The application of LC-MS/MS for metabolite identification of biotechnology drugs is a popular research topic. Meanwhile, High-resolution Mass Spectrum (HRMS), a strategy for complete protein quantification, has attracted more and more attention during recent years due to its ability to determine the content of protein drugs and metabolites directly.
The traditional "bottom-up quantitative strategy" of monoclonal antibodies and large-molecular-weight protein drugs requires the use of multiple reaction detection (MRM) scanning modes to determine the target protease after cleavage [1] (Figure 1). Now, an HRMS-based complete protein quantification protocol, also called "Top-down quantification strategy," offers an innovative, high-throughput, and simpler method that allows the direct determination of post-translational modifications of monoclonal antibodies and associated proteins.
In contrast to traditional enzymatic digestion, several pharmaceutical companies have reported in the literature that HRMS-based intact protein quantification strategies provide quantitative sensitivity at the sub-µg/mL level. For example, G0F/G1F glycoforms can be detected in 50 ng/mL rat plasma with a minimum limit of quantification (LOQ) of 100 ng/mL.
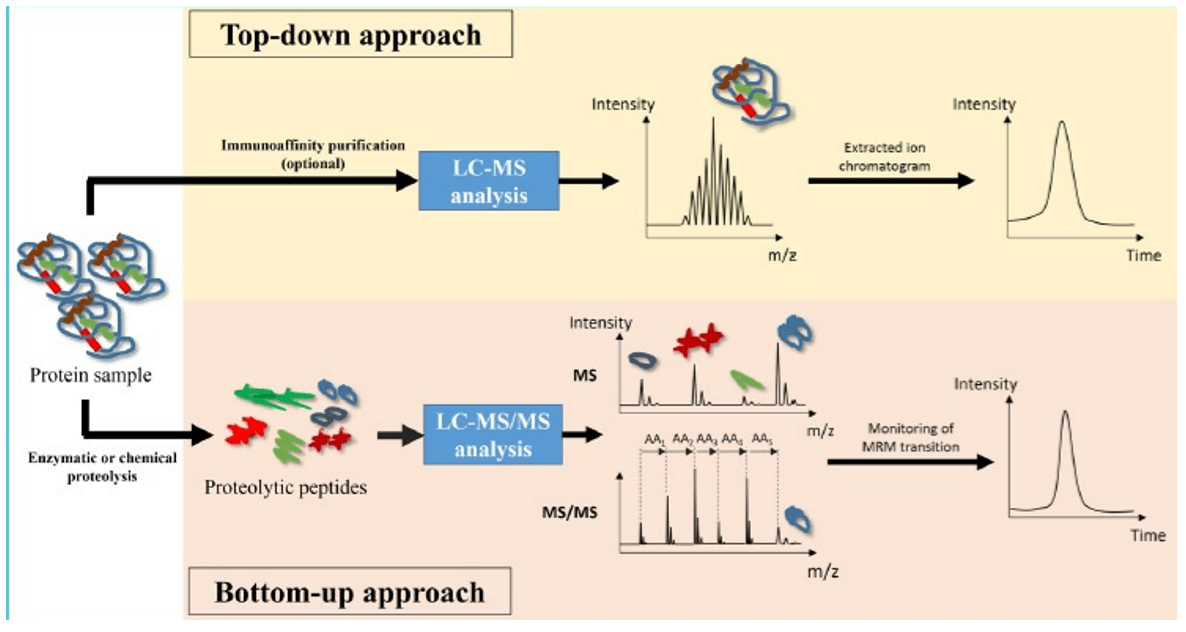
Workflow of top-down and bottom-up approaches in LC-MS-based protein analysis [1]
Our Metabolite Identification Services of Biopharmaceuticals
With our high-resolution mass spectrometer, advanced analytical software, and extensive project experience, we provide an accurate and efficient analysis of the metabolism for your biological drugs in vivo.
Metabolite Identification of Protein/Peptide Drug
After protein/peptide drugs are injected intravenously into the blood or enter cells, non-specific digestion may occur. After digestion, the amino acid sequence may also undergo further amino acid modification and be metabolized by liver P450 enzymes. In vivo metabolite studies of protein/peptide drugs usually focus on non-specific enzymatic cleavage or modification of amino acids, P450 enzyme metabolism, multi-charge distribution, etc.
Metabolites Identification of ADC
During ADC metabolism, cleavage of the linker results in a large antibody fraction, a free linker, and a small molecule drug fraction; While enzymatic cleavage of antibody results in a linker-linked small molecule drug and a small number of amino acid residues. Since the metabolism of small-molecule drugs is directly related to the toxicity of drugs, whether it is "free linkers and small molecule drugs" or "linkers connected to a small number of amino acid residues and small molecule drugs," these two have become hot spots in metabolite research.
Metabolites Identification of monoclonal antibody
In vivo modifications of monoclonal antibodies, such as oxidation, deamination, glutamate/pyroglutamate conversion, and carboxy-terminal lysine shearing, have been reported, while in vivo metabolite studies of monoclonal antibodies focus on the types and sites of amino acid modifications.
Advantages
- Customized analysis strategies: "Top-down", "Middle-up", "Middle-down", " Bottom-up", etc.
- High throughput analysis
- Good linear range, reproducibility and accuracy
References
- Richard Kin Ting Kam. et al. Serum insulin-like growth factor I quantitation by mass spectrometry: insights for protein quantitation with this technology[J]. eJIFCC. 2016, Vol27, No4, pp318-330.
- Xi Qiu. et al. Quantitation of intact monoclonal antibody in biological samples: comparison of different data processing strategies[J]. Bioanalysis. 2018 Jul 1;10(13):1055-1067.
- Yihan Li. et al. Relative Quantitation of Glycoisoforms of Intact Apolipoprotein C3 in Human Plasma by Liquid Chromatography−High-Resolution Mass Spectrometry[J]. Anal. Chem. 2013, 85, 2867−2874.
- Yihan Li1Quantitation of Intact Therapeutic Protein in Plasma Matrix by LC/MS. SCIEX Technical Note.






