Measuring HPA Axis Responses Without Sampling-Induced Stress
A fundamental challenge in studying the Hypothalamic-Pituitary-Adrenal (HPA) axis is the observer effect: the act of collecting a biological sample often induces an acute, profound stress response that dramatically distorts the true baseline being measured. In rodent models, routine restraint, cage movement, or handling can cause plasma corticosterone levels to spike exponentially within just a few minutes, completely masking the authentic pharmacodynamic (PD) effects of experimental anxiolytic or anti-depressant drug candidates. When evaluating drug efficacy, a distorted baseline severely diminishes the statistical power required to observe target engagement.
To acquire authentic biological readouts, we combine highly specific targeted liquid chromatography-tandem mass spectrometry (LC-MS/MS) with comprehensive pre-analytical guidance. By pairing precise laboratory quantification with sophisticated multi-matrix strategies—such as measuring accumulated stress in hair or feces rather than relying solely on acute blood draws—researchers can accurately separate underlying neuroendocrine states from acute sampling artifacts. This holistic approach ensures that measured fluctuations in circulating glucocorticoids are a direct result of the applied pharmacological or behavioral intervention, yielding data that withstands rigorous peer review.
Improving Cortisol and Corticosterone Specificity with Targeted LC-MS/MS
Immunoassays (such as ELISA or RIA) remain excellent tools for high-throughput, routine screening in contexts where measuring a general class of total circulating glucocorticoids is sufficient. However, targeted LC-MS/MS is highly preferred when absolute structural specificity is required. This is particularly crucial when working with complex non-invasive matrices that suffer from extreme lipid interference, or when your study design explicitly demands the clear separation of active glucocorticoids from their biologically inactive downstream metabolites.
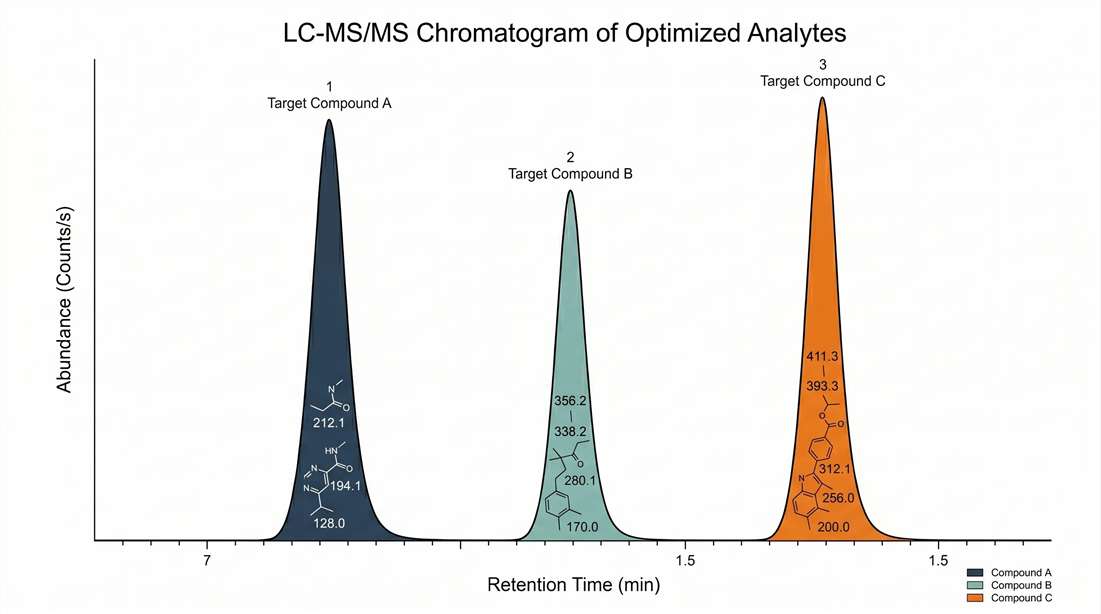
Steroid hormones possess highly conserved, rigid tetracyclic structural backbones that are notoriously difficult for antibodies to differentiate. Cortisol and its biologically inactive metabolite cortisone, for example, differ only by a single functional group (a hydroxyl versus a ketone at the C-11 position). In widely used rodent models, corticosterone and 11-dehydrocorticosterone share a similarly challenging structural resemblance. Standard antibody-based kits can easily cross-react with these structural analogs, as well as with hundreds of other exogenous dietary sterols or microbial metabolites present in complex matrices like feces, leading to artificially inflated baseline measurements and high inter-assay variability.
