Creative Proteomics offers a comprehensive protein intact mass analysis service using advanced mass spectrometry techniques to ensure the safety and efficacy of biopharmaceuticals. Our state-of-the-art mass spectrometry instruments and experienced scientists can accurately determine the molecular weight of intact proteins to help identify impurities, modifications, and structural changes.
Protein Purity Analysis: Intact mass analysis can help identify impurities in protein samples. By determining the molecular weight of intact proteins, the presence of impurities can be detected and characterized.
Structural Analysis: Intact mass analysis can provide information about the structure of proteins, including any changes that may have occurred during processing or storage.
Post-Translational Modification Analysis: Intact mass analysis can be used to identify and characterize post-translational modifications, such as glycosylation, phosphorylation, and acetylation.
Importance of Intact Mass Analysis in Protein Characterization
The characterization of protein-based therapeutics necessitates a critical step known as intact mass analysis, which provides crucial data on the primary structure of the molecule. Intact mass analysis entails the ascertainment of the weight of a protein or protein complex in its native state without fragmentation. This vital analytical technique helps discern discrepancies in protein molecular weight and glycosylation, as well as variations thereof, which can substantially impact protein function, stability, and immunogenicity.
The integrity of protein function and stability hinges on the maintenance of protein structure. Therefore, whether intentional or not, any modification can have a significant impact on the therapeutic efficacy of the protein. Post-translational modifications (PTMs), such as glycosylation, phosphorylation, and acetylation, can modify the chemical and physical properties of a protein, including its activity, stability, and pharmacokinetics. This emphasizes the criticality of assessing a protein's intact mass, which enables the identification of PTMs and structural modifications within the protein. Further, intact mass analysis of protein complexes can help determine the stoichiometry and composition of the complex.
The importance of intact mass analysis in protein characterization permeates the drug development landscape, from early discovery to late-stage clinical trials.
Early Discovery
During the early stages of drug discovery, intact mass analysis can play a pivotal role in selecting lead candidates, optimizing expression and purification conditions, and identifying process-related impurities. A protein's intact mass evaluation can give insights into the presence of post-translational modifications (PTMs), such as oxidation, deamidation, and truncation, which can arise during the recombinant protein production process. This information is crucial in determining the stability and quality of the protein, which can significantly affect the protein's bioactivity and half-life.
Late-stage Development
In the late stages of drug development, intact mass analysis becomes even more essential as it can help in the formulation development process and the assessment of biosimilarity. Formulation development requires an in-depth understanding of the protein's physicochemical properties, including solubility, stability, and compatibility with excipients. Intact mass analysis can provide vital information on the protein's oligomeric state and aggregation tendencies, which are critical in determining the protein's stability in the final drug formulation.
Biosimilarity Assessment
In the realm of drug development, ensuring the biosimilarity between a biosimilar and its reference product is a vital aspect of regulatory compliance. To achieve this, a comprehensive assessment of biosimilarity must be conducted. One analytical technique that has proven to be invaluable in this process is intact mass analysis. By examining the intact mass of both the biosimilar and the reference product, researchers can discern critical information regarding the protein's primary structure and post-translational modifications, aiding in the detection of any differences between the two products.
Mass Spectrometry Techniques for Intact Protein Analysis
Mass spectrometry (MS) represents a highly potent analytical tool, empowering researchers to unravel the molecular weight and composition of proteins. As a quintessential step in protein characterization, MS has assumed the mantle as the gold standard in analyzing the intact mass of proteins. Intact protein analysis provides a wealth of valuable information pertaining to the purity, identity, and structural integrity of the protein of interest. With a plethora of MS techniques at their disposal, researchers are presented with an abundance of options, each endowed with its own distinct advantages and limitations. The selection of a specific MS technique is thus contingent upon the attunement of experimental needs and the inherent characteristics of each technique.
Creative Proteomics utilizes various mass spectrometry techniques to perform intact protein analysis, including electrospray ionization (ESI), matrix-assisted laser desorption/ionization (MALDI), and time-of-flight (TOF) mass spectrometry. These techniques provide high-resolution and accurate analysis of protein structures.
Electrospray Ionization Mass Spectrometry (ESI-MS)
The use of Electrospray Ionization Mass Spectrometry (ESI-MS) to dissect intact proteins is an extensively utilized technique that is predicated upon the electrospray ionization of proteins into solution, ultimately resulting in the generation of ions that can be analyzed utilizing mass spectrometry. ESI-MS is a soft ionization technique that yields multiply charged ions, thus increasing the sensitivity of the analysis itself. The versatility of ESI-MS as a technique is exemplified by its applicability in analyzing a wide expanse of proteins, ranging from small peptides to large multi-subunit complexes. ESI-MS is particularly advantageous due to its ability to conserve non-covalent interactions, which renders it an ultra-valuable tool in the study of protein-protein interactions.
