Resolving CNS Heterogeneity via CSF & Biofluid Peptidomics
Neuropeptides are indispensable signaling molecules orchestrating complex physiological and pathological processes within the central nervous system (CNS). While analyzing brain tissue provides fundamental mechanistic insights, accelerating translational research requires identifying reliable signatures in biofluids. Because cerebrospinal fluid (CSF) physically bathes the brain and spinal cord, it serves as the most direct biofluid window into the neurochemical state of the CNS.
However, detecting functional, low-molecular-weight peptides in biofluids is notoriously difficult due to extreme dynamic ranges, matrix effects, and rapid ex vivo degradation. To bridge the gap between basic neuroscience and biomarker translation, R&D researchers require a highly robust and reproducible analytical platform. Through our comprehensive neuropeptidomics services, we provide an end-to-end research engine—enabling pharmaceutical companies and translational medical centers to confidently move from untargeted screening to rigorous pre-clinical cohort validation.
Translational Service Packages: Tailored for Your Development Stage
We understand that clinical research is not a "one-size-fits-all" endeavor. To meet the diverse needs of pharma R&D and clinical investigators, we have productized our workflows into three highly specialized, decision-driven translational modules:
How to Choose the Right Package:
- Choose Discovery Elite if: You are exploring completely novel biomarkers, mapping unannotated PTMs, or elucidating disease mechanisms in pilot cohorts.
- Choose Translational Bridge if: You need to rapidly validate top candidates and develop targeted PRM assays right after pilot screening to reduce biomarker attrition risk.
- Choose Targeted Pro if: You already have candidate biomarkers and require high-throughput, absolute quantification for large-cohort pre-clinical validation.
Strategic Choice: MS Peptidomics vs. Antibody-Based Panels
When designing a translational biomarker study, researchers often evaluate high-throughput antibody-based multiplex panels (such as Olink or Luminex) alongside mass spectrometry.
| Feature | MS Peptidomics (Creative Proteomics) | Antibody-Based Panels (Olink/ELISA) |
|---|---|---|
| Novel Discovery | Yes. Identifies unknown peptides/isoforms without relying on pre-existing panels. | No. Strictly limited to pre-defined, known protein targets. |
| Isoform Specificity | High. Precisely distinguishes inactive Pro-hormones from active Mature peptides. | Low. Frequent cross-reactivity between pro-forms and mature variants. |
| PTM Resolution | Direct. Detects critical modifications like C-terminal Amidation and Phosphorylation. | Indirect/Impossible without highly specific custom antibodies. |
| Validation Bridge | Seamless. Smooth transition from untargeted discovery to absolute PRM quantification. | Requires shifting to a completely different technology for validation. |
Overcoming Biofluid Complexity: Technical Performance You Can Trust
Conducting biofluid peptidomics demands highly specialized pre-analytical interventions. Our platform directly addresses decision-maker concerns regarding sensitivity, reproducibility, and data bias, delivering verified performance metrics:
- Technical Depth & Precision: We routinely resolve >1,000 to 2,000+ endogenous peptides per human CSF sample, and 500 to 1,200+ peptides in plasma/serum. We maintain stringent analytical stability with intra-batch CVs <15% and inter-batch CVs <20%.
- Proprietary Anti-Degradation SOPs: Endogenous ex vivo proteases immediately begin cleaving proteins post-collection. We provide mandatory collection protocols, utilizing rapid thermal inactivation and specialized quenching buffers to lock the peptidome in its native state, ensuring the fragments analyzed are true biological reflections.
- Bias-Free Low-Molecular-Weight Enrichment: CSF and plasma are dominated by albumin and IgGs. Rather than antibody-based depletion—which can cause the "off-target" loss of smaller peptides—we utilize a specialized combination of MWCO ultrafiltration and multi-step solid-phase extraction (SPE) to enrich the intact <10kDa fraction without immunological bias.
- Multi-Center Normalization: For large-scale multi-site research cohorts, our bioinformatics pipeline incorporates internal standard spiking and bridge-sample integration to computationally eliminate batch effects.
Integrated Workflow: From Discovery to CSF Biomarker Validation
A robust translational pipeline requires a closed-loop system. Our integrated platform ensures that findings from quantitative peptidomics discovery are immediately actionable for large-scale pre-clinical validation.
