Why Reproductive Endocrine Readouts Often Mislead
Single-analyte reproductive hormone measurements often mislead translational studies. Steroid hormones share highly similar core structures, which increases the risk of cross-reactivity or misclassification in less specific assays. Furthermore, critical markers in early-stage puberty or menopausal models circulate at sub-picogram levels, falling far below the reliable detection limits of conventional clinical assays.
| Analytical Problem | Why It Happens | Impact on R&D Decisions |
|---|---|---|
| Structural Similarity and Isomer Interference | Steroid isomers (e.g., testosterone vs. epitestosterone, estrone vs. estradiol) differ only by minor hydroxyl group orientations. | ELISA cross-reactivity causes false-positive signals, severely misguiding mechanism-of-action and toxicity interpretations. |
| Sub-Physiological Abundance | Estrogens and minor androgens in pediatric, menopausal, or rodent models circulate at extreme low pg/mL levels. | Assays with poor Lower Limits of Quantification (LLOQ) return noisy data, masking true baseline shifts. |
| Fragmented Pathway Data | Standard diagnostic assays measure isolated hormones rather than capturing the complete precursor-to-product steroidogenic flux. | Makes it impossible to accurately pinpoint in vivo enzymatic blocks (e.g., aromatase inhibition) or target-organ feedback loops. |
How This Panel Supports Fertility, HPG Feedback, and Toxicology Decisions
To make confident go/no-go decisions in drug discovery, researchers must evaluate the dynamic balance of the Hypothalamic-Pituitary-Gonadal (HPG) axis comprehensively. Our panel translates absolute quantification into actionable mechanistic insights, supporting common translational project types:
- Ovarian stimulation response studies in IVF/ART therapeutic programs.
- PCOS hyperandrogenism metabolic profiling and sub-phenotyping.
- Endocrine disruptor (EDC) screening in rodent reproductive toxicity models.
- Menopausal or pediatric low-estrogen drug efficacy studies.
- Follicular fluid or seminal plasma endocrine microenvironment characterization.
What This Panel Helps You Decide:
Who This Panel Is Best For
This solution is meticulously engineered to support the rigorous bioanalytical demands of:
- IVF / ART Researchers: Developing novel ovarian stimulation protocols, recombinant gonadotropins, or luteal phase support therapies.
- Endocrinologists (PCOS/Menopause): Investigating hyperandrogenism, metabolic syndrome crossovers, and hypogonadism mechanisms.
- Reproductive Toxicologists: Screening industrial chemicals, cosmetics, or new molecular entities (NMEs) for endocrine-disrupting properties and reproductive safety.
- Translational Scientists: Working with volume-limited rodent models, pediatric cohorts, or rare clinical matrices where sample sparing is paramount.
Panel Coverage: Gonadotropins, Steroids, and Reproductive Biomarkers
We provide a fit-for-purpose, parallel reproductive endocrine assessment across peptide/protein and steroid markers, configured to your specific sample matrix and assay sensitivity needs.
| Functional Axis | Selected Target Analytes | Analytical Advantage & Physiological Relevance |
|---|---|---|
| Steroidogenesis Full Spectrum | Estradiol (E2), Estrone (E1), Estriol (E3), Testosterone, DHT, Progesterone, DHEA, Androstenedione | Core effectors of sexual development. LC-MS/MS enables high-specificity separation of key structural isomers and closely related steroid analytes to prevent false pathway interpretation. |
| Ovarian/Testicular Reserve | Anti-Müllerian Hormone (AMH), Inhibin A, Inhibin B, SHBG | Direct indicators of the follicular pool and circulating hormone bioavailability. Multiplexed to conserve limited sample volumes. |
| Gonadotropins & Prolactin | Follicle-Stimulating Hormone (FSH), Luteinizing Hormone (LH), hCG, Prolactin | Pituitary drivers of the HPG cascade; critical for assessing central feedback inhibition versus peripheral receptor block. |
Note: When your project requires mapping upstream hypothalamic control, this panel can be seamlessly coordinated with targeted mass spectrometry quantitation of neuropeptidomics workflows to track GnRH or Kisspeptin signaling. Final analyte combinations are configured based on matrix type, sample volume, and the required LLOQ for each marker class.
Technical Superiority: Overcoming Isomers and Low-Abundance Limits
We secure bioanalytical confidence through two foundational technical pillars: absolute structural specificity and derivatization-enhanced sensitivity.
A. Isomer Baseline Separation

