Mapping Metabolic Crosstalk: The Targeted LC-MS/MS Advantage
Energy homeostasis, appetite regulation, and glucose metabolism are orchestrated by a dynamic, multi-organ neuroendocrine network. When evaluating the efficacy of highly potent metabolic therapeutics—such as unimolecular dual/tri-agonists (GLP-1/GIP/Glucagon) or novel anti-obesity Small Activating Peptides—measuring a single "total" peptide level provides an incomplete and often misleading biological picture.
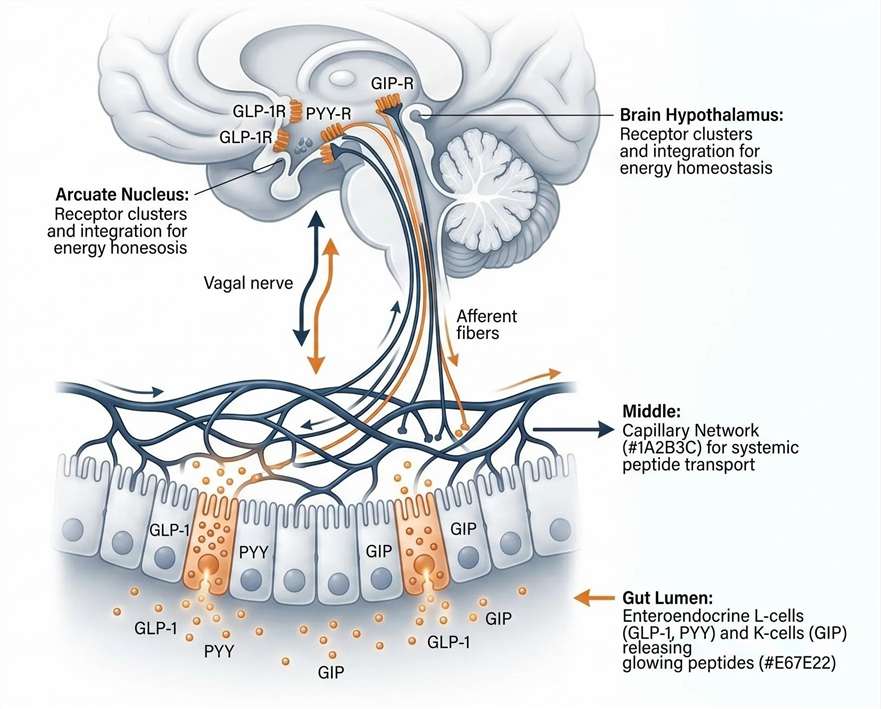
Our Gut-Brain Axis Hormone Panel employs state-of-the-art targeted liquid chromatography-tandem mass spectrometry (LC-MS/MS) via Parallel Reaction Monitoring (PRM) or Multiple Reaction Monitoring (MRM). This platform allows us to capture the complete systemic endocrine landscape with unparalleled specificity. Unlike traditional immunoassays, our mass spectrometry approach enables the precise tracking of functional peptide isoforms from local intestinal secretion (L-cells and K-cells), through systemic circulation, and across the blood-brain barrier to the metabolic control centers in the hypothalamus.
Comprehensive Pharmacological Applications for Gut Hormone Panels
Our multiplex panel is engineered to support the most rigorous experimental designs in the translational drug development pipeline, bridging the gap between discovery and IND-enabling studies:
Key Analytes in Our Multiplex Incretin and Appetite Panel
To support systems-level metabolic research, we avoid "total" hormone measurements that mask physiological reality. Endogenous peptides undergo extensive N-terminal and C-terminal processing, which drastically alters their receptor binding affinity and biological half-life. Our targeted LC-MS/MS hormone panel provides absolute quantification for the functional fraction.
