Why In Vivo Microdialysis Matters for Local Tissue PK/PD and Neurochemical Dynamics
Traditional pharmacokinetic (PK) and neurochemical studies heavily rely on sequential blood sampling and whole-tissue homogenates. However, these conventional methods present a critical blind spot in translational drug development: they fail to distinguish between the intracellular compartment, where a drug may be sequestered, and the extracellular interstitial fluid (ISF), where the drug actually interacts with its target receptors. Furthermore, tissue homogenization completely destroys the dynamic, temporal context of biochemical events.
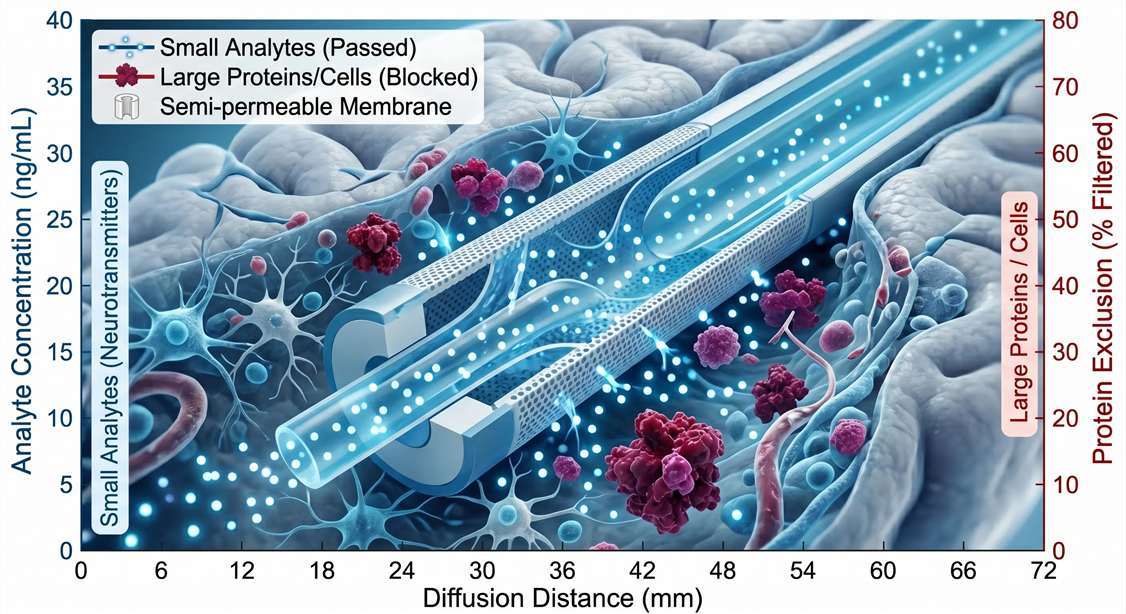
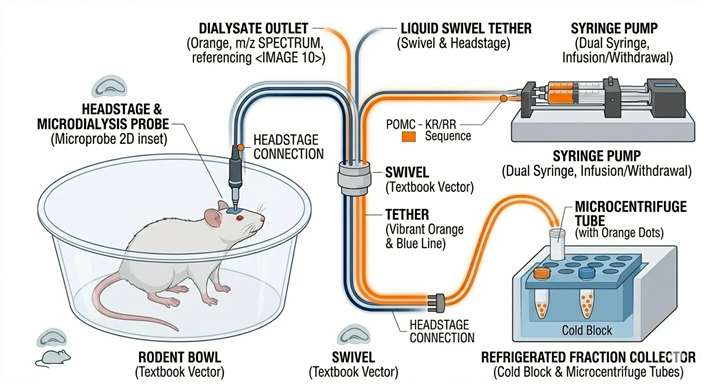
In contrast, in vivo microdialysis is one of the few techniques that enables continuous, local, extracellular sampling of free analytes in vivo with high temporal resolution. By implanting a semi-permeable membrane probe directly into a target brain region or peripheral tissue, researchers can continuously collect unbound, pharmacologically active molecules. This allows for the precise, minute-by-minute tracking of a drug's arrival at the target site and the immediate neurochemical or metabolic changes it triggers, entirely without sacrificing the animal at multiple time points.
