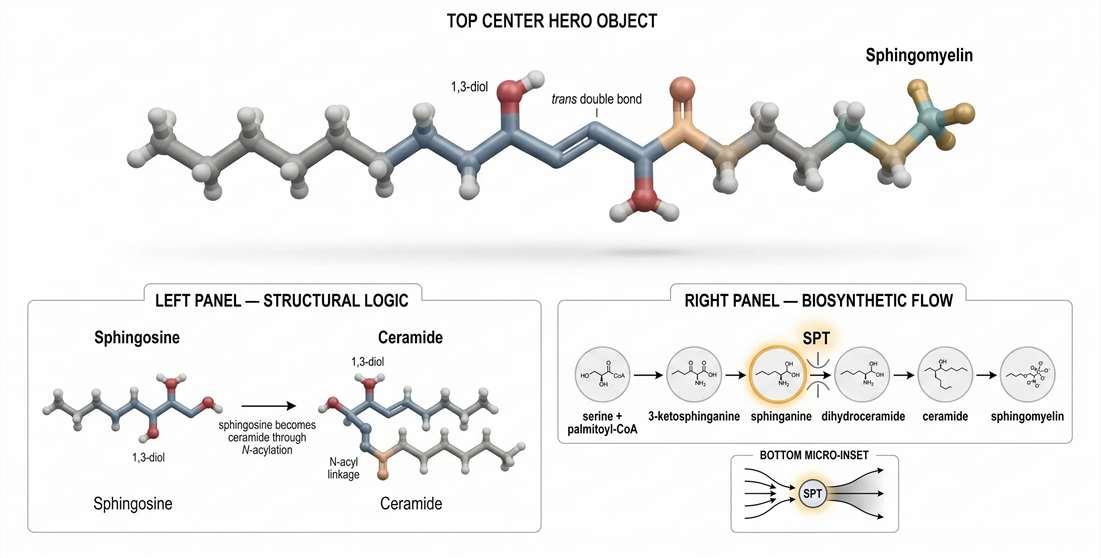
Figure 1. Sphingomyelin molecular architecture and de novo biosynthetic logic.
Biosynthetic flux: SPT is a rheostat, not a routine entry step
Many overview pages compress de novo sphingolipid synthesis into a short pathway summary. In practice, the entry step deserves much more weight. Serine palmitoyltransferase, or SPT, is not merely the first enzyme in the sequence. It functions as the rate-limiting rheostat for sphingoid backbone supply. If the input through SPT changes, the whole downstream sphingolipid landscape can change with it.
The pathway begins with condensation of L-serine and palmitoyl-CoA to produce 3-ketosphinganine. That intermediate is reduced to sphinganine, converted into dihydroceramide, desaturated to ceramide, and then diversified into downstream sphingolipid classes, including sphingomyelin. Written this way, the pathway looks linear. Functionally, it is not. It is a controlled flux architecture with upstream bottlenecks, branch decisions, and remodeling loops.
This is where many mechanistic interpretations go wrong. A rise or fall in sphingomyelin abundance is often assigned directly to sphingomyelin synthase or sphingomyelinase activity. Sometimes that is correct. But sometimes the real change starts earlier, with altered long-chain base input. If sphingoid backbone production changes, downstream ceramide and sphingomyelin pools may shift even before terminal remodeling enzymes become limiting. For that reason, species-resolved profiling is often more informative when combined with metabolic flux analysis and lipidomics pathway analysis.
Bottlenecks define phenotype better than total pool size
A useful way to think about sphingomyelin biochemistry is to divide control into three layers.
The first layer is input control. This determines how much sphingoid backbone enters the system. The second is assembly control. This determines how ceramide species are generated and how efficiently they are converted into sphingomyelin. The third is remodeling control. This determines how fast sphingomyelin is hydrolyzed, recycled, or redirected into bioactive lipids.
Phenotype emerges from the interaction of all three. A membrane can carry a large sphingomyelin pool that is kinetically stable, or a similarly sized pool that turns over quickly under stress. Those are not the same biological states. A sample can also show modest total sphingomyelin but be enriched in long-chain species that strongly favor ordered membrane organization. Again, that state is not equivalent to one dominated by shorter species. The physical consequences of the lipid pool depend on what enters the pathway, how that material is assembled, and how it is remodeled over time.
Why backbone chemistry predicts membrane behavior
The most important consequence of sphingomyelin chemistry is that it maps directly onto membrane mechanics. The sphingoid backbone and amide linkage create an interfacial architecture that favors ordered packing. That does not mean cellular membranes become rigid whenever sphingomyelin is present. It means sphingomyelin is unusually effective at supporting a membrane state that combines high local order with retained lateral mobility.
That state is the liquid-ordered phase. It is the central physical concept for understanding sphingomyelin in membranes. A phospholipid-rich liquid-disordered membrane is mobile but loosely packed. A gel phase is tightly packed but largely immobile. The liquid-ordered phase sits between those extremes. Chains remain relatively ordered, yet the membrane remains laterally dynamic enough for biological use. Sphingomyelin helps make that state possible.
This is why the phrase “sphingomyelin forms lipid rafts” is too vague to be useful on its own. A more precise statement is that selected sphingomyelin species, in the presence of cholesterol and compatible surrounding lipids, shift the membrane free-energy landscape toward local liquid-ordered organization. The outcome is not just segregation by composition. It is a change in surface readiness, protein compatibility, and signaling geometry.
Technical Sovereignty: Membrane Physics and the Liquid-Ordered Phase
The liquid-ordered phase is the most productive framework for understanding sphingomyelin. It explains why some membranes can remain dynamic while still showing high local structural order. It also explains why sphingomyelin biology cannot be reduced to total abundance.
The thermodynamics of sphingomyelin–cholesterol interaction
Sphingomyelin and cholesterol are often mentioned together, but the reason for their partnership is frequently oversimplified. They do not merely coexist in the same membrane regions because both are abundant. They interact well because their structural features are complementary.
Sphingomyelin typically presents straighter chains and a more disciplined interfacial region than highly unsaturated glycerophospholipids. Cholesterol contributes a rigid sterol ring system that suppresses chain disorder while preventing full solidification. In other words, cholesterol orders without completely freezing. Sphingomyelin provides a packing environment that makes that ordering efficient and stable.