Targeted LC-MS/MS distinctly separates highly homologous steroids based on exact mass and specific chromatographic retention times. By utilizing optimized gradients on sub-2 μm particle columns, we achieve baseline separation of isobaric pairs (e.g., testosterone vs. epitestosterone), substantially reducing cross-reactivity concerns and improving analyte discrimination.
B. Sub-pg/mL Sensitivity for Micro-Volumes
Estrogens are notoriously difficult to ionize in standard mass spectrometry. Utilizing state-of-the-art triple quadrupole (QQQ) systems paired with specialized chemical derivatization techniques (e.g., Dansyl chloride labeling), we dramatically enhance the ionization efficiency of phenolic hydroxyl groups. This achieves sub-pg/mL Lower Limits of Quantification (LLOQ) for estradiol, requiring only 50–100 μL of input volume, preserving precious rodent or pediatric samples.
Bioanalytical Workflow and Pre-Analytical QC Checkpoints
Ensuring reproducibility in complex reproductive matrices requires a strict, multi-stage workflow. Our processes are governed by rigorous Quality Control (QC) checkpoints.
Sample Requirements and Biological Context Considerations
Cycle Stage, Timing, and Biological Context Matter
Unlike steady-state metabolic markers, the HPG axis is highly cyclical. The biological interpretation of your data depends heavily on sampling context. We strongly advise controlling for menstrual phase (e.g., early follicular vs. mid-luteal), puberty stage, and diurnal timing when designing your in vivo protocols to ensure meaningful comparison between cohorts.
The Value of Local Microenvironments
Systemic blood draws do not always reflect target-organ physiology. Analyzing local matrices—such as follicular fluid (for oocyte maturation context) or seminal plasma (for testicular endocrine environments)—provides a far more accurate representation of localized steroidogenesis.
| Matrix Type | Application Value | Min Volume | Cycle/Timing & Handling SOPs |
|---|---|---|---|
| Follicular Fluid | Local microenvironment & oocyte support | 100 μL | Centrifuge to remove cellular debris (granulosa cells). Flash freeze immediately. |
| Seminal Plasma | Local endocrine context & male tox | 50 - 100 μL | Use approved collection devices without endocrine-disrupting plastics. |
| Serum (Low Volume) | Rodent models / Pediatric cohorts | 50 μL | Fasting morning samples preferred. Document exact cycle stage. Store at -80°C. |
| Tissue (Gonadal) | Preclinical toxicology / EDC studies | 30 - 50 mg | Snap-freeze in liquid nitrogen within 1 minute of excision. |
Applications in Reproductive Health and Toxicology
Our analytical workflows are designed to support precise R&D tasks across preclinical and clinical reproductive pharmacology:
Representative Deliverables and Interpretation Outputs
We deliver structured, context-rich data designed to directly answer your mechanistic questions, moving beyond raw concentrations to study-context interpretation.
HPG Feedback Ratio Analysis

Calculate LH/FSH or E2/Testosterone ratios across experimental groups to prove target engagement and feedback loop modulation.
Steroidogenesis Pathway Heatmaps

Visualize upstream/downstream metabolite abundance to pinpoint specific enzyme inhibition (e.g., 5α-reductase or aromatase blocks).
Local vs. Systemic Comparison

Overlay follicular fluid versus circulating serum endocrine profiles to confirm microenvironment drug penetration.
Bioanalytical QC Package