Biofluid Sample Submission Requirements
Proper pre-analytical handling is the primary determinant of success in biofluid peptidomics workflows.
| Biofluid Type | Min. Required Volume | Collection SOP & Stabilization Notes | Exclusion Criteria |
|---|---|---|---|
| Human CSF | 150 – 200 µL | Low-protein-binding tubes. Add protease inhibitors immediately. Snap-freeze in LN2. | Visible RBC (Hemolysis index > 500 mg/dL). |
| Plasma / Serum | 200 – 500 µL | EDTA-Plasma is preferred. Rapid centrifugation (4°C). Avoid freeze-thaw cycles. | Repeated freeze-thaw (>2 cycles). |
| Mouse / Rat CSF | 20 – 50 µL | Pooling of samples from the same group recommended. Requires Nano-LC setup. | N/A (Consult for micro-volume protocols). |
Demo Results: Translational Insights from Biofluid Cohorts
Our platform translates complex multidimensional mass spectrometry data into publication-ready visual insights. These demo results represent typical data outputs designed to accelerate your peptide biomarker discovery and downstream modeling.
Biomarker Performance (ROC/AUC Modeling)
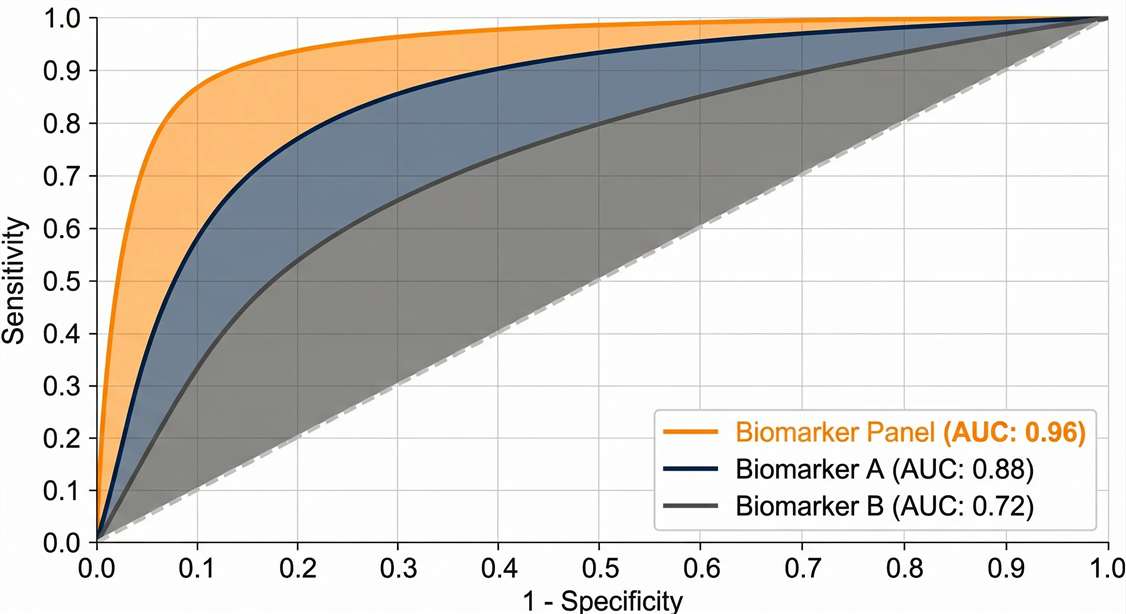
Statistical evaluation of the sensitivity and specificity of individual peptides or multiplexed biomarker panels for cohort classification.
Cohort Stratification (PCA/PLS-DA Analysis)
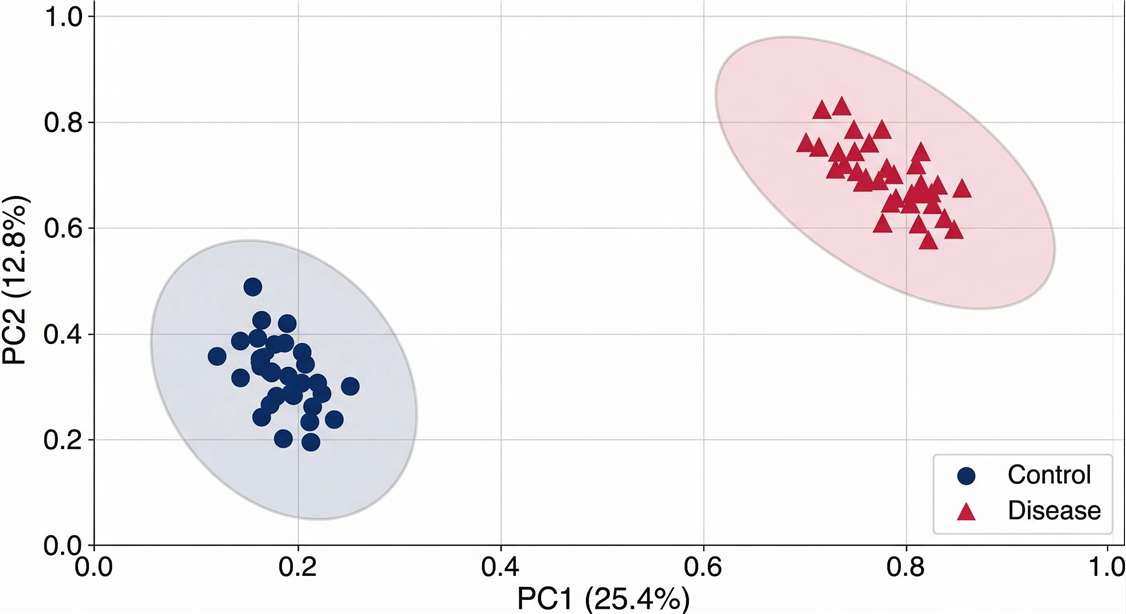
Validation of molecular distinctness between clinical groups and demonstration of batch-effect correction across large samples.