Establishing Tissue-Level PK/PD Relationships
For CNS and solid tumor indications, systemic exposure does not guarantee local efficacy. A potent candidate may exhibit excellent plasma stability but fail to engage its target due to poor tissue penetration or rapid local clearance.
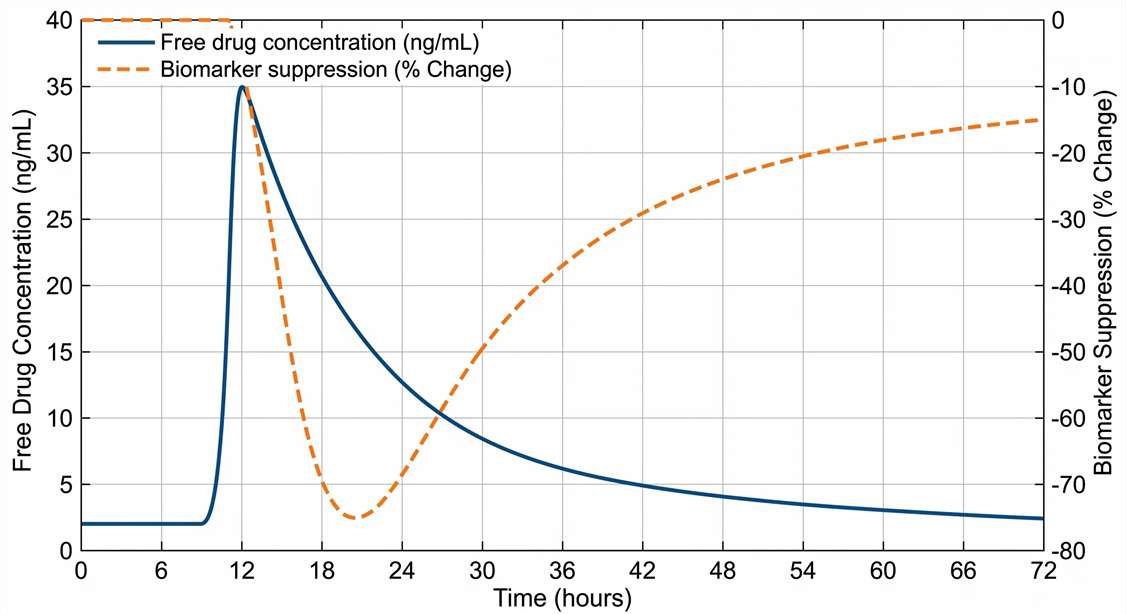
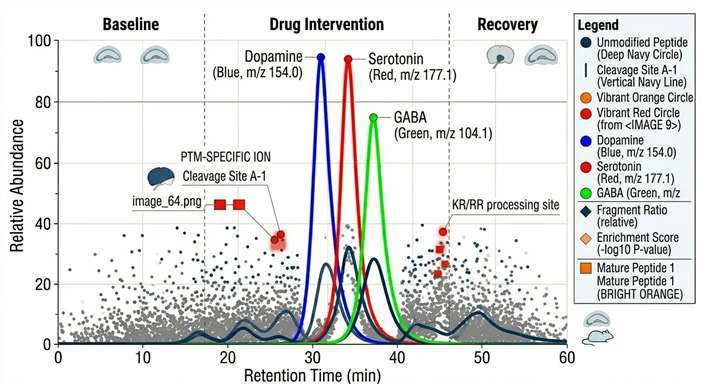
Our continuous sampling microdialysis service directly correlates local exposure with pharmacological effect. By simultaneously monitoring plasma PK and local ISF free drug concentrations, we help you construct accurate tissue-specific PK/PD models. When coupled with downstream analysis of biomarkers or neurotransmitters from the same dialysate, this approach validates target engagement. You can directly observe the physiological response—such as a transient spike in dopamine or a drop in local glucose—precisely synchronized with the local extracellular drug concentration.