| Endocrine Axis & Function | Targeted Analytes & Isoform Specificity | Biological Relevance & Mechanism |
|---|---|---|
| Incretins & Glycemic Control | GLP-1 (Active 7-36 amide vs Inactive 9-36) GIP (Active 1-42 vs Inactive 3-42) Glucagon, Oxyntomodulin (OXM), Glicentin |
Direct regulation of postprandial insulin secretion, gastric emptying, and hepatic glucose output. |
| Anorexigenic (Satiety / Fullness) | PYY (Total vs Active 3-36) CCK (CCK-8, CCK-33) Amylin, Leptin, Pancreatic Polypeptide (PP) |
Induction of meal termination via vagal afferent activation and direct hindbrain receptor targeting. |
| Orexigenic (Hunger / Adiposity) | Ghrelin (Acylated/Active vs Unacylated/Des-acyl) AgRP, NPY |
Fasting-induced hunger signaling, fat storage promotion, and central growth hormone regulation. |
| Gut Motility & Secretion | VIP, Neurotensin, Somatostatin, Motilin, Secretin | Regulation of peristaltic rhythm, gastric acid secretion, and paracrine inhibition of neighboring cells. |
Active vs. Inactive GLP-1 Quantification: Overcoming the Isoform Blindspot
The most significant analytical hurdle in incretin profiling is the rapid enzymatic degradation of peptide hormones. Active GLP-1 (7-36 amide) and GIP (1-42) are cleaved by Dipeptidyl Peptidase-4 (DPP-4) within 1-2 minutes of entering circulation. Furthermore, the biologically active form of Ghrelin depends on an octanoyl group on its third serine residue, which is highly susceptible to hydrolysis by circulating esterases.
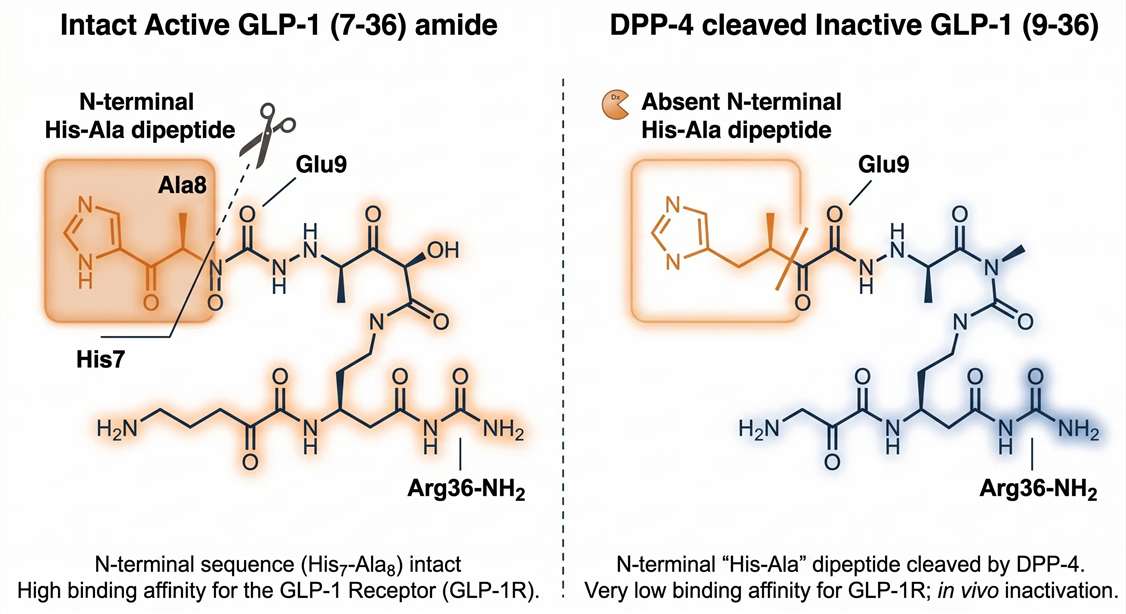
Traditional immunoassays fundamentally struggle with this active vs inactive GLP-1 quantification. Antibodies designed for "active" GLP-1 frequently cross-react with the inactive (9-36) metabolite because they cannot reliably differentiate the loss of just two N-terminal amino acids (His-Ala). This cross-reactivity leads to artificially inflated active hormone readings and compromised PK/PD models.
By utilizing targeted neuropeptide quantification (PRM/MRM), we quantify hormones based on their exact mass-to-charge (m/z) ratio and unique amino acid fragmentation patterns.