Precursor Cleavage & Processing Maps
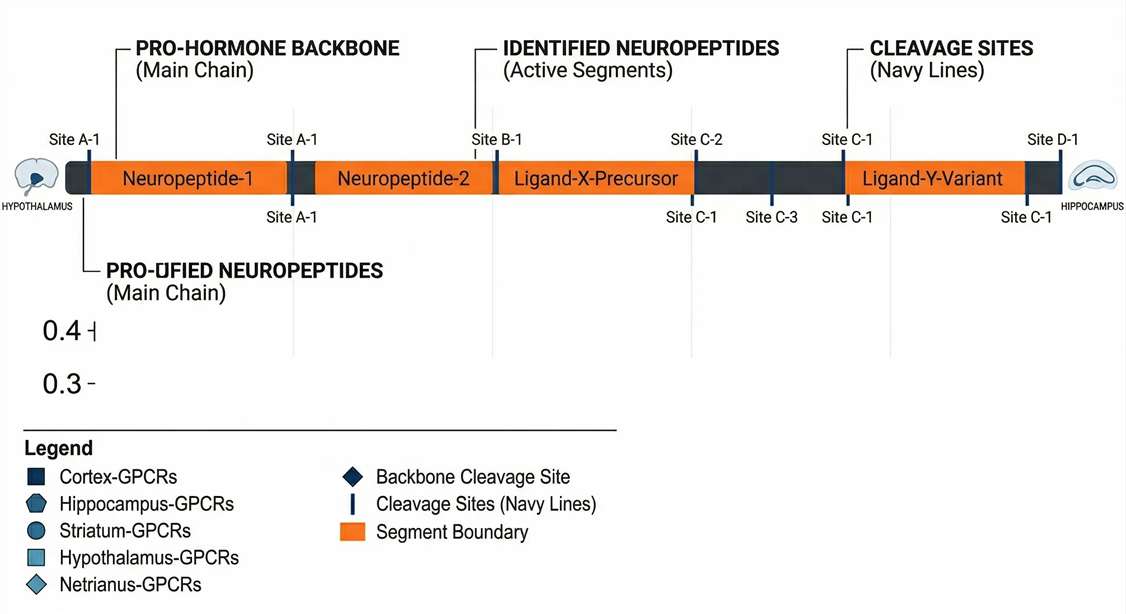
Mapping identified CSF peptides back to their source pro-hormones to reveal abnormal proteolytic activity in disease models.
Targeted Quantification (PRM Scatter Plots)
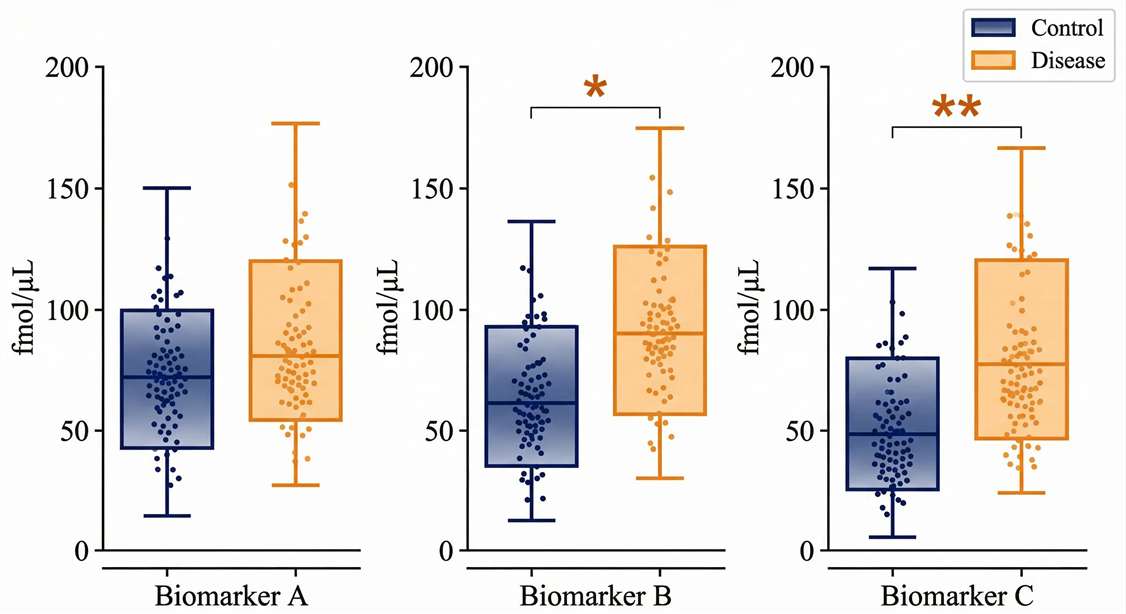
Definitive absolute concentration distributions (fmol/µL) across clinical groups, providing confidence for pre-clinical decision-making.