Recommended Research Applications
Our microdialysis platform is engineered to support advanced preclinical evaluations and mechanistic discovery. This service is highly recommended for the following research applications:
(Note: While our primary focus is on endogenous metabolites and drug kinetics, advanced stable isotope tracing is available as an optional fluxomics module for specialized metabolic pathway investigations).
Integrated In Vivo Sampling Across Brain ISF, Peripheral Tissues, and Tumor Microenvironments
Microdialysis is highly versatile and can be adapted to almost any solid tissue that accommodates probe implantation. Our surgical and analytical teams specialize in integrating sampling platforms across diverse biological compartments:
- Brain Interstitial Fluid (ISF): Stereotaxic implantation targeting specific cerebral nuclei (e.g., striatum, prefrontal cortex, hippocampus) to monitor localized neurochemical circuitry.
- Peripheral Tissues: Implantation of flexible linear or concentric probes into skeletal muscle, subcutaneous adipose tissue, or hepatic compartments to track systemic metabolic conditions or peripheral drug delivery.
- Tumor Microenvironment (TME): Sampling intratumoral fluid to assess local drug penetration, hypoxic metabolic signatures (e.g., lactate accumulation), or immune-modulating metabolites.
- Dual-Probe & Blood Synchronized Sampling: Simultaneous implantation in multiple regions (e.g., bilateral brain regions, or brain ISF combined with continuous blood sampling) to map systemic vs. localized kinetics.
Advanced Analytical Technologies for Targeted Quantification and Broad Response Mapping
The dialysate collected during an in vivo microdialysis experiment is highly pure, free of large proteins and cellular debris, making it an ideal matrix for mass spectrometry. However, sample volumes are exceptionally small (often 1–2 μL per time point), and analyte concentrations can be in the low picogram/mL range.

To overcome these analytical limits, we employ state-of-the-art mass spectrometry platforms:
- High-Sensitivity Triple Quadrupole LC-MS/MS: Used for the absolute quantitation of classic neurotransmitters, their metabolites, and free drug concentrations. We strongly recommend our targeted neuropeptide quantification workflows when assessing low-abundance signaling peptides in the dialysate.
- High-Resolution Orbitrap Mass Spectrometry: Utilized for broad, discovery-phase mapping. If your goal is to discover novel biochemical responses to a stimulus, our untargeted neuropeptidomics and neuropeptidome profiling platforms can identify unexpected molecular shifts in the extracellular space.
Step-by-Step Microdialysis Workflow and QC Checkpoints
Our microdialysis workflows incorporate strict surgical and analytical quality controls to ensure data reliability and animal welfare.
Typical Results: Visualizing Dynamic Neurochemical and PK/PD Responses
We deliver comprehensive data packages that transform continuous sampling points into clear pharmacokinetic and neurochemical models.
Stimulus-Induced Flux Dynamics
PK/PD Exposure-Response Bridge
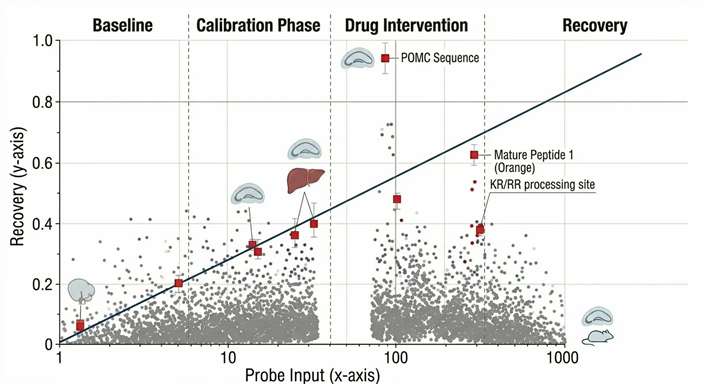
Zero-Net-Flux Calibration
Trace-Level LC-MS/MS Quantitation
Microdialysis vs. CSF Sampling vs. Tissue Homogenate
When planning an in vivo study, choosing the correct sampling methodology is crucial for data accuracy. Microdialysis offers unique advantages over bulk tissue or fluid collection.