The result is a membrane regime that is more ordered than a liquid-disordered phospholipid-rich region but less immobilized than a gel. This is the core of the liquid-ordered state. It is not a static chemical complex. It is a thermodynamically favored organization in which chain alignment, sterol positioning, and interfacial interactions all reinforce one another.
The umbrella effect helps explain part of this behavior. Cholesterol has a small polar hydroxyl group attached to a bulky hydrophobic body. In membranes, neighboring phospholipid headgroups help shield that hydrophobic structure from water. Sphingomyelin is especially compatible with this arrangement because its headgroup region and interfacial geometry support tight packing around cholesterol. The more efficient this local shielding and packing become, the more favorable ordered organization becomes.
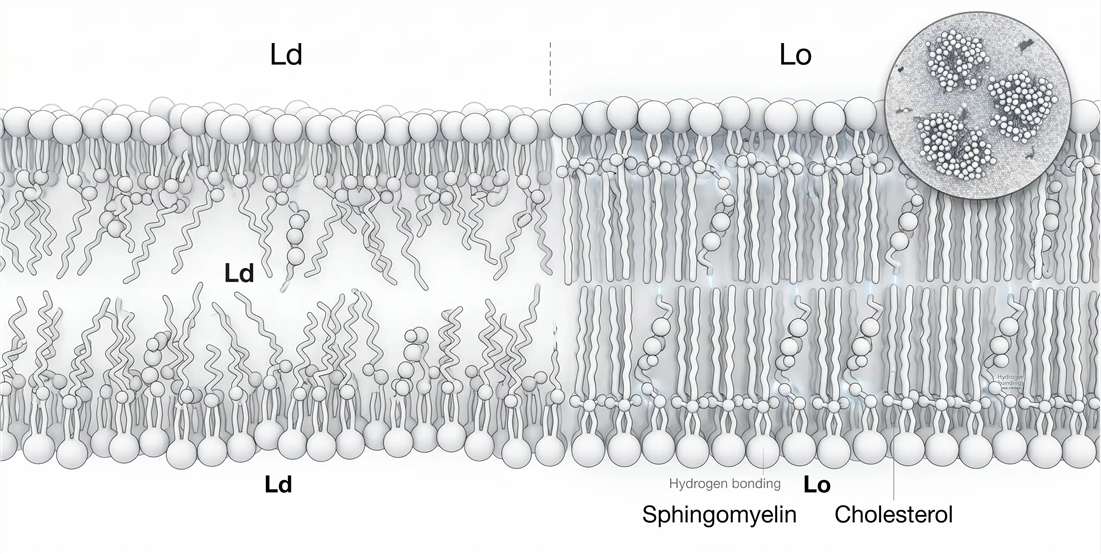
Figure 2. Liquid-ordered phase formation driven by sphingomyelin–cholesterol interactions.
The liquid-ordered phase is not a uniform sheet
One of the most useful corrections to older raft language is that ordered membrane regions are not always uniform. The liquid-ordered phase can contain local heterogeneity. That means a membrane may contain ordered neighborhoods, internal nanoscopic variation, and transient boundaries rather than one seamless, homogeneous ordered slab.
That distinction matters because proteins do not experience only average membrane order. They experience local order, local defects, local thickness, and local residence time. A receptor may prefer the edge of an ordered nanodomain rather than its center. A lipidated adaptor may partition into one ordered region but not another. A protein complex may stabilize only when surrounding order stays above a threshold for long enough. Once the problem is framed this way, sphingomyelin becomes more than a composition variable. It becomes a domain-regulating variable.
Phase separation kinetics depend on structure, not just class identity
Not all sphingomyelin species support the liquid-ordered phase equally. Acyl-chain length matters. Saturation matters. Hydroxylation can matter. Even subtle changes in interfacial structure can alter packing density, bilayer thickness, and sterol accommodation.
Longer, more saturated sphingomyelin species generally favor tighter packing and more stable ordered regions. Shorter species weaken that tendency. If the interfacial region becomes more capable of organized hydrogen-bond-like interactions, ordered domain stability can increase further. These changes influence how readily a membrane shifts from a more disordered state toward an ordered one, how large ordered regions become, and how long they persist.
This is why total sphingomyelin is often a misleading mechanistic endpoint. Two samples with the same class-level abundance can occupy very different membrane states if their species composition differs. One may support thicker, more cohesive ordered domains. Another may support smaller, shorter-lived domains with weaker sterol retention. The biological readout can diverge sharply even when the class total looks stable.
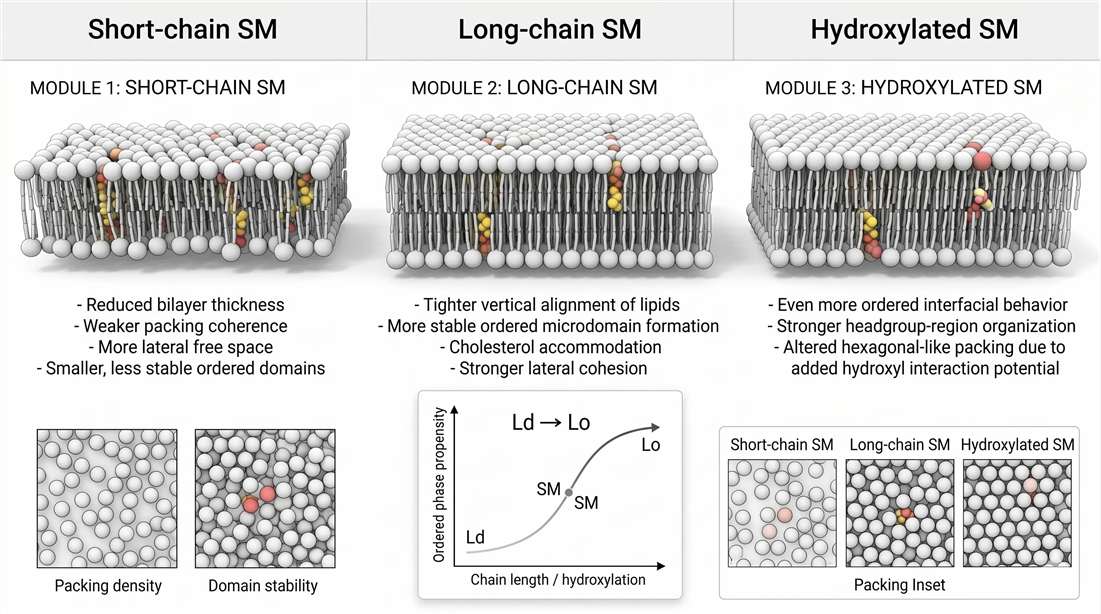
Figure 3. Phase separation kinetics and structural variables controlling membrane order.
