- Services
- FAQ
- Case Study
- Demo
- Related Services
- Support Documents
- Inquiry
Our SV-AUC Services for Antibody Therapeutic Characterization
Our SV-AUC services deliver precise, high-resolution analysis of proteins in their natural solution state. By tracking how molecules move under strong centrifugal force, we can clearly separate monomers, small clusters (oligomers), and larger aggregates in antibodies and protein therapeutics. This approach lets you accurately measure particle size distribution and sample heterogeneity—without altering the molecules through labeling or surface binding.
We design SV-AUC experiments tailored to your specific project goals, whether it is early formulation screening, stability assessment, or comparability studies. Our team integrates SV-AUC data with complementary methods:
Advantages of Our SV-AUC Services
- Analyze proteins in native solution without labeling or immobilization
- Detect and quantify aggregates with high resolution
- Resolve monomers, oligomers, and subvisible aggregates
- Integrate seamlessly with DLS, SEC-MALS, and mass spectrometry
- Deliver expert interpretation and actionable recommendations for formulation and stability
Precise Detection of Antibody Aggregates
Aggregation can compromise the efficacy and safety of antibody therapeutics. Our SV-AUC services enable detailed characterization of soluble and subvisible aggregates, providing critical data for formulation optimization.
- Soluble and Subvisible Aggregates: We detect dimers, trimers, and higher-order assemblies, allowing you to identify potential stability issues early.
- Quantitative Aggregation Profiles: Using sedimentation coefficient distributions, we quantify the abundance of each species for informed decision-making.
- Real-Time Aggregation Monitoring: We track aggregation dynamics under stress conditions such as temperature shifts, agitation, or buffer changes.
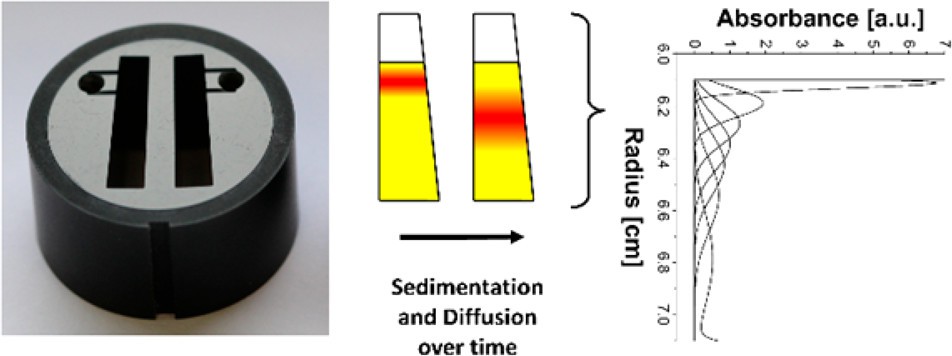 Figure 1. The band sedimentation experiment in AUC (Schneider C M, et al., 2018).
Figure 1. The band sedimentation experiment in AUC (Schneider C M, et al., 2018).Accurate Particle Size and Molecular Weight Analysis
Our SV-AUC service provides hydrodynamic and molecular weight measurements in near-native conditions, enabling you to understand molecular conformation and complex formation.
- Sedimentation Coefficient Analysis: We generate high-resolution c(s) profiles to resolve heterogeneous protein populations and identify aggregates.
- Hydrodynamic Radius Calculation: Diffusion-corrected analysis allows precise determination of protein size, shape, and assembly state.
- Molecular Weight Determination: We calculate molecular weights of monomers, oligomers, and complexes without labeling.
SV-AUC for Antibody Stability and Developability
Understanding stability and developability is crucial for successful biopharmaceutical development. Our SV-AUC services help you monitor stability under stress and optimize formulations for long-term storage.
- Stress-Induced Aggregation Monitoring: We evaluate heat, pH, and excipient effects on antibody stability.
- Long-Term Stability Assessment: Track sedimentation profiles over time to predict shelf life and storage conditions.
- Developability Evaluation: Determine if your candidate maintains monomer integrity and resists aggregation under various conditions.