Our analytical platform utilizes state-of-the-art Triple Quadrupole (QQQ) mass spectrometry systems operating in Multiple Reaction Monitoring (MRM) mode. LC-MS/MS identifies molecules based on a dual-verification system: exact chromatographic retention times paired with specific precursor-to-product ion fragmentation patterns. This provides absolute structural specificity across both human/NHP (cortisol-dominant) and rodent (corticosterone-dominant) models, completely bypassing the cross-reactivity risks inherent to standard immunoassays and delivering unparalleled quantitative confidence.
Configurable HPA Panel: Upstream Peptides, Downstream Steroids, and Rhythm Markers
Evaluating the stress response effectively often requires a holistic view beyond the measurement of a single downstream steroid. The HPA axis functions as an intricate feedback loop. We offer a highly configurable panel approach, enabling researchers to track the endocrine signaling cascade from initial hypothalamic activation, through pituitary amplification, down to peripheral adrenal execution. When studies require parallel screening of upstream peptides (CRH/ACTH) alongside highly lipophilic steroids, we utilize High-Resolution Accurate Mass (HRAM) Orbitrap systems to support robust, comprehensive profiling.
| Biological Target Area | Selected Analytes | Physiological Relevance in Stress Models |
|---|---|---|
| Upstream HPA Peptides | CRH (Corticotropin-Releasing Hormone), ACTH (Adrenocorticotropic Hormone) | Primary hypothalamic and pituitary drivers initiating the neuroendocrine stress cascade; crucial for evaluating central feedback inhibition. |
| Downstream Steroids | Cortisol, Corticosterone, Cortisone, Aldosterone | Primary active glucocorticoids (species-dependent) and mineralocorticoids mediating peripheral metabolic adaptation and immune suppression. |
| Rhythm & Buffering Markers | Melatonin, DHEA, DHEA-S | Key markers of circadian sleep/wake cycles and neuroactive steroids that actively buffer the brain against chronic glucocorticoid toxicity. |
| SAM Axis (Catecholamines) | Epinephrine, Norepinephrine | Rapid, acute "fight-or-flight" sympathetic nervous system signaling molecules indicating immediate stress arousal. |
For clients aiming to integrate both peptides and steroids from ultra-low sample volumes (e.g., < 50 μL of mouse plasma), analytical feasibility and extraction efficiency are evaluated on a case-by-case basis depending on the biologically relevant Lower Limit of Quantification (LLOQ) requested. For research requiring deep characterization of central nervous system signaling, neuroinflammation, or CSF partitioning, our assays seamlessly adapt for CSF and biofluid translational neuropeptidomics, serving as a powerful, interconnected extension to our targeted neuropeptide quantification platform.
Translational Applications in Neuropsychopharmacology & Toxicology
Our fit-for-purpose LC-MS/MS assays are engineered to deliver the quantitative rigor and reproducibility required for advanced preclinical drug discovery and complex translational studies:
Multi-Matrix Strategy: Acute Spikes vs. Cumulative Stress Burden
Matching the appropriate biological matrix to the experimental timeline is critical for experimental validity. An acute panic or startle model requires a fundamentally different analytical strategy than a progressive, chronic depression model. We deploy multiple validated extraction routes tailored to overcome the unique chemical complexities of each distinct matrix.
Plasma & Saliva: Acute Dynamics and Diurnal Slopes
Blood (plasma/serum) and saliva represent the systemic, real-time physiological status of the HPA axis. They are the matrices of choice for high-frequency temporal sampling over a 24-hour period to map precise diurnal slopes, or for tracking the immediate neuroendocrine spike and subsequent negative feedback recovery following an acute, short-duration stressor (e.g., forced swim test, restraint stress, or fear conditioning).
Hair & Feces: Cumulative Stress Burden
Repeated, high-frequency blood sampling is highly counterproductive in chronic models, as the sampling itself becomes a chronic stressor. As steroids circulate systemically, they are continuously incorporated via passive diffusion into growing hair shafts, and heavily metabolized via enterohepatic circulation before being excreted into feces.
- Hair cortisol/corticosterone provides a stable, retrospective window into the cumulative stress burden accumulated over several weeks.
- Fecal corticosterone metabolites offer a pooled, integrated 12-to-24-hour average of overall HPA activity.
Both of these non-invasive matrices naturally buffer the acute hormone spikes caused by daily laboratory handling, yielding a much more accurate reflection of chronic pathological states and long-term neuroendocrine adaptations.
Pre-Analytical SOPs: Preventing Blood Draw Stress in Rodent Models
Reliable, high-fidelity bioanalytical data begins long before the sample reaches the mass spectrometer; it begins in the vivarium. To support precise pharmacological profiling and reduce group variance, we provide expert guidance on managing the fragile HPA baseline in rodent models prior to sample submission.
