S-palmitoylation (S-PALM) is a post-translational lipid modification because of its reversibility that associated the linkage of a fatty acid chain to cysteine residues of the substrate protein, adjusted membrane-protein and protein-protein interactions in cell signaling, which commands membrane targeting of many proteins underlying fundamental cellular signaling pathways and diseases, for example, cancer and microbial infections. Thence, it can act as a switch equal to phosphorylation or ubiquitination that adjust various crucial physiological pathways within the cell. What should be noticed is that the dynamic turnover of palmitate on proteins at the synapse may offer a crucial mechanism for rapidly changing synaptic strength. Several evidence displays a linkage between severe neurological disorders and impairments in S-PALM. Protein S-PALM can be reckoned by using metabolic labeling methods and biochemical tools. Unfortunately, there exists obvious limitation continues to be the restriction to use them in living cells. It is important for membrane targeting to conclude the spacing of palmitoylated cysteines. But it is still a mystery on how these specific features of S-palmitoylation work for protein sorting. S-prenylation implies to the post-translational installation of either farnesyl (15-carbon) or geranylgeranyl (20-carbon) isoprenoid lipid chains on proteins at one or two cysteine residues located at or near the C-terminus through a thioether linkage.
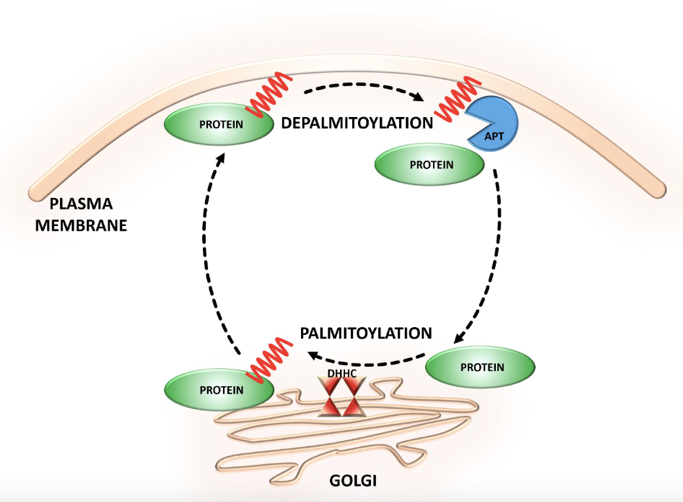 Figure 1: S-palmitoylation
Figure 1: S-palmitoylationRecent research shows that palmitoylation may be crucial for directing proteins, palmitoylation can clear transport vesicles and allows a selection of specific cytosolic proteins to restrict to secretory vesicles. Enough proteins are put into different transport carriers and transported to the specific axonal or dendritic sections. By sorting adaptors and next concentrate, sorting signals conclude the cytoplasmic region of transmembrane proteins, of which are recognized in the appropriate vesicles. Various sorting motifs are submitted in the majority of proteins, for example, tyrosine- or dileucine-based motifs, and interact with different adaptor protein complexes. Besides the secretory vesicles, other palmitoylated proteins also transport palmitoylated SCG10 and N-Ras proteins, for examples, the microtubule associated protein MAP6.
Creative Proteomics has a strict workflow to analyze S-palmitoylation to meet your requirements.






