Biopharmaceutical Glycan Analysis Service
- Home
- Applications
- Biopharmaceutical Characterization Services
- Biopharmaceutical Variation Analysis Services
- Biopharmaceutical Glycan Analysis Service
Service Details
The biopharmaceutical industry harnesses the power of multiple gene expression systems to produce therapeutic proteins for personalized treatment of diseases ranging from cancer to autoimmune diseases. These therapeutic proteins, including monoclonal antibodies (mAbs), fusion proteins, hormones, cytokines, and enzymes, are highly specific. Therapeutic glycoproteins, which make up a large proportion of biotherapeutic products, are post-translationally modified by glycan linkages at specific sites in the protein, typically nitrogen atoms on the side chain of asparagine (N linkage) or serine or threonine oxygen atom on the amino acid side chain (O linkage). With this modification, glycans (carbohydrate moieties) are attached to specific sites on the protein, in turn , these glycans are key determinants of the pharmacokinetics, solubility, stability, and immunogenicity of these therapeutic glycoproteins. Therefore, biopharmaceutical manufacturers must characterize and analyze glycans for safety and consistency, which must rely on techniques including high-performance liquid chromatography (HPLC), mass spectrometry (MS), and fluorescent labeling.
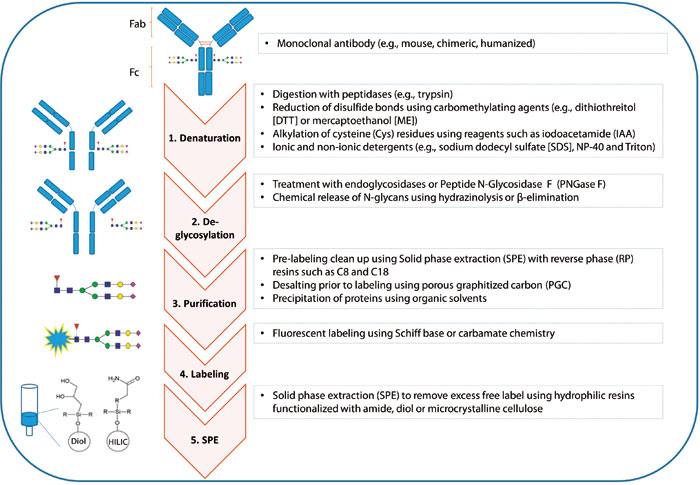 Fig.
1. Schematic illustrating the steps of a typical sample processing method for N-glycan analyses of monoclonal. (Duke
R M, et al., 2015)
Fig.
1. Schematic illustrating the steps of a typical sample processing method for N-glycan analyses of monoclonal. (Duke
R M, et al., 2015)
Glycan characterization is important in the protein drug development process, from early drug candidate selection to late regulatory submission. Glycan variants of glycoprotein products must be adequately analyzed and controlled to ensure product quality. With many years of experience in the field of biopharmaceutical glycan analysis, Creative Proteomics can design and execute a series of analyzes to suit your needs, including the analysis of the total number of glycans on the glycoprotein, the glycan structure of each protein glycosylation site in the molecule.
As a competent partner for biopharmaceutical analysis, we are pleased to provide you with high-quality biopharmaceutical glycan characterization services, including N-glycan analysis and O-glycan analysis. Our glycan analysis service can be performed at the level of intact glycoproteins, but also at the level of proteolytic glycopeptides, released glycans and monosaccharides.
Our intact glycoprotein analysis provides you with direct analysis of general glycan patterns and glycan heterogeneity in intact glycoproteins.
Characterization of protein glycosylation sites and occupancy is achieved at the glycopeptide level by using ESI-MS or MALDI-MS.
Released glycans are rapidly analyzed by MALDI-TOF MS after permethylation.
Quantification of released monosaccharides gives you information on the content of specific terminal monosaccharides, such as sialic acid, mannose-6-phosphate (M6P) or O-linked monosaccharides.
The launch of new products and biosimilars requires detailed characterization of product glycosylation information. Depending on regulatory agency requirements, you can use biopharmaceutical glycan analysis in a variety of areas including, but not limited to.
Creative Proteomics is a reliable biopharmaceutical partner. Our professional team can offer a range of glycan analysis services to our clients worldwile. Our services guarantee accurate and reliable results, at quick turnaround time! If you would like more information about specific aspects of our services, feel free to contact us and we will be happy to answer any questions.
References
For research use only, not intended for any clinical use.