Applications of Antibody Therapeutic Development
Our SV-AUC services are versatile and support multiple stages of development:
- Monoclonal Antibody Aggregation: Quantify monomers, dimers, and higher-order aggregates for quality control.
- Bispecific and Complex Proteins: Resolve heterogeneous species in multi-domain constructs.
- Nanoparticle and Viral Vector Characterization: Evaluate size, homogeneity, and aggregation in gene therapy vectors.
- Biosimilar Comparability Studies: Assess aggregate profiles and molecular stability to support biosimilar development.
Creative Proteomics' SV-AUC Workflow
- Sample Preparation: Optimize protein concentration, buffer composition, and viscosity while minimizing particulate contamination.
- Experimental Design: Select rotor speed, temperature, and duration tailored to monomers, oligomers, or aggregates.
- Data Acquisition: Run SV-AUC in analytical ultracentrifuge cells and capture absorbance, interference, or fluorescence data.
- Advanced Data Analysis: Apply c(s) distribution, diffusion-corrected sedimentation, and hydrodynamic modeling to quantify molecular populations.
- Comprehensive Reporting: Provide sedimentation coefficient distributions, hydrodynamic radii, aggregation quantification, stability assessment, and formulation recommendations.
Sample Requirements
| Parameter | Recommended Requirement |
|---|---|
| Sample Type | Purified proteins, monoclonal antibodies, bispecific antibodies, protein complexes, nanoparticles. |
| Protein Concentration | Typically 0.1–1.0 mg/mL (adjustable depending on sample properties). |
| Sample Volume | 350–450 μL per cell. |
| Buffer Composition | Physiological buffers recommended (e.g., PBS, HEPES, Tris). |
| Buffer Matching | Dialyzed or buffer-exchanged samples preferred. |
| Particulate Removal | Filtered through 0.22 μm filter or centrifuged before analysis. |
Why Choose Creative Proteomics for SV-AUC?
- Over decades of experience in protein characterization.
- Custom experiment design for antibody and complex biologics.
- Integration with proteomics and stability workflows.
- Rapid turnaround and expert data interpretation.
- Trusted by R&D teams and CROs for reliable biopharmaceutical analysis.
FAQ
Q1: What is Sedimentation Velocity Analytical Ultracentrifugation (SV-AUC)?
A1: SV-AUC is a solution-based method that separates biomolecules based on how they move under high centrifugal force. By tracking this movement in solution, it allows us to determine particle size distribution, molecular weight, and the extent of aggregation.
Q2: What types of biomolecules can we analyze with SV-AUC?
A2: We routinely analyze monoclonal antibodies, bispecific antibodies, recombinant proteins, protein complexes, nanoparticles, viral vectors, and macromolecular assemblies in solution.
Q3: What information can SV-AUC provide for antibody therapeutics?
A3: SV-AUC enables quantification of monomers, oligomers, and aggregates, determination of sedimentation coefficient distributions, and evaluation of molecular heterogeneity and stability in antibody formulations.
Q4: How does SV-AUC detect protein aggregates?
A4: Aggregated proteins sediment faster than monomeric species during centrifugation. By analyzing sedimentation coefficient distributions, we identify and quantify aggregates, including dimers, trimers, and larger complexes.
Q5: What is a sedimentation coefficient?
A5: The sedimentation coefficient reflects how fast a particle moves under centrifugal force. It depends on particle size, shape, and density, and is commonly expressed in Svedberg units (S).
Q6: How does SV-AUC compare with DLS?
A6: DLS provides rapid particle size estimation, while SV-AUC offers higher resolution for heterogeneous mixtures, enabling quantitative separation of monomers and aggregates.
Q7: How does SV-AUC compare with SEC?
A7: SEC separates molecules using a column matrix, while SV-AUC analyzes samples directly in solution without stationary-phase interactions, reducing the risk of artifact formation.
Case Study
Case: Comprehensive Size Distribution and Composition Analysis of Adeno-Associated Virus Vector by Multiwavelength SV-AUC
Authors: Maruno T, et al.
