PCR Based Antibody Sequencing
- Home
- Services
- Antibody Sequencing
- PCR Based Antibody Sequencing
Service Details
Hybridoma cells are susceptible to contamination, gene loss, poor cell condition and even death during storage, resulting in the permanent loss of important mAbs. Therefore, obtaining the gene sequences of monoclonal antibodies from hybridoma cells is of great significance for the large-scale stable expression of monoclonal antibodies. Compared with mass spectrometry sequencing, monoclonal antibody gene sequencing based on PCR amplification technology can provide more accurate and reliable results, and can more accurately distinguish leucine/isoleucine of amino acids that are difficult to distinguish in mass spectrometry identification.
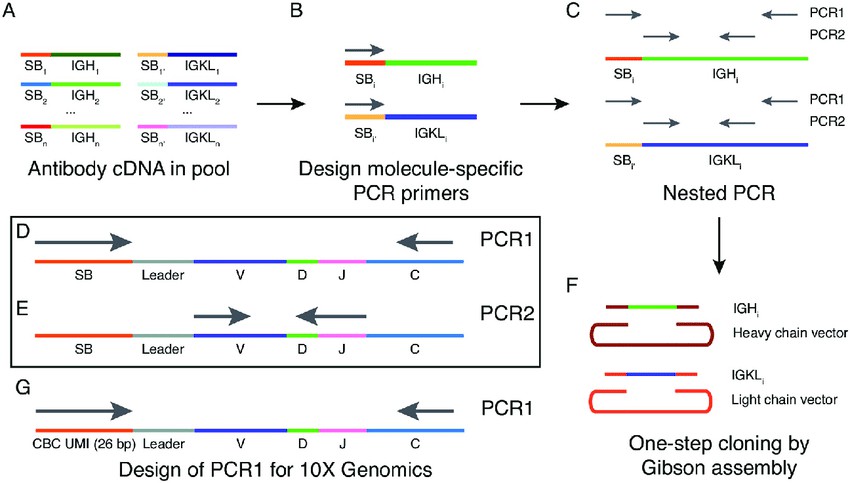 Fig. 1.
Schematic of workflow for Selective PCR for Antibody Retrieval (SPAR). (Horns F, et al., 2020)
Fig. 1.
Schematic of workflow for Selective PCR for Antibody Retrieval (SPAR). (Horns F, et al., 2020)
Creative Proteomics has developed a PCR based antibody sequencing workflow combined with degenerate primer design, which extracts and sequences the antibody genes from hybridoma cells to obtain the corresponding nucleotide sequences, ensuring the stable expression and production of exogenous antibodies in the later stage. In this method, professional degenerate primer design (degenerate primer) and sequencing solutions were applied to provide customers with fast and accurate antibody variable region and full-length gene sequencing services. We provide a one-stop service for the entire analytical steps of antibody sequencing. At the same time, we can also provide corresponding customized services according to the specific needs of customers.
At the Creative Proteomics, we customize a complete set of antibody sequencing service processes for your research.
Ⅰ. Extraction of total RNA from hybridoma cells.
Ⅱ. Reverse transcription of total RNA to cDNA.
Ⅲ. Design degenerate primers for the variable region sequence of the monoclonal antibody, and amplify the light chain and heavy chain region DNA fragments of the variable region of the monoclonal antibody.
Ⅳ. Clone the DNA fragment into a plasmid vector dedicated to sequencing.
Ⅴ. Sequencing analysis of the DNA cloned antibody to obtain the DNA report of the monoclonal antibody.
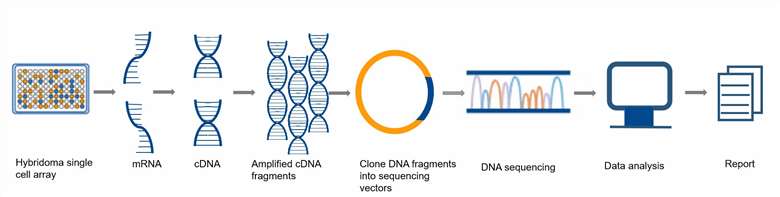 Fig. 2. Workflow of the
monoclonal antibody sequencing service.
Fig. 2. Workflow of the
monoclonal antibody sequencing service.
Creative Proteomics provides fast, reliable, and high-quality sequencing services for customers around the world to advance your discoveries with a team of experts who can help you determine the best path forward. Whether your ultimate goal is simple sequence or recombinantly expressed purified antibody, we have a solution for you. Please contact us today to find out how we can advance your next drug discovery project.
References
For research use only, not intended for any clinical use.