Lipid rafts are a membrane mechanics problem before they are a signaling label
The raft concept is often discussed in the language of signaling, but the underlying issue is physical. Cells need membrane regions that are neither fixed solids nor featureless fluids. They need local environments that can recruit selected proteins, exclude others, and change on demand. Sphingomyelin helps create those environments because it promotes order, increases local thickness, and cooperates with cholesterol to form membranes with distinct lateral organization.
That change in physical environment can have direct biological consequences. Proteins with longer transmembrane segments may partition differently into thicker ordered regions. Peripheral proteins with lipid anchors may show altered membrane residence time. Receptor complexes may cluster more efficiently when the surrounding bilayer reduces entropic noise and stabilizes lateral proximity. Conversely, if sphingomyelin-rich order is disrupted, a receptor pathway may weaken even when receptor abundance is unchanged.
This is why membrane composition studies often benefit from direct pairing with protein-side measurements. In practice, sphingomyelin-focused projects may expand into membrane protein identification, cell surface proteomics, or subcellular proteomics when the central question shifts from “what lipids changed?” to “what membrane organization changed?”
Membrane asymmetry is not a detail. It is part of the mechanism.
One of the most important features of sphingomyelin is its preferential enrichment in the exoplasmic leaflet of the plasma membrane. This asymmetry changes how the entire bilayer behaves. If one leaflet is enriched in a lipid that strongly supports ordered packing, then the other leaflet does not remain unaffected. The two leaflets are coupled by thickness constraints, hydrophobic matching, sterol exchange, and in some cases direct interdigitation of long chains.
This becomes especially important for very-long-chain sphingomyelin species such as C24 forms. These molecules can extend deeply into the bilayer and may partially interdigitate into the opposite leaflet. Once that happens, the outer leaflet is no longer simply an outer leaflet. It becomes a mechanical influence on the whole membrane.
A useful consequence of this model is that outer-leaflet sphingomyelin composition can alter how cholesterol partitions across the bilayer. In asymmetric membranes, long-chain sphingomyelin in the outer leaflet can bias sterol distribution toward the inner leaflet. That means the sterol economy of the membrane is not determined by cholesterol alone. It is co-authored by leaflet-specific sphingomyelin structure.
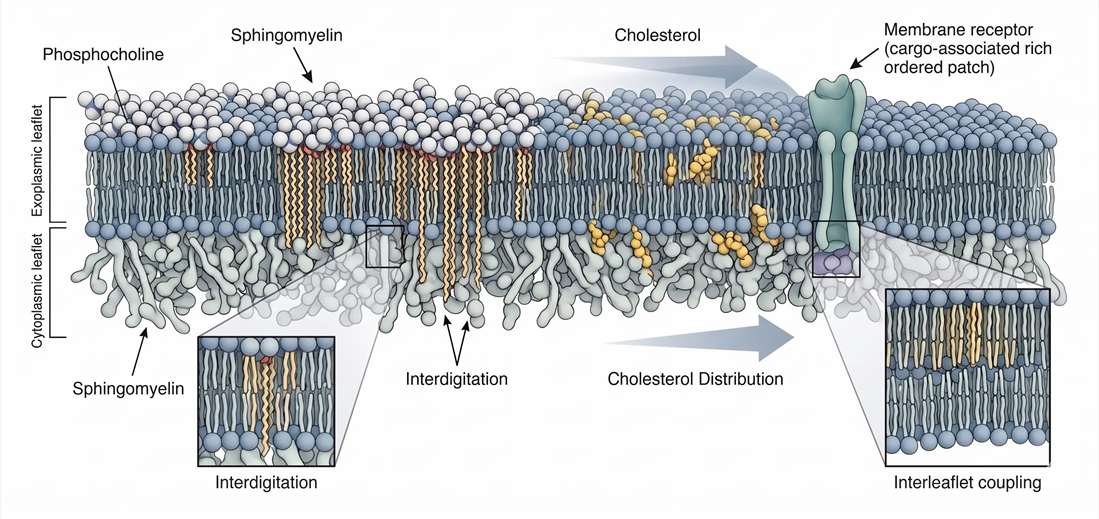
Figure 4. Membrane asymmetry, outer leaflet enrichment, and interleaflet coupling.
Why asymmetry changes trafficking and signaling logic
Once sphingomyelin asymmetry is treated as a core variable, several membrane questions become easier to frame. Why do some receptors change localization after lipid remodeling without obvious expression changes? Why do some trafficking phenotypes emerge after subtle chain-length shifts? Why do some signaling effects appear first at the extracellular membrane surface and only later in the cytoplasmic response network?
A strong answer is that the membrane is not a neutral solvent. It is a layered physical filter. The exoplasmic leaflet, when enriched in sphingomyelin, presents a specific combination of order, hydration, sterol compatibility, and thickness. Transmembrane proteins, extracellular domains, glycans, and membrane-associated complexes all interact with that filter. When sphingomyelin species shift, the filter shifts.
This is where multi-omics can become especially useful. A project that combines untargeted lipidomics, integrated transcriptomic and lipidomics analysis, and selected protein-side assays can distinguish between a simple abundance change and a deeper membrane-state transition. In receptor trafficking systems, that distinction is often the difference between a descriptive dataset and a mechanistic one.
