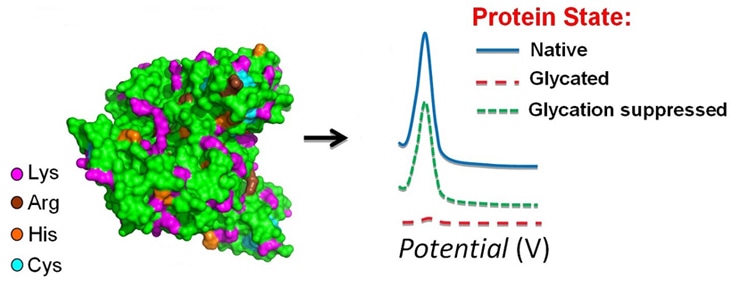
As one of the chemical modifications, glycation, initially reported by Maillard, refers to the nonenzymatic reaction of sugars with proteins. This process, involves a series of stages starting with the reaction between the reducing carbonyl group of a carbohydrate, and an amino group present on proteins, usually at ε-amine of lysine residues or the N-terminus. Since glycation is one of main resources of protein heterogeneity, it’s quite important to monitor the glycation states of protein therapeutics.
Advances in the LC-MS system, have enabled the detection of specific chemical modifications at any given residue within a therapeutic protein. As a non-enzymatic reaction between reducing sugars and specific sites of proteins, glycation may occur during cell culture, formulation and storage, where reducing sugars are used or generated from non-reducing sugars. In principle, glycation susceptibility is altered by the surface accessibility of the Lys residues, and can also be catalyzed by amino acids in close proximity. Glycation can be controlled by optimizing cell culture conditions. A mAb containing 17% glycation did not show any structural difference compared with the main peak with no glycation. However, glycation increases the propensity of aggregation of recombinant mAbs. Several studies demonstrated glycation of Lys in various CDR regions from 10% glycation to about 100% had no effect on antigen binding and potency. Extensive glycation has no effect on binding to FcgRIIIa and FcRn and protein A. However, those results can only demonstrate that those Lys residues are not critical for various ligand binding. A significant effect is expected if the glycated residues are localized in the binding pockets because of the loss of the positive charge of Lys upon glycation. Acidic species with 17% glycation did not show.
Creative Proteomics, are glad to learn more details about your projects, and issue the optimal analytical proposal, to speed up your research.





