From Static Snapshots to Dynamic Neuropeptide Flux
To truly understand complex brain functions, measuring the total abundance of peptides in brain tissue is insufficient. Conventional tissue homogenates act as static snapshots—they conflate intracellular precursor stores with actively released peptides and inherently fail to capture the critical dimension of time. While approaches like spatial neuropeptidomics are excellent for mapping regional distribution, they cannot record the continuous neurochemical dialogue of a living animal.
Neuropeptides operate via volume transmission, acting as broad neuromodulators that dictate behavioral states, stress responses, and metabolic shifts over minutes to hours. To decode this signaling layer, you must monitor the extracellular fluid in vivo. Our in vivo neuropeptide microdialysis service shifts your research from static abundance to dynamic flux. By continuously sampling the interstitial fluid of freely moving animals, we empower you to track the temporal kinetics of peptide release, linking molecular events directly to observable phenotypic and pharmacological outcomes.
When to Use Time-Resolved In Vivo Neuropeptide Microdialysis
This platform is specifically designed as a dynamic functional readout. It is the necessary methodological choice for your project when:
- Extracellular Release Dynamics are Required: When you need to measure what is actively secreted and available for receptor binding in the interstitial space, rather than just measuring total peptide stores locked inside vesicles.
- Behavior or Drug Responses Must Be Linked to Time: When you need to establish a direct temporal relationship between a specific event (e.g., drug administration, acute stress, feeding) and minute-level peptide flux.
- Classical Neurotransmitter Monitoring is Insufficient: When monoamines (DA, 5-HT) fail to explain prolonged neuromodulation or complex, long-lasting behavioral states.
- Rigorous Dynamic Evidence is Demanded: When high-tier journal reviewers or grant committees require direct, in vivo evidence of endogenous peptide release to substantiate a proposed mechanistic pathway.
Decision Matrix: Microdialysis vs. Tissue Peptidomics
To help you select the most scientifically appropriate assay, we contrast the distinct goals of dialysate analysis versus standard tissue homogenate profiling:
| Research Goal | Microdialysis Neuropeptidomics | Tissue Neuropeptidomics |
|---|---|---|
| Monitor extracellular release over time | Yes | No |
| Link peptide signals to specific behavior/drug timing | Yes | Limited |
| Capture total tissue peptide content (incl. precursors) | Limited | Yes |
| Resolve minute-level kinetic changes in a single animal | Yes | No |
(For comprehensive baseline profiling, explore our untargeted neuropeptidomics service utilizing brain tissue.)
Overcoming the Microdialysis Dilution Barrier
The most significant barrier to dialysate peptidomics is sample dilution. Neuroactive peptides exist in the extracellular space at sub-picomolar to femtomolar concentrations. When collected via microdialysis, these signals are further diluted and often lost entirely on standard LC-MS platforms or degraded by adsorption to collection tubing.
We have engineered a specialized analytical pipeline to conquer this dilution barrier. By utilizing state-of-the-art timsTOF Pro / HT and Orbitrap Exploris / Eclipse mass spectrometers coupled with specialized nano-flow liquid chromatography (Nano-LC), we achieve attomole-level sensitivity. This ensures that even the lowest abundance peptides are accurately quantified against challenging matrix suppression effects.
How We Ensure Confidence in Dialysate Peptide Measurements
Detecting low-concentration, highly dynamic peptides from minute volumes of biofluid requires rigorous analytical control. We secure the credibility of your temporal data through six mandatory quality checkpoints:
Core Applications: PK/PD, Behavioral Neuroscience, and Circuit Dynamics
Our dynamic neuropeptide flux analysis platform serves as an essential tool for high-level systems neuroscience and pharmaceutical development:
- CNS Drug PK/PD Correlation: For DMPK leaders, we directly monitor the extracellular brain microenvironment. Map how a systemic pharmacological intervention crosses the blood-brain barrier and triggers secondary endogenous peptide release over time.
- Behavior-Synchronized Neuroscience: Correlate specific behavioral events—such as acute stress, social interaction, or feeding—with immediate, minute-level surges in neuropeptide release in targeted circuits.
- Circuit-Level Metabolic Dynamics: Track the slow, rhythmic fluctuations of metabolic signaling peptides (e.g., orexins, NPY) in the hypothalamus over extended temporal windows without sacrificing the animal.