Deliverables | What You Will Receive
Upon project completion, we provide a comprehensive documentation package designed to support your regulatory submissions and internal archiving:
- Identified Neuropeptide Inventory (CSV/Excel): Detailed list of sequences, PTMs, charge states, and confidence scores (1% FDR).
- Quantitative Abundance Matrix: Normalized intensity data for all cohorts, ready for independent statistical re-analysis.
- PTM & Cleavage Annotation Report: Deep mapping of amidation, phosphorylation, and pro-hormone processing patterns.
- Executive Statistical Summary (PDF): High-level overview including PCA, Volcano plots, Heatmaps, and ROC analysis.
- Targeted PRM Assay Pack: For validation projects, includes transition lists, collision energies, and SIS-normalized absolute concentration tables.
- Raw Data Access: Full access to high-resolution MS/MS raw files.
Disclaimer: All services and platforms described are for Research Use Only (RUO). Not for use in diagnostic procedures.
Case Study: CSF Peptidomics Reveals Disease-Associated Peptide Signatures in Neurodegeneration
Journal: Molecular & Cellular Proteomics
Published: 2024
Summary
Recent studies using LC-MS/MS–based peptidomics have demonstrated that cerebrospinal fluid (CSF) contains a diverse repertoire of endogenous peptides reflecting central nervous system (CNS) activity. By combining peptide enrichment strategies with high-resolution mass spectrometry, researchers have successfully profiled CSF samples from neurodegenerative disease cohorts and identified disease-associated peptide signatures beyond conventional protein biomarkers.
These findings highlight the value of CSF peptidomics as a complementary approach to traditional proteomics for biomarker discovery and translational research.
Methods
The study applied a biofluid-specific peptidomics workflow optimized for endogenous peptide preservation:
- Sample Type: Human CSF from control and neurodegenerative disease cohorts
- Pre-analytical control: Rapid stabilization and protease inhibition to minimize ex vivo degradation
- Peptide enrichment: Molecular weight cut-off (MWCO) filtration combined with solid-phase extraction (SPE) to isolate peptides <10 kDa
- LC-MS/MS profiling: High-resolution mass spectrometry for deep endogenous peptide detection
- Data analysis: Multi-step database searching and statistical filtering to control false discovery rate (FDR <1%)
Results
The workflow enabled comprehensive profiling of the CSF peptidome, revealing:
- Hundreds to thousands of endogenous peptides detected per sample
- Identification of differential peptide signatures between disease and control groups
- Detection of precursor-derived peptide fragments associated with neurosecretory and synaptic proteins
- Evidence of disease-associated proteolytic processing changes, reflected in altered peptide cleavage patterns
Several peptide candidates demonstrated strong discriminatory potential between cohorts, supporting their relevance as translational biomarkers.
Key Findings
- Peptide-level profiling provides additional biological resolution beyond total protein measurements
- Cleavage-specific peptide variants can indicate altered enzymatic activity in neurodegenerative pathways
- Low-abundance peptides remain detectable with optimized enrichment and stabilization workflows
- The approach supports a direct transition from discovery to targeted validation workflows
Relevance to Translational Peptidomics Services
These findings illustrate the critical role of optimized CSF peptidomics workflows in biomarker discovery:
- Accurate detection depends on anti-degradation sample handling
- Reliable results require peptide-focused enrichment rather than standard proteomics digestion
- Translational value is enhanced by integrating discovery with targeted validation (e.g., PRM)
These principles are directly reflected in our CSF & Biofluid Translational Neuropeptidomics platform, which supports both early-stage discovery and large-cohort validation in CNS research.