Matrix-Assisted Laser Desorption Ionization (MALDI-TOF-MS)
The utilization of Matrix-assisted Laser Desorption Ionization (MALDI-MS) serves as another renowned technique employed in intact protein analysis. This method entails the implementation of a matrix that is co-crystallized with the coveted protein. Upon irradiation of this matrix with a laser, ions emanate from the protein, enabling them to be subsequently analyzed utilizing mass spectrometry. MALDI-MS is deemed a highly potent technique that is capable of conducting high-throughput analyses of intact proteins, while still maintaining compatibility with a vast array of sample types, inclusive of tissue and cell lysates.
Undoubtedly, each MS technique brings to the table its own set of advantages and limitations, and thus the selection of a specific technique is ultimately dependent upon the particular needs of the experiment. Nonetheless, it is unequivocal that the analysis of intact proteins represents an indispensable step in protein characterization. This bedrock of protein-based product assessment is instrumental in ensuring the quality and integrity of these products.
Applications of Intact Mass Analysis
Intact mass analysis is a versatile technique that finds applications in various fields, including pharmaceuticals, biotechnology, and proteomics.
Pharmaceuticals:
Intact mass analysis plays a crucial role in the development and quality control of protein therapeutics. The accurate determination of intact mass and identification of modifications such as glycosylation, oxidation, and deamidation are essential for ensuring the safety and efficacy of protein drugs. Creative Proteomics offers intact mass analysis services for protein drugs that comply with regulatory requirements.
Proteomics:
Intact mass analysis is a valuable tool in proteomics, allowing researchers to identify and characterize proteins. This analysis provides accurate mass measurements that can be used to confirm protein identities and the presence of post-translational modifications. Creative Proteomics offers intact mass analysis services for proteomic research, including top-down proteomics, which involves the analysis of intact proteins rather than peptides.
Other Applications:
Apart from the above-mentioned applications, intact mass analysis also finds utility in other fields. For instance, it can be used to identify proteins in complex mixtures, such as food products, environmental samples, and forensic materials. Additionally, intact mass analysis can be used to study protein-protein interactions and protein-ligand binding events.
Workflow of Intact Mass Analysis
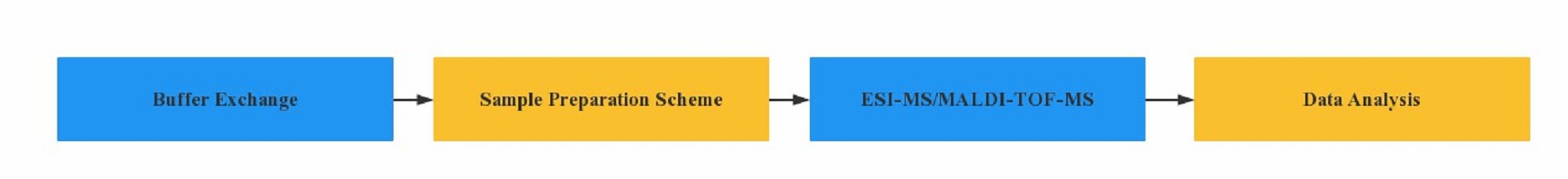
Advantages of Intact Mass Analysis at Creative Proteomics
Creative Proteomics is a leading provider of protein analysis services, offering an exceptional protein intact mass analysis service with several advantages. Here are some reasons why you should choose us for your protein analysis needs:
High-Resolution Mass Spectrometry: At Creative Proteomics, we utilize high-resolution mass spectrometry to provide precise and accurate analysis of intact proteins. Our state-of-the-art technology can detect even subtle differences in protein structure and modifications.
Experienced Scientists: Our team of experienced scientists has a deep understanding of protein analysis and can provide valuable insights into protein structure and modifications. We are constantly expanding our knowledge and expertise in the field, staying up-to-date with the latest technologies and methodologies.
Customized Analysis: We offer customized analysis services to meet specific client needs and requirements. Our team will work with you to design a tailored analysis plan that fits your specific needs, ensuring the best possible results.
Regulatory Compliance: Creative Proteomics complies with regulatory guidelines for the analysis of biopharmaceuticals, ensuring the safety and efficacy of these products. Our team follows strict quality control procedures, ensuring the highest standards of quality and accuracy.