Workflow for Time-Resolved In Vivo Neuropeptide Microdialysis
We operate on a consultative "Hybrid Model." While your team performs the stereotaxic surgery and in vivo microdialysis using established behavioral paradigms, we provide rigorous protocols for sample preservation before you send the dialysate to our facilities.
Microdialysis Peptidomics vs. Classical Neurotransmitter Monitoring
Traditional microdialysis services are optimized exclusively for classical, small-molecule neurotransmitters using basic electrochemical detection. They are structurally incapable of resolving the peptidergic neuromodulation layer.
| Analytical Dimension | Microdialysis Neuropeptidomics (Our Service) | Traditional MD (e.g., Dopamine, Serotonin) |
|---|---|---|
| Target Molecules | Large endogenous peptides, complex prohormone cleavage products | Small-molecule monoamines and amino acids |
| Biological Implication | Neuromodulation, volume transmission, and long-lasting behavioral states | Fast, localized point-to-point synaptic transmission |
| Detection Method | High-Resolution Nano-LC-MS/MS | HPLC with Electrochemical Detection (ECD) |
| Sample Challenge | Extreme MS matrix effects, severe tube adsorption, rapid peptidase degradation | Straightforward electrochemical oxidation |
| Probe Requirement | High MWCO (> 20-100 kDa) mandatory | Low MWCO (e.g., 6 kDa) is generally sufficient |
Demo Results: Visualizing the Brain's Chemical Pulse
We deliver a publication-ready Mechanism Package that translates complex longitudinal mass spectrometry data into intuitive biological kinetics:
Temporal Flux Curves
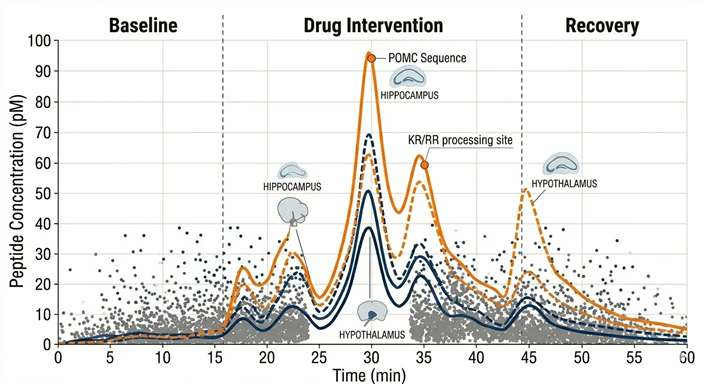
X-axis tracking time (min) across Baseline, Intervention, and Recovery phases. Y-axis tracking the precise concentration spikes of critical peptides as they surge and return to baseline.
Behavior-Peptide Sync Maps
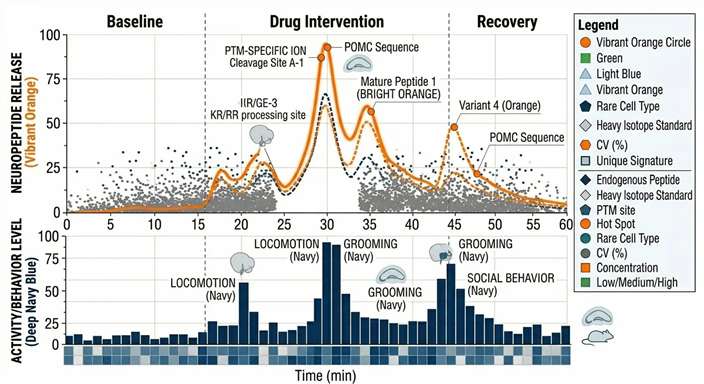
Directly overlaying animal activity tracking data with peptide release pulses, proving the correlation between neurochemistry and phenotype.
Processing Dynamics
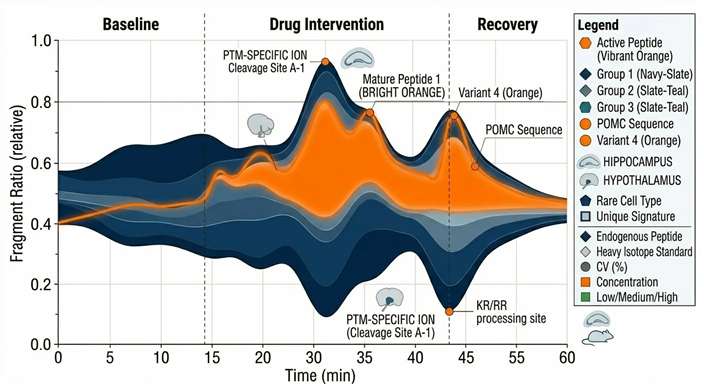
Visualizing the dynamic ratio of precursor proteins to their active, enzymatically cleaved downstream products over the duration of the experiment.
