The overall three-dimensional shape of an entire protein molecule is the tertiary structure. The protein molecule will bend and twist in such a way as to achieve maximum stability or lowest energy state. Although the three-dimensional shape of a protein may seem irregular and random, it is fashioned by many stabilizing forces due to bonding interactions between the side-chain groups of the amino acids.
Under physiologic conditions, the hydrophobic side-chains of neutral, non-polar amino acids such as phenylalanine or isoleucine tend to be buried on the interior of the protein molecule thereby shielding them from the aqueous medium. The alkyl groups of alanine, valine, leucine and isoleucine often form hydrophobic interactions between one-another, while aromatic groups such as those of phenylalanine and tryosine often stack together. Acidic or basic amino acid side-chains will generally be exposed on the surface of the protein as they are hydrophilic.
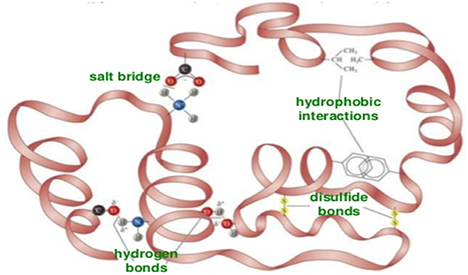
Fig 1. Tertiary Structure
The formation of disulfide bridges by oxidation of the sulfhydryl groups on cysteine is an important aspect of the stabilization of protein tertiary structure, allowing different parts of the protein chain to be held together covalently. Additionally, hydrogen bonds may form between different side-chain groups. As with disulfide bridges, these hydrogen bonds can bring together two parts of a chain that are some distance away in terms of sequence. Salt bridges, ionic interactions between positively and negatively charged sites on amino acid side chains, also help to stabilize the tertiary structure of a protein. Tertiary Structure Analysis services at Creative Proteomics are as below.
- Circular Dichroism Spectra (Near UV)
- Hydrogen Deuterium Exchange Mass Spectrometry (HDX-MS)
- X-ray Crystallography
For more details, please feel free to contact us or send inquiry directly.