Journal: Journal of Pharmaceutical Sciences
Year: 2021
DOI: 10.1016/j.xphs.2021.06.031
Background
Recombinant adeno-associated virus (AAV) vectors are commonly used in gene delivery applications. During production, not all particles are fully packaged, which can lead to empty particles, partially filled particles, and aggregates. These impurities can reduce effectiveness and may increase the risk of immune responses. Traditional analysis methods, such as chromatography and electron microscopy, often have limitations in resolution, accuracy, or the ability to reliably quantify these different particle types.
Methods
- AAV samples (AAV5-EP and AAV5-FP) were prepared in PBS, with some containing isotopically enriched water for density contrast experiments.
- SV-AUC determined sedimentation coefficient distributions (c(s)) and partial-specific volumes.
- MW-SV-AUC captured UV absorption profiles at multiple wavelengths (230–280 nm).
- Spectral deconvolution and multiple regression analysis quantified the protein-to-nucleic acid ratio of all particles.
- Molar extinction coefficients were calculated to enable accurate EP/FP ratio assessment.
Results
- SV-AUC accurately resolved EPs and FPs.
- MW-SV-AUC allowed quantification of IPs and aggregates by determining ssDNA/capsid ratios.
- Isosbestic wavelength analysis enabled EP/FP ratio measurement without extinction coefficients.
- SV-AUC showed superior accuracy and resolution compared to UV A260/A280 ratios or chromatography.
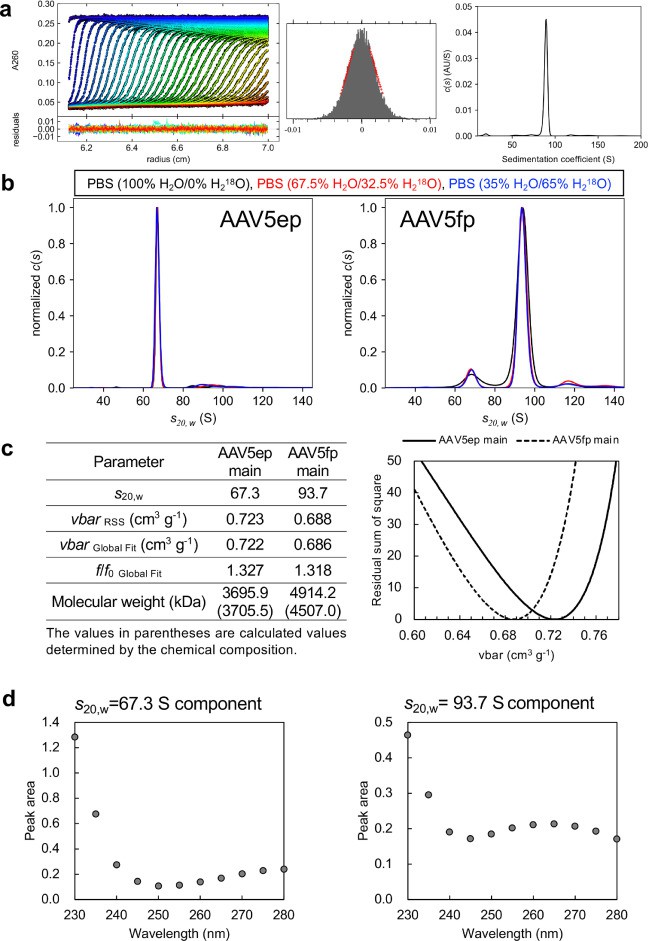 Figure 2. Characterization of AAV-EP and AAV-FP components.
Figure 2. Characterization of AAV-EP and AAV-FP components.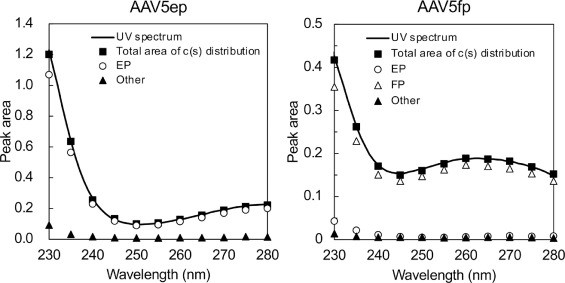 Figure 3. Examination of the consistency of the UV signals.