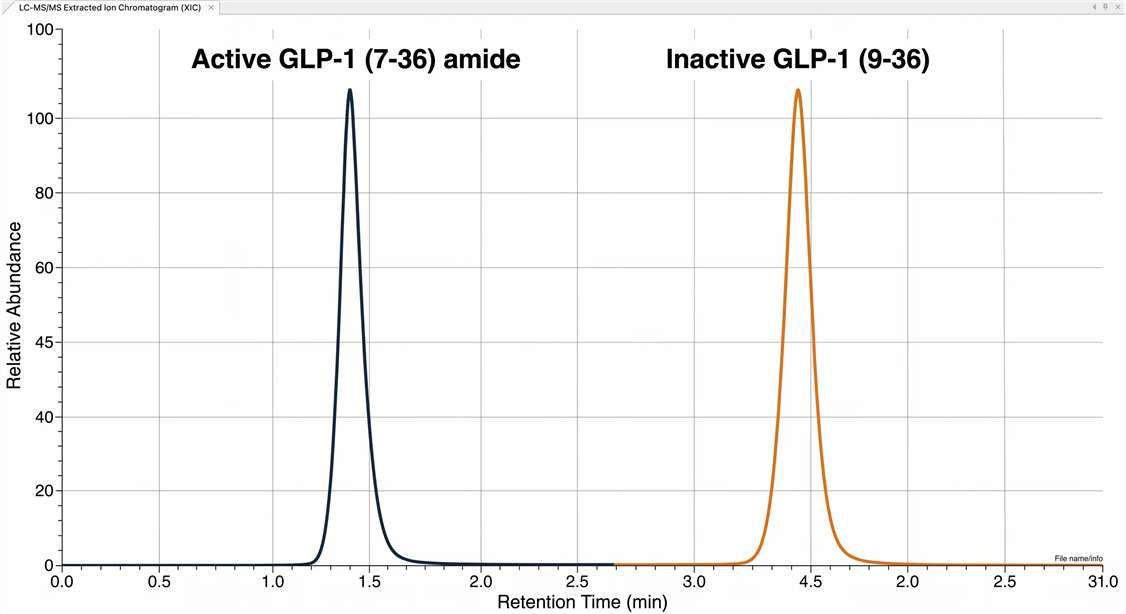
As shown in our high-resolution chromatograms, mass spectrometry provides unambiguous baseline separation of these isoforms. This mass-based resolution ensures that you receive the absolute quantification of the biologically active fraction, providing a definitive molecular readout for your drug efficacy studies.
Seamless Cross-Species Metabolic Hormone Profiling: Mouse to NHP
A major bottleneck in preclinical drug development is the limited availability of high-quality commercial immunoassay kits for non-standard animal models. Because LC-MS/MS quantifies hormones based on their conserved amino acid sequences rather than antibody-epitope binding, we can seamlessly translate your assay across different species.
Whether your study involves Rodent models (Mouse/Rat), Canine, Minipig, or Non-Human Primate (NHP) cohorts, our platform maintains analytical consistency. This allows for direct comparisons of hormonal responses from the early pharmacology stage through to human clinical trials without the confounding variables of differing antibody lots or cross-species cross-reactivity.
Bioanalytical Workflow and Pre-Analytical Stabilization Protocols
In metabolic peptidomics, the sample collection phase is the most critical quality gate. Even the most sensitive mass spectrometer cannot rescue data from degraded samples. Our end-to-end workflow is built around the strict preservation of endogenous signaling states:
Sample Requirements for Targeted Hormone Panel Analysis
Our multiplex gut hormone panel is specifically optimized for ultra-low volume inputs, enabling serial sampling in longitudinal rodent studies or the analysis of precious clinical CSF.
| Matrix Type | Minimum Volume / Mass | Critical Pre-Analytical Stabilization Protocols | Typical Study Design Context |
|---|---|---|---|
| Mouse / Rat Plasma | 50 - 100 μL | Add DPP-4 inhibitor & Protease Inhibitor Cocktail (e.g., Aprotinin) within seconds of collection. Acidification required for Ghrelin. | Acute metabolic response; post-dose PK/PD; Fasting vs. Refeeding. |
| Human / NHP Plasma | 200 - 500 μL | Collect in pre-loaded P800/EDTA tubes. Keep strictly on ice and centrifuge in a refrigerated rotor. | Oral Glucose Tolerance Tests (OGTT); Mixed Meal Tolerance Tests (MMTT). |
| Cerebrospinal Fluid (CSF) | 20 - 50 μL | Snap-freeze immediately. Avoid blood contamination during collection. | Validating CNS penetrance of peripherally administered peptide drugs. |
| Tissue (Gut/Brain) | 20 - 50 mg | Snap-freeze in liquid N2 immediately. Do not wash in PBS to prevent protease activation. | Chronic treatment endpoint analysis; evaluation of local hormone reservoirs. |
Typical Results and Delivering High-Impact PD Data
Our bioanalytical reports are designed to be "submission-ready," providing the quantitative depth needed for regulatory filings and high-impact publications. We provide integrated data visualization that correlates systemic endocrine shifts with Real-Time In Vivo Microdialysis & Temporal Fluxomics.