- The 2-Minute Window: Endogenous corticosterone levels begin to rise rapidly upon environmental disturbance. We strongly recommend sampling techniques such as submandibular or tail-nick bleeding completed strictly within 2 minutes of the investigator first opening the home cage. This rapid execution is essential to capture a true, un-stressed physiological baseline before the adrenal cortex can respond to the handling stimuli.
- Habituation Protocols: Acclimating animals with 3 to 5 days of deliberate mock handling and familiarization with restraint tubes prior to the actual study day drastically reduces procedural stress artifacts. Habituation effectively normalizes the baseline, greatly enhancing the statistical signal-to-noise ratio when evaluating the effects of a test compound.
- Alternative Sampling: Whenever biologically feasible for the study design, we encourage transitioning to non-invasive fecal bolus collection, or integrating real-time in vivo microdialysis and temporal fluxomics to continuously monitor dynamic neurochemical shifts in freely moving animals with absolute minimal handling.
Sample Requirements for Diurnal Slopes and Chronic Accumulation Studies
The stabilization and collection context are just as important as the analytical method. Below are general guidelines optimized to ensure target integrity for our HPA axis panel.
| Matrix Type | Species & Model Context | Min Volume / Mass | Critical Pre-Analytical SOPs & Context |
|---|---|---|---|
| Blood (Plasma/Serum) | Rodent Acute Stress, Human Circadian. | 50 μL | Require rapid bleed (< 2 mins from cage opening) to prevent handling-induced corticosterone spikes. Rapid handling, chilled processing, and EDTA tubes with matrix-appropriate stabilization are recommended. |