Recovery & LOD Curves
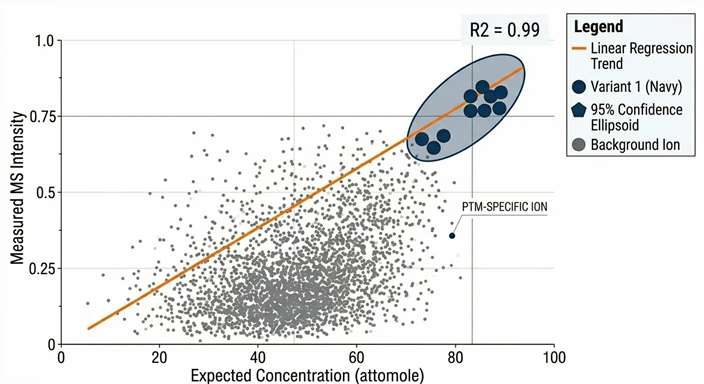
Providing hard analytical proof of our quantitative linearity and attomole detection limits within your specific aCSF matrix.
(To further validate these temporal biomarkers across larger cohorts, these targets are ready for downstream assay design using our targeted neuropeptide quantification services.)
Sample and Probe Requirements for Dialysate Peptidomics
To guarantee analytical success, precise adherence to physical and chemical parameters during your sampling phase is critical:
| Parameter | Recommended Specification | Critical Notes for Peptidomics |
|---|---|---|
| Compatible Models | Mice, Rats, Non-Human Primates (NHP) | Probe size and flow rate must be optimized according to the specific brain region and animal model. |
| Probe MWCO | > 20 kDa (e.g., 38 kDa or 100 kDa) | Standard 6 kDa probes will filter out most neuropeptides. High MWCO is mandatory. |
| Perfusion Fluid | Artificial CSF (aCSF), unbuffered | Avoid adding carrier proteins (like BSA) unless completely necessary, as they cause massive MS ion suppression. |
| Flow Rate | 0.5 – 2.0 µL/min | Slower flow rates increase relative recovery but reduce temporal resolution. |
| Temporal Bins | 10 – 20 min intervals | Yields 5–40 µL per fraction. Optimize based on physiological time window. |
Disclaimer: All services and platforms described are for Research Use Only (RUO). Not for use in diagnostic procedures.
Dynamic In Vivo Detection of Enkephalins Following Acute Stress
Journal: eLife
Published: 2025
Background & Challenge
Endogenous opioid peptides, such as enkephalins, play critical roles in pain modulation, reward circuitry, and stress responses. A neuroscience research team needed to map the real-time extracellular release of these specific peptides in the mouse brain to establish a direct temporal link between an acute stress event and neurochemical adaptation.
Enkephalins are secreted in extremely low quantities (sub-picomolar) and have an extraordinarily short half-life in the extracellular fluid due to rapid cleavage by endogenous peptidases. Traditional microdialysis coupled with standard detection methods repeatedly failed to capture the signal above the noise floor, missing the transient release window entirely.
Analytical Strategy (Methods)
To overcome the dilution and degradation barriers, an optimized in vivo microdialysis pipeline was deployed. Researchers utilized high-MWCO probes perfused with an artificial cerebrospinal fluid (aCSF) containing a customized peptidase inhibitor cocktail. Dialysate fractions were collected at strict 10-minute intervals to maintain high temporal resolution and were immediately flash-frozen. The extracellular fluid was then analyzed using an ultra-high-sensitivity Nano-LC-MS/MS platform, leveraging advanced retention-time scheduling to pinpoint low-abundance targets.
Outcomes & Results
- Attomole Sensitivity: The analytical pipeline achieved an unprecedented limit of detection in the low-attomole range, successfully isolating the native peptide signals from the high-salt microdialysate matrix.
- Behavior-Synchronized Flux: As clearly demonstrated in Figure 3 (Panels F and G) of the referenced study, the generated Temporal Flux Curves captured a distinct, transient "pulse-like" spike in enkephalin concentrations. This surge perfectly synchronized with the exact onset of the acute stressor (predator odor), followed by a rapid return to baseline within the subsequent window.
