It has been found in all life form that protein lipidation, which is crucial in post-translational modifications (PTMs) that enable direct interaction with cell membranes. In the pattern of lipidation changing of membrane related proteins, the proteins have been detected in cancer and neurological disorders. It is a key determinant of its immunogenicity for lipidation at the N-terminal of recombinant outer surface protein A. Lipid modifications include Glycosyl-phosphatidylinositol anchor (GPI). At the C-terminus of proteins, it happens in the endoplasmic reticulum that the post-translational linkage between glycosylphosphatidylinositol and the modified proteins are translocated to the membrane, which are remain anchored to the outer leaflet of the cell membrane. Cholesterylation is also one of the the modifications. C-terminal attachment of a cholesterol molecule, which is another key lipidation detected in members of the mammalian Hedgehog family, important morphogens of a great importance in regulating cellular proliferation and differentiation. S-Isoprenylation: The recognition motif for isoprenylation is a C-terminus sequence known as CAAX box. Acylation is mainly limited to cysteine residues, thereby forming a labile and reversible thioester linkage.
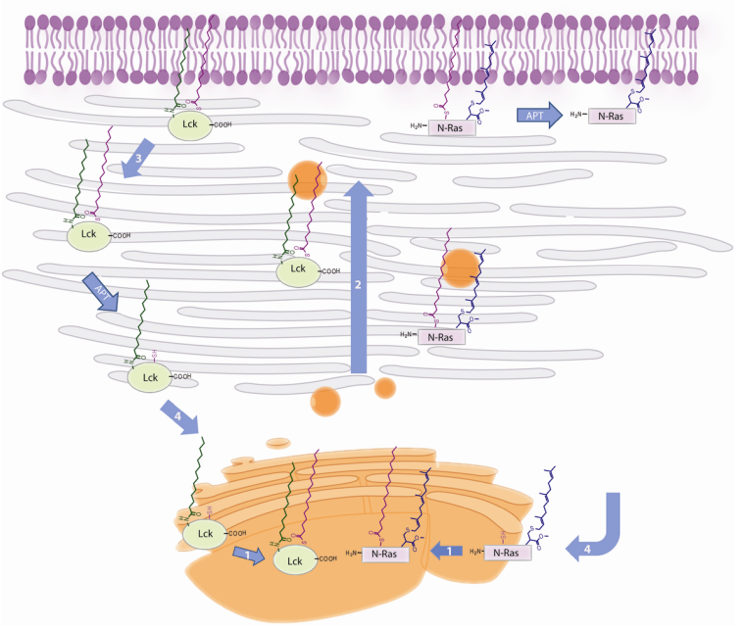 Figure 1: Protein Lipidation
Figure 1: Protein LipidationMethods for detecting protein lipidation have been exploited to predict GPI-anchored proteins, N-myristoylated, S-isoprenylated or S-palmitoylated proteins, and so on. The traditional approaches are based on the incorporation of radioactive precursors and subsequent detection of the proteins by fluorography. Mass spectrometry (MS) methods are universally applied, which are for the mapping of post-translational modifications. This technology submits crucial advantages. Bio-orthogonal probes, reactions compatible with biomolecules that allow their study in their native settings.
Creative Proteomics have a strict workflow to analyze lipidation to meet your requirements.