Figure 3. Examination of the consistency of the UV signals.Conclusion
This research provides a reliable, high-resolution tool for comprehensive characterization of AAV vectors, enabling precise quantification of EPs, FPs, IPs, and aggregates. This method enhances quality control in viral vector production and supports the development of safer, more effective gene therapies.
Demo
Demo: Sedimentation Velocity Analytical Ultracentrifugation of Oxidized Recombinant Full-Length Factor VIII
This study used SV-AUC to assess whether oxidation of recombinant factor VIII increases protein aggregation. Although oxidation reduced biological activity, no increase in aggregates was detected, indicating that immunogenicity is likely driven by structural changes rather than aggregation.
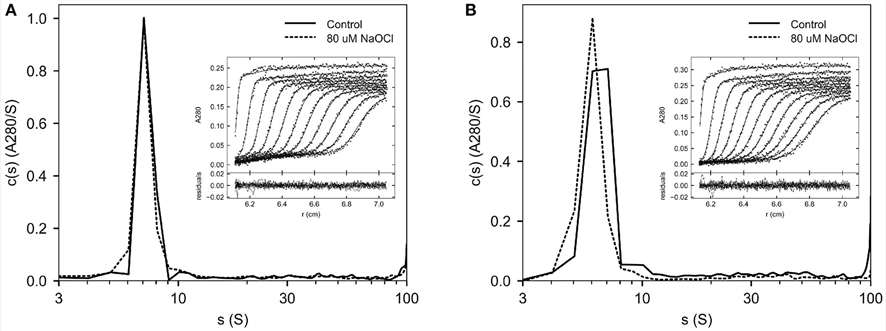 Figure 4. SV AUC of oxidized Helixate.
Figure 4. SV AUC of oxidized Helixate.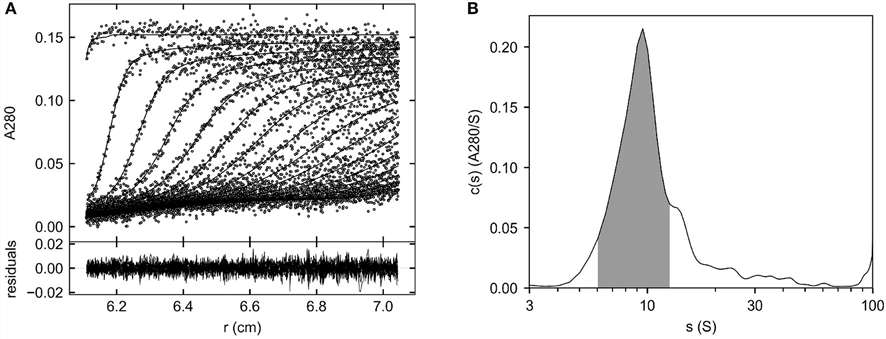 Figure 5. Major species of Helixate in formulation buffer determined by SV AUC.
Figure 5. Major species of Helixate in formulation buffer determined by SV AUC.References
- Schneider C M, Haffke D, Cölfen H. Band sedimentation experiment in analytical ultracentrifugation revisited. Analytical chemistry, 2018, 90(18): 10659-10663. DOI: 10.1021/acs.analchem.8b02768.
- Brown P H, Schuck P. Macromolecular size-and-shape distributions by sedimentation velocity analytical ultracentrifugation. Biophysical journal, 2006, 90(12): 4651-4661. DOI: 10.1529/biophysj.106.081372
- Maruno T, et al. Comprehensive size distribution and composition analysis of adeno-associated virus vector by multiwavelength sedimentation velocity analytical ultracentrifugation. Journal of Pharmaceutical Sciences, 2021, 110(10): 3375-3385. DOI: 10.1016/j.xphs.2021.06.031
- Zakas P M, et al. Sedimentation Velocity Analytical Ultracentrifugation of Oxidized Recombinant Full-Length Factor VIII. Frontiers in Immunology, 2020, 11: 150. DOI: 10.3389/fimmu.2020.00150
Related Services
Support Documents
KNOWLEDGE CENTER