From membrane order to membrane readiness
The real significance of sphingomyelin is not that it creates order for its own sake. It creates readiness. Ordered domains can recruit or exclude proteins. They can stabilize receptor proximity. They can delay or accelerate lateral mixing. They can change how easily a membrane accommodates curvature-sensitive events. And because sphingomyelin is metabolically connected to ceramide, the same membrane that supports ordered structure can be rapidly converted into one primed for condensation, curvature, and stress signaling.
That transition is the critical bridge to the second half of the article. Sphingomyelin is not only a builder of ordered membrane platforms. It is also the substrate for one of the fastest lipid-based state changes in the cell. When sphingomyelinase acts, membrane order does not just decline or increase in a linear way. The topology of the membrane can change. Domain architecture can reorganize. Curvature can emerge. Signaling thresholds can shift.
Technical Sovereignty: Bioactive Signaling — The SM–Ceramide Rheostat
If the first half of the sphingomyelin story is about membrane order, the second half is about reversible state change. Sphingomyelin is not only a structural lipid that helps stabilize organized membrane environments. It is also a stored signaling substrate. That dual identity gives the SM–ceramide axis unusual power. A cell can use sphingomyelin to maintain ordered membrane architecture under baseline conditions, then rapidly convert part of that same pool into ceramide during stress, injury, inflammatory signaling, or receptor-driven remodeling. Chemically, that is a hydrolysis event. Physically, it is a membrane-state switch.
This is why the term SM–ceramide rheostat is still useful when handled carefully. It does not mean that sphingomyelin is always “pro-survival” and ceramide is always “pro-death” in a simplistic binary sense. It means that the balance between these two lipid states can change how a membrane behaves, what proteins it favors, how much curvature it tolerates, and how easily signaling assemblies form. A sphingomyelin-rich patch and a ceramide-rich patch are not interchangeable surfaces. Their interfacial geometry differs. Their sterol compatibility differs. Their condensation behavior differs. And those differences feed directly into signaling logic.
Sphingomyelinase kinetics: a fast way to change membrane physics
Sphingomyelinases cleave sphingomyelin into ceramide and phosphocholine. That reaction looks chemically straightforward, but its membrane consequence is disproportionately large. Before hydrolysis, the membrane region contains sphingomyelin with a bulky phosphocholine headgroup and a cholesterol-compatible ordered packing tendency. After hydrolysis, that same region becomes enriched in ceramide, which has a much smaller headgroup and a stronger tendency toward self-association.
The effect is immediate at the level of surface behavior. The headgroup environment becomes less bulky. Local hydration changes. Lateral condensation increases. A region that previously functioned as an ordered but still cholesterol-friendly platform may become a ceramide-rich condensate with very different protein preferences and curvature behavior.
That is why sphingomyelinase activity is better described as a physical switch than as a simple catabolic event. In stress biology, the timing matters just as much as the endpoint. A transient hydrolysis pulse can destabilize one membrane platform and nucleate another. Receptor residency can change. Lateral diffusion constraints can change. The membrane can become more competent for budding, invagination, or high-order clustering of signaling proteins.
This is also where assay design starts to matter. If the hypothesis centers on rapid sphingomyelin turnover rather than only steady-state abundance, a broader discovery screen often needs support from ceramide analysis, targeted lipidomics, and, in pathway-centered studies, signaling molecule analysis. Otherwise, the biological interpretation may collapse a dynamic membrane event into a flat class-level measurement.
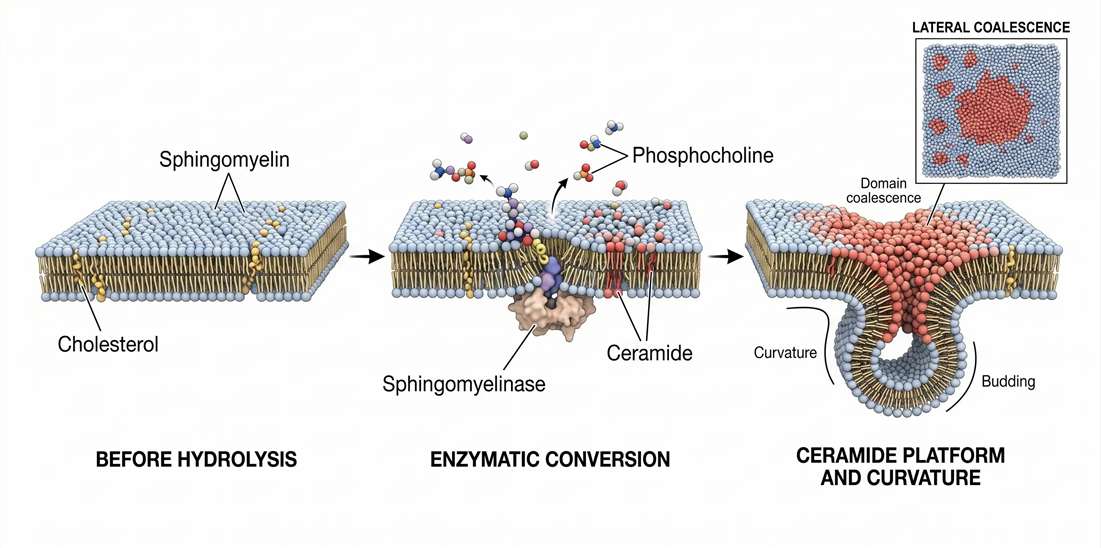
Figure 5. Sphingomyelinase-driven conversion to ceramide and membrane curvature remodeling.
Why ceramide behaves like a membrane condenser
Ceramide is often introduced as a bioactive lipid mediator, but that phrasing only becomes meaningful when its membrane physics are made explicit. Ceramide has a relatively small polar headgroup compared with sphingomyelin. It can pack tightly. It can self-associate efficiently. And it does not present the same phosphocholine-dominated interfacial surface to the aqueous phase. The result is a strong tendency toward local condensation.
That condensation matters because small changes in local packing can scale into much larger changes in membrane organization. A ceramide-enriched area may begin as nanoscopic remodeling. But once local concentration rises and neighboring ceramide molecules start to coalesce, the membrane can transition into a larger platform with altered line tension, altered sterol compatibility, and altered protein partitioning behavior. What begins as chemistry becomes mesoscale membrane reorganization.
A useful way to frame this is to split the event into two linked stages.