| Feature | In Vivo Microdialysis (ISF) | Cerebrospinal Fluid (CSF) Sampling | Whole Tissue Homogenate |
|---|---|---|---|
| Analyte Captured | Unbound, free molecules only | Free molecules (but generalized) | Total (bound + intracellular + free) |
| Spatial Resolution | Highly localized (specific brain nucleus) | Global / Ventricular pool | Poor (destroys spatial context) |
| Temporal Resolution | High (Continuous, minute-level) | Moderate (Intermittent discrete samples) | None (Single endpoint snapshot) |
| Animal Survival | Longitudinal monitoring in one animal | Typically serial sampling (can cause stress) | Terminal per time point |
| Primary Limitation | Low recovery rates require sensitive MS | CSF does not perfectly equal ISF | Cannot assess pharmacological availability |
Sample and Experimental Design Requirements
Proper study design is vital for successful in vivo sampling. We customize probe selection, flow rates, and analytical methods to align with your specific target and model.
| Model / Tissue | Probe Type | Recommended Sampling Frequency | Key Experimental Inputs | Typical Deliverables |
|---|---|---|---|---|
| Freely Moving Brain (Rat/Mouse) | 1-2 mm active membrane (e.g., 20 kDa MWCO) | 10 – 20 minutes | Target brain region, analyte class, dosing schedule, behavior pairing | Neurotransmitter baseline & flux curves |
| Anesthetized Brain / Acute | Ultra-fine or standard concentric | 5 – 15 minutes | Exact stereotaxic coordinates, specific localized stimuli | High-resolution transient peak analysis |
| Peripheral Tissue (Muscle/Adipose) | Linear or flexible concentric probe | 20 – 30 minutes | Tissue type, physiological state (fasted/fed) | Local metabolite flux, lipolysis markers |
| Tumor Microenvironment | Large surface area, high MWCO | 30 – 60 minutes | Tumor model, cell line, tumor volume at implantation | Intratumoral free drug AUC |
Recovery, Calibration, and Data Interpretation in In Vivo Microdialysis
A critical component of in vivo microdialysis is understanding that it is a non-equilibrium technique. The concentration of an analyte in the collected dialysate is lower than its true concentration in the extracellular space. Interpreting this data correctly requires rigorous methodological calibration.
- Relative Recovery vs. Flow Rate: The amount of analyte crossing the membrane (relative recovery) is inversely proportional to the perfusion flow rate. We optimize the flow rate (typically 0.5 to 2.0 μL/min) to balance the need for high temporal resolution (frequent sampling) with the need for sufficient analytical sensitivity (higher recovery).
- Probe Calibration: To estimate actual tissue concentrations, we perform in vitro recovery calibrations for specific analytes across different temperatures and matrices to correct the raw dialysate concentrations.
- Zero-Net-Flux Considerations: For studies requiring absolute, rather than relative, extracellular concentrations, we can employ the zero-net-flux (ZNF) or quantitative microdialysis method. By perfusing the probe with varying known concentrations of the analyte and plotting the net gain or loss, we can precisely calculate the true baseline concentration in the ISF.
- Data Interpretation: Our final reports provide extensive guidance on interpreting these values, ensuring you understand the difference between relative dialysate shifts and absolute tissue exposure when constructing your PK/PD models.
Disclaimer: All services and analytical platforms described are intended for Research Use Only (RUO). Not for use in diagnostic procedures.