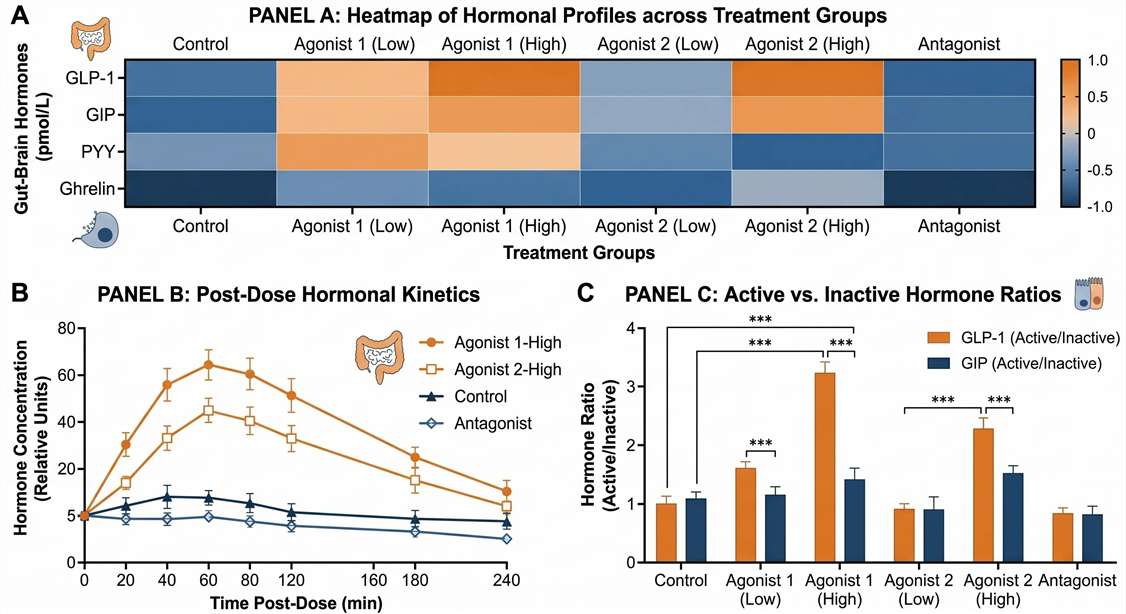
- Absolute Concentration Data: Accurate quantification (pg/mL or pM) calibrated against multi-point SIL-IS standard curves.
- Time-Course Endocrine Kinetics: Kinetic line graphs mapping the acute spikes of active GLP-1 and PYY 3-36 following meal challenge or drug dosing.
- Isoform Ratio Statistics (Active vs. Cleaved): Statistical bar charts demonstrating the percentage of biologically active hormone vs. degraded metabolites across different treatment groups.
- Integrated Multi-Omics Mapping: Correlation matrices linking the hormone panel readouts with microbiome features (16S/Metagenomics) or small molecule metabolites.
- Methodology & QC Report: Comprehensive documentation of LLOQ, recovery, linearity, and intra-batch precision (TAT: typically 4-6 weeks).
Evaluating Your Options: LC-MS/MS vs. ELISA for Incretin Assays
Selecting the right analytical platform is a strategic decision for your pipeline. While immunoassays (ELISA/Luminex) remain useful for rapid, routine screening where total hormone levels are sufficient, our targeted LC-MS/MS panels are the definitive choice for IND-enabling studies.
| Capability Metric | Targeted LC-MS/MS Hormone Panel | Conventional Immunoassays (ELISA/Luminex) |
|---|---|---|
| Isoform Specificity (Active vs Total) | Absolute Specificity: Distinguishes active GLP-1 (7-36) from inactive fragments based on exact mass. | Prone to Cross-Reactivity: Antibodies often cannot resolve small N-terminal truncations. |
| Multiplex Capacity | High: Quantifies 10-20+ hormones simultaneously without antibody cross-talk interference. | Moderate: Limited by antibody-analyte specificity in multiplex wells. |
| Species Flexibility | Universal: Can be applied to NHP, Canine, and Minipig without species-specific kit development. | Restricted: Highly species-dependent; requires specific validated kits for each model. |
| Drug-Hormone Differentiation | Definitive: Can separate synthetic drug analogs from endogenous host hormones by mass. | Difficult: Synthetic analogs often bind to the same capture antibodies as endogenous peptides. |
Disclaimer: All services and analytical platforms described are intended for Research Use Only (RUO). Not for use in diagnostic procedures.