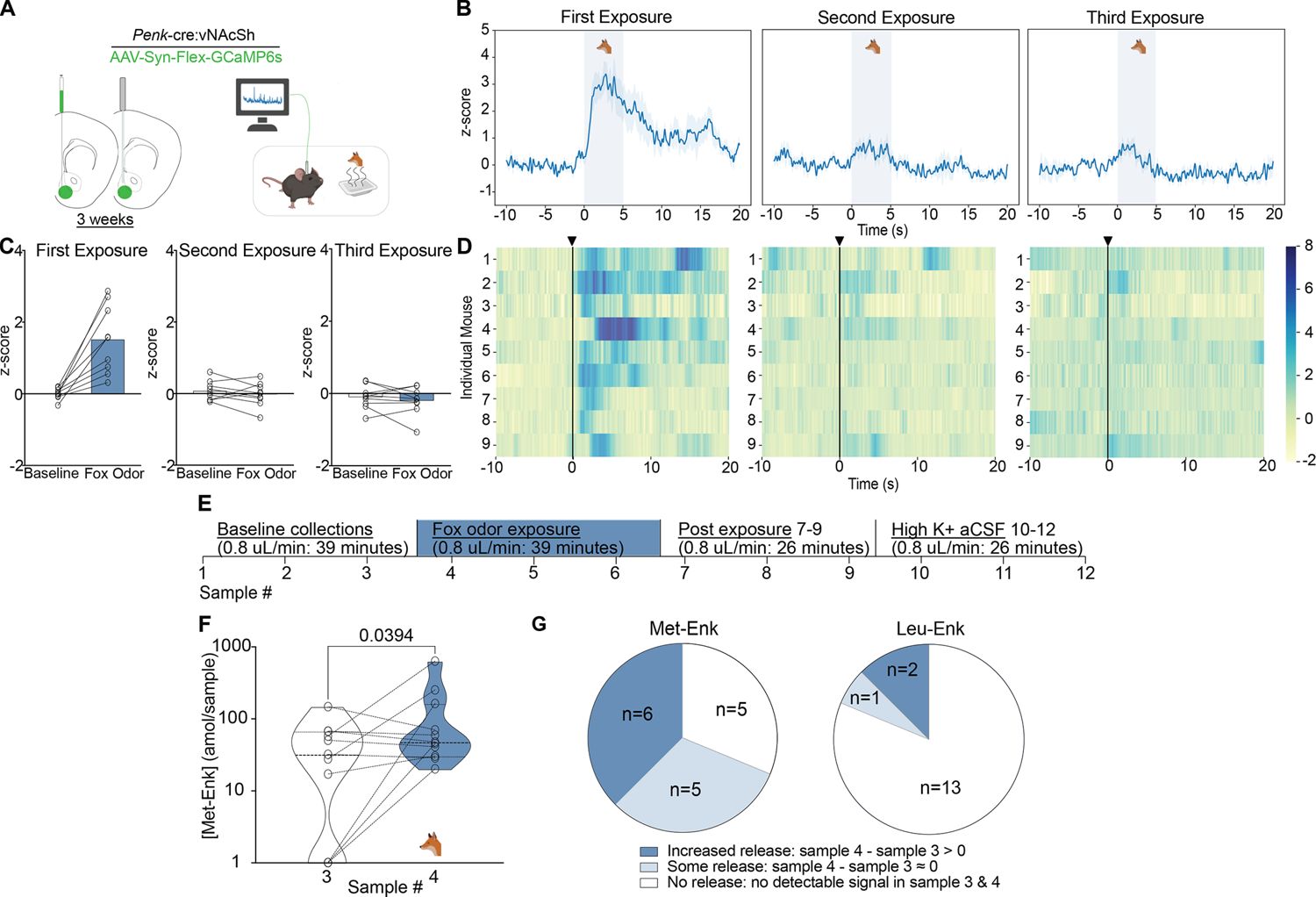
Scientific Impact: This case firmly validates that pairing meticulous, degradation-aware microdialysis collection with advanced nano-LC-MS/MS can overcome extreme dilution barriers. It provides researchers with the definitive capability to reveal the minute-by-minute kinetic release of endogenous neuropeptides during complex behavioral and physiological events.
Reference
- Highly sensitive in vivo detection of dynamic changes in enkephalins following acute stress. eLife (2025). https://elifesciences.org/reviewed-preprints/91609v2


