High performance liquid chromatography (HPLC) is one of the most powerful techniques in analytical chemistry, which can separate, identify, and quantitate drugs and drugs-related impurities. A typical HPLC system consists of a stationary phase, a mobile phase of varying polarity, and an ultraviolet detector. The stationary phase is usually a solid column casing of silica support particles and a bonded substance immobilized on them. High pressure forces the components of the analyte to move through the column depending on their relative polarities. There are four different categories: normal phase chromatography, reverse phase chromatography, hydrophilic interaction chromatography, and ion exchange chromatography.
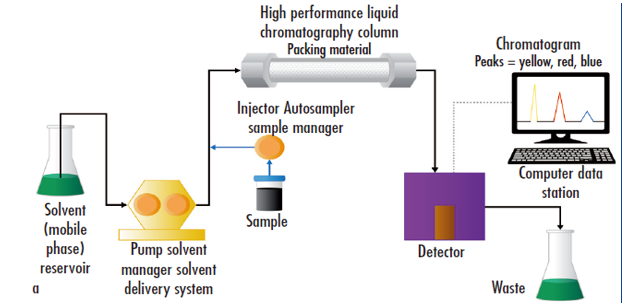
High performance liquid chromatography (Blum et al., 2014)
Normal phase chromatography
Normal phase chromatography separates components of the analyte based on their affinity for the stationary surface in the column. The unmodified silica particles are usually used with a gradient moving from non-polar to polar solvents. The absorption strength is a function of the polarity of the molecule. The more polar the molecule will be more strongly retained by the stationary phase, and be sequentially eluted from the column by solvents with increasing polarity.
Revers phase chromatography
Reverse phase chromatography utilizes with silica modified with 4- to 18-carbon chains, and separates components of the analyte based on hydrophobicity. On stationary phase, the more lipophilic compounds will adsorb more strongly. Then the molecule will be eluted from the column by polar solvent on the mobile phase.
Hydrophilic interaction liquid chromatography (HILIC)
HILIC is a variant of normal phase chromatography, and an alternative technique to effectively separate small polar compounds on polar stationary phases. The separation mechanism of HILIC is more sophisticated than that in normal phase chromatography, and HILIC can be complementary to other both normal and reverse phase chromatography. HILIC deploys traditional stationary phases, and mobile phases with rich organic solvents to enhance hydrophilic interactions between the compounds and the stationary phase. HILIC can separate small organic acids, drugs, many neutral and charged substances, and be successfully employed to analyze carbohydrates, peptides, polar pharmaceuticals, and so forth.
Ion exchange chromatography (IEC)
IEC separates the components of the analyte on basis of their total charge. In IEC, the stationary phases with anion-exchange resin or cation-exchange resin are used to separate different ionizable molecules based on their affinity for stationary surface in the column. The pH at which the net charge of a molecule equals zero is known as its isoelectric point (pI). The overall charge of a protein varies from the pH of their environments, which enables the separation of different molecules in a solution with certain pH.
We Can Provide Service Based on HPLC but Not Limited to:
- Abundant protein depletion
- Aggregate & fragment analysis
- Amino acid & cell culture analysis
- Antibody titer determination
- Charge variant analysis
- Glycan analysis
- Intact & subunit analysis
- Oligonucleotide analysis
- Peptide mapping
References
- Sahu P.K., Rao R.N., Cecchi T., et al. An Overview of Experimental Designs in HPLC Method Development and Validation. Journal of Pharmaceutical and Biomedical Analysis. Volume 147, 5 January 2018, Pages 590-611.
- Buszewski B. and Noga S. Hydrophilic interaction liquid chromatography (HILIC)—a powerful separation technique. Anal Bioanal Chem. 2012 Jan; 402(1): 231–247.
- Blum F. High performance liquid chromatography. British Journal of Hospital Medicine, February 2014, Vol 75, No 2.
- Jungbauer A and Hahn R. Ion-exchange chromatography. Methods Enzymol. 2009; 463:349-71.






