Chimeric fusion proteins are used as biologic therapeutic agents consisting of a carefully selected and relatively short-lived effector domain (generally peptide) coupled to a 'carrier' (usually protein or peptide) that contributes to the functional properties of the resultant fusion protein. Fusion proteins are genetic engineering products, and linking genes of separate proteins involved to afford novel polypeptides from the incorporated domains together with their functional properties. Examples of biologically active functional proteins include cytokines, toxins, receptors, enzymes, antigenic peptides, etc.
Fusion proteins that the FDA has approved to date can be used as agonists of receptor function, such as romiplostim and alefacept; as antagonists that block receptor function, such as etanercept, belatacept, abatacept, rilonacept, and aflibercept; as direct cytotoxic agents, such as denileukin-diftitox. Substantial potential candidate peptides targeting metabolic, cardiovascular, central and peripheral nervous systems, endocrine, immunological, hematologic, allergic, and other malignant disorders are under early investigation or clinical trials.
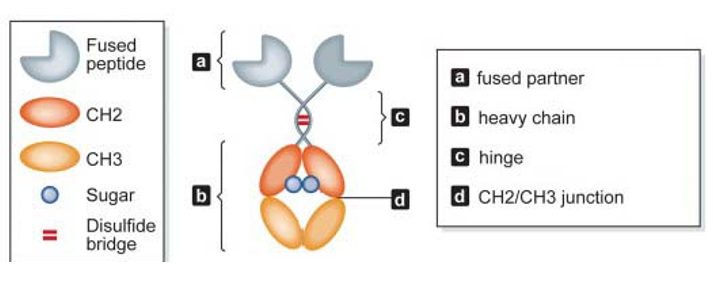 Structure of Fc fusion proteins
Structure of Fc fusion proteinsAn Important Class of Fusion Proteins
Fc fusion proteins are novel proteins produced by fusing the Fc segment of immunoglobulins (IgG, IgA, etc.) with a biologically-active compound using genetic engineering techniques. Fc fusion proteins not only exert biological activities of proteins but also antibody properties, such as resulting in antibody-dependent cell-mediated cytotoxicity (ADCC), complement-dependent cytotoxicity (CDC), and antibody-dependent cell-mediated phagocytosis (ADCP). Fusion of functional proteins with the Fc segment of immunoglobulins can prolong half-life in plasma, reduce immunogenicity, and have important implications in the diagnosis and treatment of diseases.
The development and production process of therapeutic fusion proteins is complicated, and the generation of inevitable impurities may affect therapeutic effects. In order to ensure the production of safe and effective therapeutic fusion proteins, a comprehensive characterization analysis must be performed.
Our Fusion Proteins Characterization Services
By combining advanced technology and an in-depth understanding of biochemistry, Creative Proteomics can perform a wide range of characterization for fusion proteins, such as Fc fusion proteins, applying different analytical techniques.
Our Fusion Proteins Characterization Services Include But Are Not Limited to
High-resolution molecular weight analysis (electrospray mass spectrometry)
Peptide identification analysis (LC-MS/MS)
Peptide sequencing
- N-terminal sequence analysis
- C-terminal sequence analysis
- Sequence coverage
- HPLC peptide mapping
Purity and impurities analysis
- Residual organic solvent determination per USP <467>
- Purity/Potency assessment by HPLC
- Host cell protein analysis
- Purity assessment by capillary electrophoresis (CE, CGE, cIEF)
Others
- Clinical samples preparation/testing for pharmacodynamics and pharmacokinetics studies using multiple reactions monitoring and LC-MS/MS
- Forced degradation studies
Advantages
- Capable of identifying and characterizing target molecules of >0.01%, low-level impurities
- Unique sequence analysis tools enhance additional values
- 100% quality control
Reference
- Brian A. Baldo. Chimeric Fusion Proteins Used for Therapy: Indications, Mechanisms, and Safety. Drug Saf. 2015.