Comprehensive validation metrics including extraction recovery rates in complex matrices (seminal plasma) and LLOQ confirmation.
Evaluating Analytical Platforms: When LC-MS/MS Is Preferred
CLIA and ELISA remain highly useful and cost-effective tools for routine, single-analyte screening in standard human volumes (e.g., basic hospital clinical tracking).
However, LC-MS/MS is strongly preferred when your R&D study demands:
- The absolute separation of structural isomers without antibody cross-reactivity.
- Quantification of ultra-low abundance steroids (e.g., pediatric estradiol) in severely volume-limited samples (rodent models).
- Overcoming severe matrix effects in complex, lipid-rich biofluids like follicular fluid.
- Multiplexed pathway analysis to interpret complete steroidogenic enzyme flux from a single aliquot.
Frequently Asked Questions (FAQ)
Insights From Liquid Chromatography-Mass Spectrometry-Measured Androgens in Women With Polycystic Ovary Syndrome
Journal: Journal of the Endocrine Society
Published: 2024
https://pmc.ncbi.nlm.nih.gov/articles/PMC10904685/Background
Hyperandrogenemia is the defining feature of Polycystic Ovary Syndrome (PCOS). The role of LC-MS/MS over immunoassay in the diagnosis of PCOS has been debated. Standard immunoassays rely on antigen-antibody complexes and are prone to cross-reactivity with structurally similar steroids, frequently lacking the precision required at critical female testosterone thresholds. This diagnostic ambiguity complicates fertility risk stratification and metabolic phenotyping.
Methods
In a prospective cross-sectional study, researchers compared targeted LC-MS/MS androgen profiling against standard electrochemiluminescence immunoassays (ECLIA). The study analyzed comprehensive androgen profiles—including Total Testosterone, Androstenedione (A4), and Dehydroepiandrosterone (DHEA)—in a cohort of women with PCOS phenotypes compared to age- and BMI-matched controls.
Results
Immunoassay Limitations Exposed
- The study revealed significant discrepancies when relying solely on traditional immunoassays to identify biochemical hyperandrogenism.
- When using a clinical cut-off for normal testosterone (≤ 45 ng/dL), standard assay approaches misclassified a large portion of the hyperandrogenic phenotype cohort as "normal".
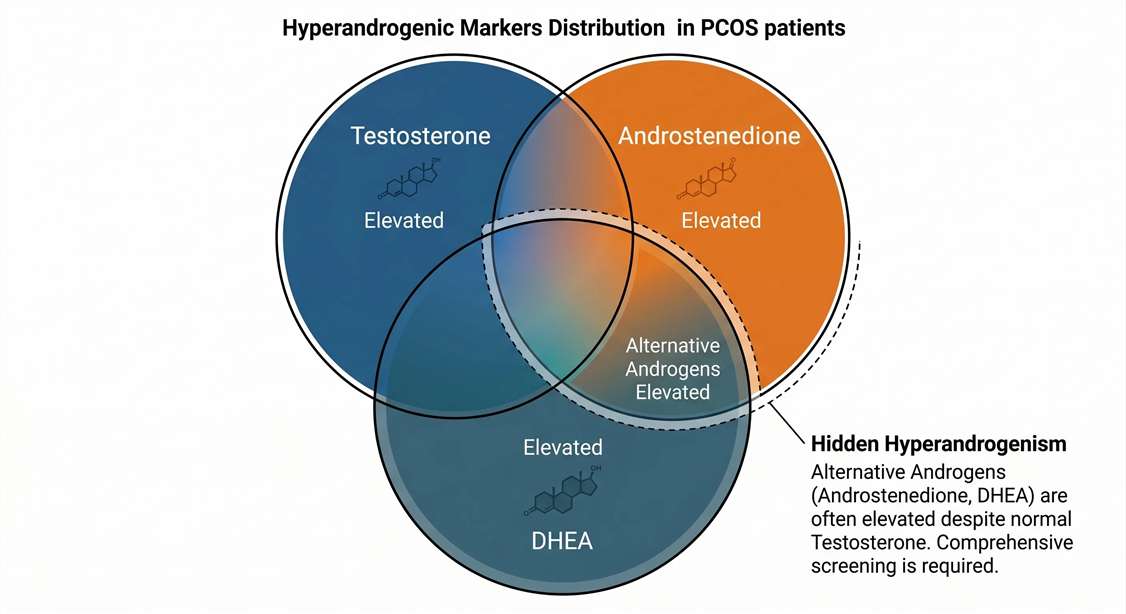
Hidden Hyperandrogenism Identified by LC-MS/MS
- By employing high-specificity LC-MS/MS, researchers identified that 75% (14 out of 19) of the subjects who presented with "normal" testosterone levels actually possessed significantly elevated alternative androgens (specifically Androstenedione or DHEA).
- The targeted mass spectrometry data demonstrated that Androstenedione had the highest Area Under the Curve (AUC = 0.89) for diagnosing PCOS, outperforming Total Testosterone (AUC = 0.81).
- This overlapping pattern of androgen elevation is visually detailed in the study's Venn Diagram (Figure 1 in the original paper), illustrating how LC-MS/MS uncovers hidden hyperandrogenism missed by single-analyte immunoassay screening.
Conclusion
Relying solely on immunoassay-based testosterone measurements results in an incomplete understanding of HPG-axis dysfunction. For fertility drug discovery and reproductive cohort phenotyping, targeted LC-MS/MS provides the methodological rigor required to accurately quantify the entire steroidogenic cascade. This comprehensive profiling refines the diagnosis of biochemical hyperandrogenism and helps properly stratify patient sub-groups for adverse metabolic and fertility outcomes.