First, there is conversion: sphingomyelin is hydrolyzed into ceramide.
Second, there is reconstruction: the membrane reorganizes around the physical properties of ceramide.
The second stage is often where the mechanistic interpretation lives. Two experiments may detect similar ceramide increases, yet only one produces a strong signaling phenotype because only one crosses the threshold required for domain coalescence or curvature generation.
The rheostat is spatial, not just quantitative
The phrase “SM–ceramide rheostat” can mislead readers into thinking of a simple slider. More sphingomyelin, less ceramide, one phenotype. More ceramide, less sphingomyelin, another phenotype. Real systems are more spatial than that.
The impact of sphingomyelin hydrolysis depends on where it occurs, which species are involved, how large the remodeled region becomes, and how long the state persists. A small ceramide pulse in an already ordered exoplasmic patch may have a larger structural effect than a larger pulse in a disordered internal membrane. A remodeling event involving very-long-chain species may propagate mechanical consequences differently from one involving shorter chains. A transient change may prime signaling without obvious membrane deformation, while sustained remodeling may support budding or vesicle release.
This spatial logic is one reason membrane-state projects often benefit from pairing lipid readouts with protein-state readouts. If a sphingomyelin-to-ceramide transition is expected to alter receptor clustering or stress-complex assembly, a stronger design may combine lipid profiling with phosphoproteomics service, protein-protein interaction analysis, or IP-MS protein interactomics solution. That kind of design asks the right question: not just whether the lipids changed, but whether the signaling architecture built on those lipids changed.
Ceramide-induced curvature: why lipid shape becomes membrane shape
One of the most important consequences of the sphingomyelin-to-ceramide switch is curvature remodeling. This follows from molecular geometry. Sphingomyelin carries a bulky phosphocholine headgroup and supports relatively broad, ordered packing at the membrane interface. Ceramide reduces headgroup bulk while maintaining strong hydrophobic packing. As ceramide accumulates, the effective shape of the local lipid population changes.
That shift pushes the membrane toward negative curvature. In practical terms, it makes the bilayer more competent for inward bending, neck formation, invagination, and budding-related topology changes. This does not mean ceramide alone explains every vesiculation event. Protein crowding, cytoskeletal tension, leaflet asymmetry, and sterol content all contribute. But ceramide can lower the energetic barrier for curvature by condensing the membrane and altering interfacial area balance.
This is why ceramide repeatedly appears in discussions of membrane stress, endocytic-like remodeling, extracellular vesicle formation, and stress-induced budding. The key insight is that curvature is not merely an output layered on top of lipid chemistry. It is one of the direct physical consequences of lipid chemistry when enough remodeling accumulates in one place.
Budding, invagination, and domain coalescence
Ceramide-rich domains do not always produce the same morphological result. In some systems, the dominant phenotype is lateral platform formation. In others, invagination becomes more obvious. In still others, budding and vesicle release are the clearest outcomes. The difference usually reflects local context: protein crowding, membrane tension, cytoskeletal attachment, sterol distribution, and the exact species composition of the ceramide pool.
Even so, the general sequence is often recognizable. A sphingomyelin-rich ordered patch exists. Sphingomyelinase acts locally. Ceramide begins to accumulate. Small ceramide-rich nanodomains merge. Lateral condensation rises. Boundary tension changes. Curvature becomes easier to generate. And the membrane shifts into a state more compatible with budding or signal-amplifying platform formation.
For experimental interpretation, that sequence matters because it explains why endpoint measurements can be deceptive. A class-level decrease in sphingomyelin and increase in ceramide may look modest, yet the topological consequence can be large if the remodeling is spatially concentrated. That is another reason species-aware sphingolipid metabolism analysis is often more informative than broad totals alone.
Technical Sovereignty: Advanced Sphingolipidomics via LC-MS/MS
Modern sphingomyelin research eventually becomes an analytical problem. The membrane physics may be elegant, but experiments still stand or fall on readout quality. In practice, that means LC-MS/MS. And once sphingomyelin enters LC-MS/MS workflows, one analytical reality becomes unavoidable: phosphocholine chemistry makes the class easy to detect, but not always easy to assign cleanly.
ESI ionization physics: why positive mode is the natural entry point
Sphingomyelin carries a phosphocholine headgroup with strong positive-ion response characteristics. That makes positive electrospray ionization the natural starting point for many profiling workflows. In positive mode, sphingomyelin generally produces strong precursor response and recognizable tandem-MS behavior. For routine lipidomics, that is a major advantage. It improves detectability, supports class-level screening, and makes sphingomyelin one of the more accessible sphingolipid classes in broad LC-MS pipelines.
But strong ionization can create false confidence. Good signal is not the same thing as high-confidence structural interpretation. Sphingomyelin often looks analytically cooperative at first glance, yet the difficult part begins once the workflow moves from class detection to species annotation.
This is where method design starts to matter. A discovery-oriented untargeted lipidomics workflow can reveal whether sphingomyelin remodeling is likely present. But when the biological question depends on precise species calls, source efficiency alone is not enough. Extraction conditions, chromatographic separation, collision energy, adduct handling, internal standards, and annotation rules all become consequential.
MS/MS fragmentation deconvolution: why m/z 184.1 is important and insufficient
The phosphocholine fragment at m/z 184.1 is the best-known tandem-MS feature in sphingomyelin analysis. It is useful because it confirms the presence of a phosphocholine-containing lipid class in positive-ion fragmentation. In many workflows, it serves as a reliable class-level diagnostic anchor.
The problem is that sphingomyelin is not the only lipid that produces it. Phosphatidylcholine generates the same phosphocholine fragment. So while m/z 184.1 is highly informative, it does not uniquely identify sphingomyelin. It tells the analyst that a phosphocholine headgroup is present. It does not, by itself, prove that the backbone is sphingoid. It does not define the N-acyl composition. And it does not solve ambiguity arising from closely related precursors in complex matrices.
This is the central deconvolution challenge in sphingomyelin analysis. The fragment is not wrong. It is incomplete. Problems arise when a workflow treats the 184.1 signal as a stand-alone sphingomyelin identifier instead of a class-diagnostic clue that must be combined with additional evidence.