| Hair / Fur | Chronic Stress Models (e.g., CUMS), Aging. | 10 - 20 mg | Shave at baseline, regrow, collect. Measures cumulative burden over weeks. Avoid chemical washing prior to lab submission. |
| Feces | Wildlife Toxicology, Chronic Stress. | 50 - 100 mg | Freeze immediately at -20°C. Normalizes acute handling stress; represents a 12-24 hour pooled metabolic excretion. |
| Saliva | Human Clinical Cohorts, Sleep Studies. | 100 μL | Use specific collection swabs (e.g., Salivettes). Ideal for mapping diurnal cortisol slopes and awakening responses non-invasively. |
Representative Data Deliverables: Circadian Curves and Matrix Recovery
We deliver comprehensive, fit-for-purpose datasets meticulously designed to reflect what chronobiology and stress pharmacologists value most. Absolute quantification is achieved using robust surrogate matrix calibration curves paired with representative class-specific Stable Isotope-Labeled Internal Standards (SIL-IS) to correct for ion suppression. We provide strict bioanalytical validation reporting (LLOQ, recovery rates, and CV%), while biological interpretation is reliably anchored to the client’s own vehicle/control groups.
Baseline Chromatographic Separation
Circadian Rhythm & Acrophase Mapping
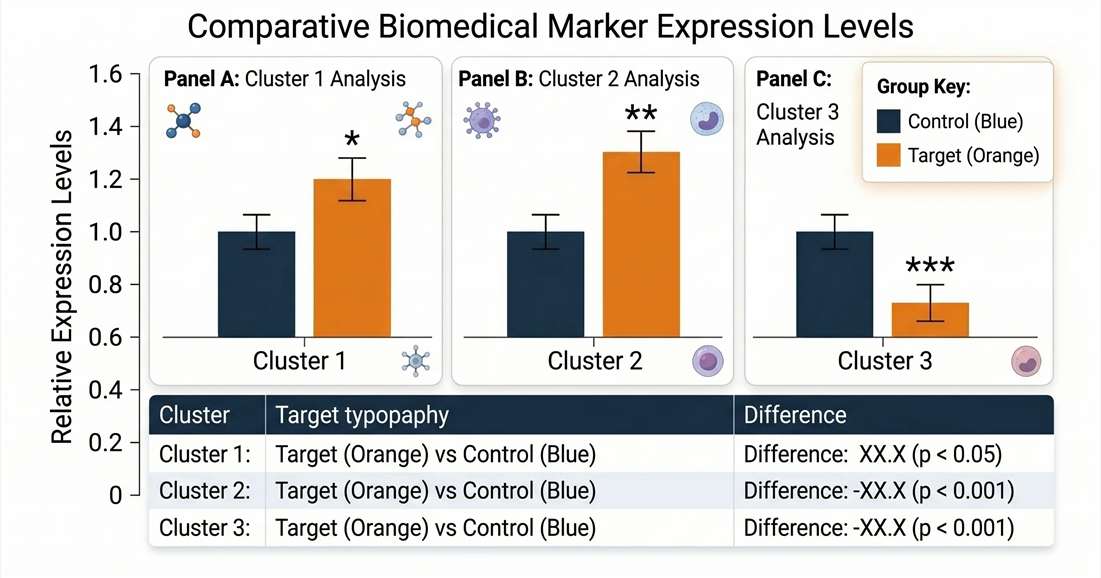
Acute vs. Chronic Matrix Comparison
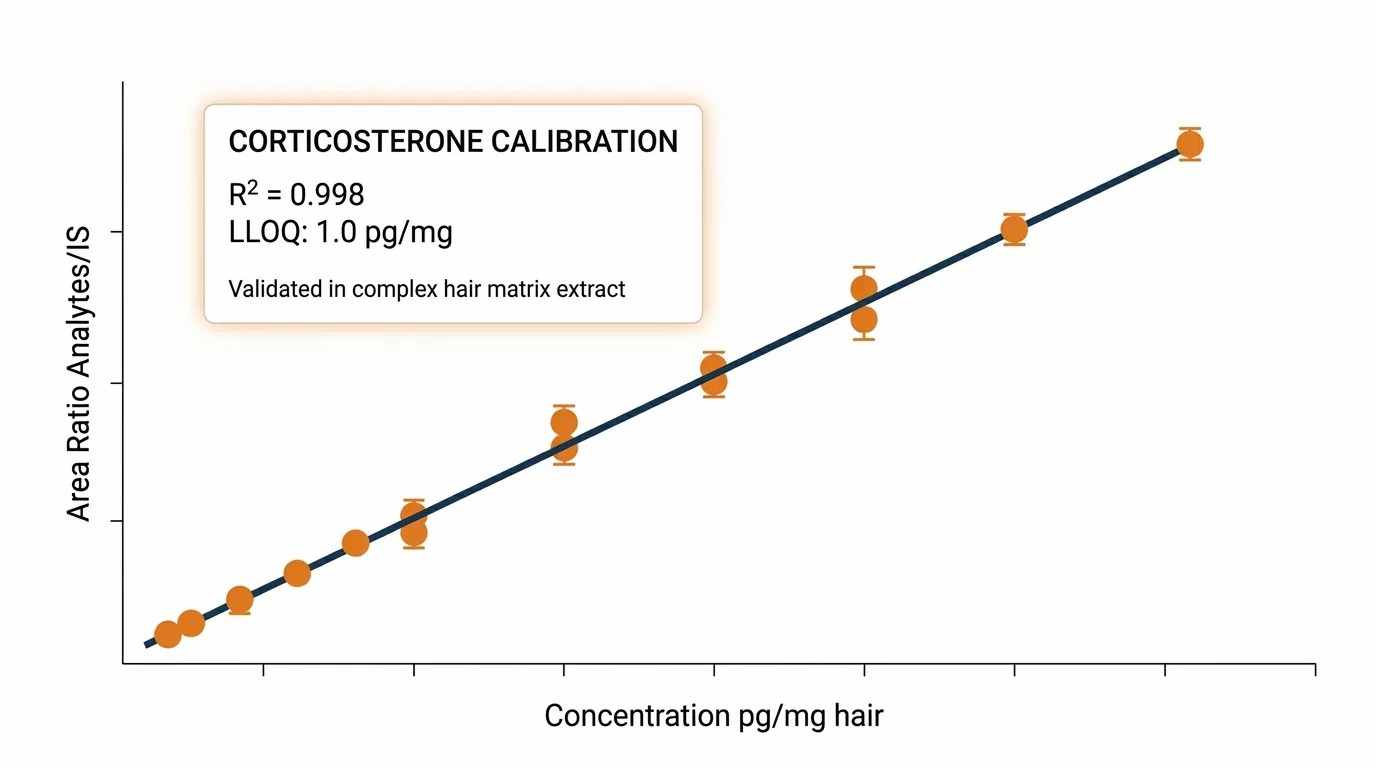
Matrix Recovery QC Data
Frequently Asked Questions (FAQ)
References
- Guidelines for the measurement of hair cortisol and cortisone in psychoneuroendocrinology
- Methodological considerations for corticosterone analysis in rodent stress models
- LC-MS/MS analysis of steroid hormones in saliva and hair: Applications in translational research
Disclaimer: All services and analytical platforms described are intended for translational research and preclinical support. Research Use Only (RUO). Not for use in diagnostic procedures.