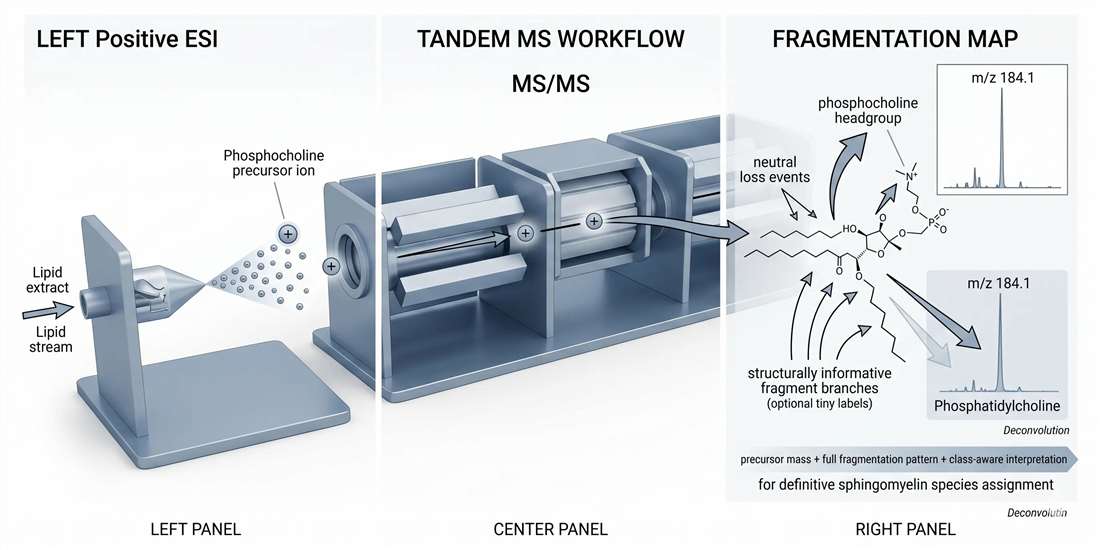
Figure 6. LC-MS/MS sphingomyelin fingerprinting and diagnostic fragmentation logic.
What reliable sphingomyelin assignment actually requires
A robust sphingomyelin call usually depends on several layers of evidence used together.
The first is precursor mass logic. The intact mass constrains which class and species are even plausible. The second is fragment pattern logic. The phosphocholine ion supports class placement, but additional fragments and neutral-loss relationships help refine species interpretation. The third is chromatographic logic. Retention behavior can help separate closely related lipids and provide an orthogonal check when the method is well characterized. The fourth is context logic. The assignment should make biochemical sense inside the observed lipid landscape.
In practical terms, confident deconvolution often means integrating:
- precursor ion mass
- phosphocholine fragment presence
- neutral-loss behavior
- supporting fragment pattern
- retention time behavior
- isotope correction
- class-aware annotation rules or curated libraries
That is the correct way to use m/z 184.1. Keep it as a core signal, but never let it function as the only signal.
Species-level sphingomyelin profiling is harder than it looks
At the class level, sphingomyelin can seem analytically simple. It ionizes well. The headgroup is familiar. The MS/MS signature looks recognizable. But the real biological questions usually live at the species level, and that is where complexity rises fast.
Chain length matters. Unsaturation matters. Hydroxylation matters. Near-isobaric possibilities matter. Matrix effects matter. Relative abundance of co-eluting phosphocholine-containing lipids matters. And once the biological claim depends on a specific SM-to-ceramide remodeling pattern, annotation discipline becomes non-negotiable.
The most common failure mode is not that sphingomyelin is missed entirely. It is that it is oversimplified. A dataset may return a clean list of putative sphingomyelin species while still carrying unresolved ambiguity in class separation, isotopic contribution, or species-level assignment. That may be acceptable for an initial survey. It is not strong enough when the manuscript or decision path depends on a particular mechanistic interpretation.
This is why broad screens often need a second stage. Discovery may begin with lipidomics service or untargeted lipidomics, but high-confidence interpretation often benefits from follow-up targeted lipidomics, phospholipids analysis, or dedicated sphingolipid metabolism analysis.
Positive mode helps, but preprocessing and normalization still decide outcome quality
Even excellent LC-MS acquisition can be weakened by poor downstream handling. That is especially true in sphingomyelin projects, where subtle species shifts may carry major mechanistic meaning. If peak grouping is inconsistent, isotope correction is weak, batch normalization is underpowered, or statistical filtering is too generic, the final interpretation can flatten the real biological pattern.
This is why advanced sphingomyelin workflows often need explicit data-analysis support rather than just instrument time. In cohort studies or perturbation series, bioinformatic data preprocess and normalization service, multivariate analysis service, and statistical analysis service can make the difference between a noisy lipid list and a defensible membrane-state model.
When untargeted becomes insufficient
Untargeted lipidomics is often the right starting point because it provides breadth and discovery power. But sphingomyelin biology frequently reaches a point where breadth is no longer enough. That point arrives when the claim depends on a specific species family, a subtle chain-length shift, a ceramide-linked conversion pattern, or a phosphocholine deconvolution-sensitive readout.
At that stage, a stronger workflow usually becomes layered rather than singular. An initial untargeted lipidomics pass defines the landscape. A follow-up targeted lipidomics panel confirms priority species. A cholesterol LC-MS analysis module can test the sterol-coupling hypothesis. And if pathway interpretation becomes central, integrated transcriptomic and lipidomics analysis can help distinguish enzyme-driven remodeling from passive compositional drift.
Sphingomyelin vs. Phosphatidylcholine
Sphingomyelin and phosphatidylcholine are often grouped together because both are major membrane phosphocholine lipids. That shared headgroup is real, but it can obscure deeper differences. Structurally, biophysically, and analytically, these lipids are not interchangeable.
| Parameter | Sphingomyelin | Phosphatidylcholine |
| Backbone architecture | Sphingoid base with amide-linked fatty acyl chain | Glycerol backbone with two ester-linked fatty acyl chains |
| Interfacial behavior | More structurally constrained, often more order-promoting | More variable and highly dependent on chain composition |
| Phase tendency | Strong contributor to liquid-ordered behavior with cholesterol | Often supports more fluid behavior unless highly saturated |
| Cholesterol partnership | Frequently strong in ordered membrane environments | Variable and often less dominant in nanodomain stabilization |
| Role in membrane thickness | Commonly associated with thicker, more ordered regions | More compatible with thinner or more disordered regions depending on species |
| Leaflet distribution | Strong enrichment in exoplasmic leaflet | Broader membrane distribution |
| Signaling connection | Direct precursor reservoir for ceramide generation | Not part of the SM–ceramide rheostat |
| MS/MS hallmark | Produces m/z 184.1; requires class-aware species interpretation | Also produces m/z 184.1; overlaps with SM at headgroup-fragment level |
| Interpretation risk | Assuming phosphocholine fragment proves sphingomyelin | Assuming headgroup similarity means membrane-behavior similarity |
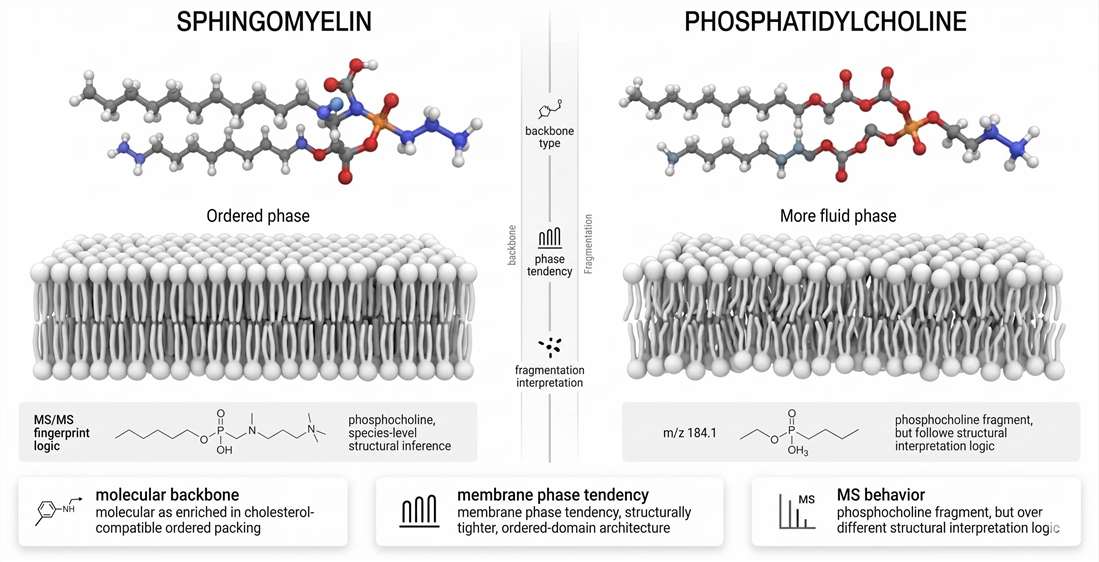
Figure 7. Sphingomyelin versus phosphatidylcholine: structure, phase behavior, and MS behavior.
The most important lesson from this comparison is that headgroup similarity does not equal functional equivalence. The same phosphocholine fragment can sit on two very different molecular frameworks. Those frameworks create different membrane states and require different analytical logic.
A Systems View: From Sphingoid Flux to Membrane Signaling Output
The real strength of sphingomyelin as a research topic is that it links multiple biological scales without losing mechanistic coherence.
At the metabolic scale, sphingoid backbone synthesis determines how much material enters the pathway. At the species scale, ceramide assembly and phosphocholine transfer determine which sphingomyelin molecules are produced. At the membrane scale, those species shape cholesterol accommodation, liquid-ordered domain stability, leaflet coupling, and sterol asymmetry. At the signaling scale, sphingomyelinase-driven hydrolysis converts a structural pool into a ceramide-rich stress-responsive state. And at the analytical scale, LC-MS/MS translates that remodeling into a species-resolved readout that can be compared across samples, interventions, and cohorts.
That continuity is what makes sphingomyelin such a valuable molecule for advanced lipid biology. It is not only a membrane constituent. It is a bridge between flux, structure, topology, signaling, and measurement.
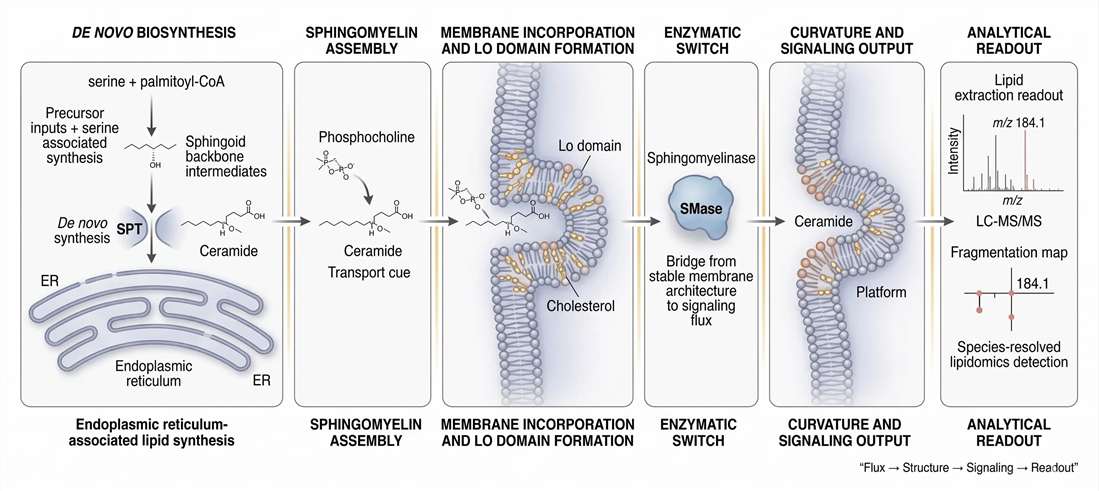
Figure 8. End-to-end systems map from sphingoid backbone synthesis to membrane signaling and lipidomics readout.
For the same reason, strong sphingomyelin projects are often multidisciplinary by design rather than by accident. A single biological question may begin with mammals untargeted lipidomics, expand into sterol lipids analysis service, move into integrated proteomics and lipidomics analysis, and then require network analysis service to connect lipid remodeling to phenotype-level interpretation. In that sense, sphingomyelin is not merely a lipid class to be measured. It is an organizing principle for membrane-state research.
FAQ
What makes sphingomyelin structurally different from phosphatidylcholine?
Sphingomyelin uses a sphingoid backbone and an amide-linked fatty acyl chain, while phosphatidylcholine uses a glycerol backbone with two ester-linked fatty acyl chains.
Why is sphingomyelin strongly associated with liquid-ordered domains?
Because its molecular architecture supports tight packing and efficient cholesterol accommodation, allowing a membrane to remain ordered without becoming fully rigid.
Does total sphingomyelin abundance predict membrane behavior well?
Not reliably. Species composition, chain length, localization, and turnover can all change membrane behavior even when total sphingomyelin looks stable.
Why is outer-leaflet sphingomyelin so important?
Because exoplasmic-leaflet enrichment affects whole-bilayer mechanics through interleaflet coupling, sterol redistribution, and in some cases long-chain interdigitation.
What is the SM–ceramide rheostat?
It is the functional balance between sphingomyelin-rich structural states and ceramide-rich remodeled states. Changes in that balance alter membrane order, signaling readiness, and curvature behavior.
Why does sphingomyelinase activity have such a large physical effect?
Because it removes the bulky phosphocholine headgroup and generates ceramide, which condenses efficiently and can promote domain coalescence and curvature formation.
Why is m/z 184.1 important in sphingomyelin MS/MS?
Because it is the classic phosphocholine fragment in positive mode and is a useful diagnostic signal for phosphocholine-containing lipids.
Why is m/z 184.1 not enough to identify sphingomyelin by itself?
Because phosphatidylcholine also produces the same fragment. Correct annotation needs precursor mass, fragment pattern, retention behavior, and class-aware interpretation.
When should untargeted sphingomyelin work be followed by targeted analysis?
When the biological claim depends on specific species, subtle remodeling patterns, SM-to-ceramide conversion logic, or phosphocholine deconvolution-sensitive assignments.
References
- Hannun YA, Obeid LM. Sphingolipids and their metabolism in physiology and disease. Nat Rev Mol Cell Biol. 2018;19(3):175–191. DOI: 10.1038/nrm.2017.107 (Nature)
- Slotte JP. Biological functions of sphingomyelins. Prog Lipid Res. 2013;52(4):424–437. DOI: 10.1016/j.plipres.2013.05.001 (Scilit)
- Róg T, Vattulainen I. Cholesterol, sphingolipids, and glycolipids: what do we know about their role in raft-like membranes? Chem Phys Lipids. 2014;184:82–104. DOI: 10.1016/j.chemphyslip.2014.06.003
- Castro BM, Prieto M, Silva LC. Ceramide: a simple sphingolipid with unique biophysical properties. Prog Lipid Res. 2014;54:53–67. DOI: 10.1016/j.plipres.2014.01.004 (PubMed)
- Pinto SN, Silva LC, Futerman AH, Prieto M. Effect of ceramide structure on membrane biophysical properties: The role of acyl chain length and unsaturation. Biochim Biophys Acta Biomembr. 2011;1808(11):2753–2760. DOI: 10.1016/j.bbamem.2011.07.023 (PubMed)
- Courtney KC, Pezeshkian W, Raghupathy R, et al. C24 sphingolipids govern the transbilayer asymmetry of cholesterol and lateral organization of model and live-cell plasma membranes. Cell Rep. 2018;24(4):1037–1049. DOI: 10.1016/j.celrep.2018.06.104 (Cell)
- Hutchison JM, Shih KC, Scheidt HA, Fantini J, Epand RM. Insights into the molecular mechanism of sphingomyelin and cholesterol interactions. Biochimie. 2022;203:120–128. DOI: 10.1016/j.biochi.2022.09.003
- Murata M, Matsumori N, Kinoshita M, London E. Molecular substructure of the liquid-ordered phase formed by sphingomyelin and cholesterol: sphingomyelin clusters forming nano-subdomains are a characteristic feature. Biophys Rev. 2022;14:703–716. DOI: 10.1007/s12551-022-00967-1 (Springer)
- Ekroos K, Ejsing CS, Bahr U, et al. Charting molecular composition of phosphatidylcholines by fatty acid scanning and ion trap MS3 fragmentation. J Lipid Res. 2003;44(11):2181–2192. DOI: 10.1194/jlr.D300020-JLR200 (PubMed)
- Hsu FF. Mass spectrometry-based shotgun lipidomics – a critical review from the technical point of view. Anal Bioanal Chem. 2018;410:6387–6409. DOI: 10.1007/s00216-018-1252-y (PMC)
- Merrill AH Jr. Sphingolipid and Glycosphingolipid Metabolic Pathways in the Era of Sphingolipidomics. Chem Rev. 2011;111(10):6387–6422. DOI: 10.1021/cr2002917 (X-MOL)
Related Services
- Lipidomics Service
- Untargeted Lipidomics
- Mammals Untargeted Lipidomics
- Targeted Lipidomics
- Sphingolipid Metabolism
- Membrane Protein Identification
- Cell Surface Proteomics
- Subcellular Proteomics
- Metabolic Flux Analysis (MFA)
- Integrated Proteomics and Lipidomics Analysis
- Bioinformatic Data Preprocess and Normalization Service
For Research Use Only. Not for use in diagnostic procedures.
More Articles
- What is Phytosterols
Learn about phytosterols - plant-based compounds that have cholesterol-lowering properties and potential health benefits. - Preparation and Development of GC Capillary Column
Learn about the preparation and development of GC capillary columns, an important component in gas chromatography analysis. - What Is Acyl-CoAs Metabolism
Discover the vital role of Acyl-CoAs in cellular metabolism, including fatty acid oxidation, synthesis, cholesterol metabolism, and ketogenesis. Learn how Creative Proteomics' innovative Acyl-CoAs assay can advance your research.


