- Services
- FAQ
- Case Study
- Demo
- Related Services
- Support Documents
- Inquiry
Our AF4 Services for Biopharmaceutical Analysis
Asymmetrical Flow Field-Flow Fractionation (AF4) is a highly versatile, matrix-free separation technique designed for the precise analysis of proteins, peptides, antibodies, viral vectors, and nanoparticle-based formulations. This method offers a broad measurement range, spanning 1 nm to 1 μm, enabling detailed characterization of hydrodynamic size, aggregation states, and molecular weight distributions. AF4 is particularly valuable for orthogonal validation alongside conventional techniques (SEC, or SV-AUC), providing high-resolution data without introducing artifacts from stationary-phase interactions.
Our AF4 services support every stage of biopharmaceutical development, from early research to method validation and analytical support for regulatory or comparability studies. By combining advanced fractionation strategies with multi-detector systems, we provide actionable insights into molecular and particle behavior under physiologically relevant conditions.
What Our AF4 Services Deliver
AF4 offers a unique combination of matrix-free separation, broad size coverage, and compatibility with multiple detection methods. It serves as an orthogonal technique for validating traditional size-based methods and provides high-resolution analysis for complex biological samples.
Protein Aggregation and Stability Profiling
AF4 resolves monomers, oligomers, and higher-order aggregates, enabling precise quantification for stability and degradation studies. Forced degradation and real-time stability analysis allow researchers to monitor changes in protein and peptide formulations under controlled conditions.
Antibody Drug Characterization Services
Our AF4 platform enables detailed assessment of antibody drug candidates, including size variants, fragmentation, and structural heterogeneity. The method supports comparability studies, biosimilarity assessment, and lot-to-lot consistency evaluations.
Viral Vector and Lipid Nanoparticle Analysis
AF4 accurately separates and quantifies empty and full capsid populations in viral vector preparations. For lipid nanoparticles (LNPs) and other nanoformulations, AF4 provides information on size distribution, drug loading efficiency, and release profiles, essential for formulation optimization and quality assessment.
Nanoparticle and nanoformulation profiling
We characterize lipid-based carriers, polymeric nanoparticles, and surface-modified systems (e.g., PEGylated particles)—measuring size distribution, polydispersity, and proxies for drug encapsulation and release using spectroscopic detectors.
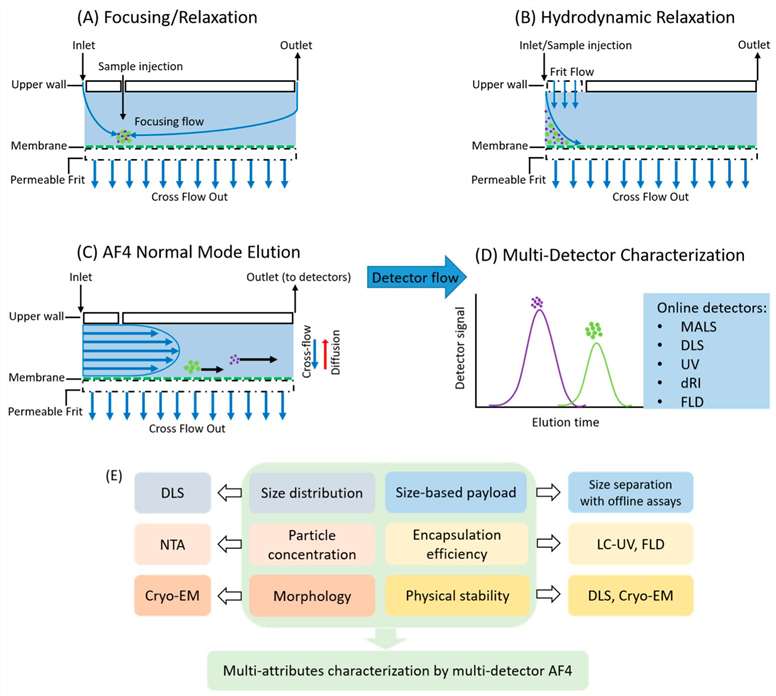 Figure 1. Representative diagram of relaxation and elution process in AF4.
Figure 1. Representative diagram of relaxation and elution process in AF4.Instrumentation and Detection Systems
AF4 instrumentation combines a flow-controlled separation channel with advanced detection systems to ensure high sensitivity, precision, and reproducibility.
AF4 Channel Design and Focused Separation
The channel design, including frit-inlet options, is optimized to minimize focus stress and sample perturbation. AF4 supports sample volumes from 4 µL to 100 µL and allows measurements under controlled temperatures, preserving the integrity of fragile biomolecules.
Detection Systems
AF4 can be coupled with multiple detectors:
- Multi-Angle Light Scattering (MALS) for molecular weight and radius of gyration.
- Dynamic Light Scattering (DLS) for hydrodynamic size confirmation.
- UV, Refractive Index (RI), Fluorescence, and Diode-Array Detectors (DAD/FLD) for concentration, drug encapsulation, and release analysis.
Visualization and standard deliverables
We provide:
- High-resolution chromatograms with aligned detector traces (UV, RI, MALS).
- Tables summarizing numeric outputs and QC metrics.
- Comparative overlays to past runs or orthogonal method outputs.
- Narrative interpretation focusing on development decisions.
Orthogonal strategy — integrating AF4 with multi-method analytics
We embed AF4 within a multi-method analytical plan to increase confidence and satisfy regulatory expectations.
How to Choose AF4 versus other methods
- Use AF4 when sample fragility or potential stationary-phase interactions may bias SEC, or when a broad size range must be covered in one workflow.
- Use SEC for rapid, robust QC monitoring where stationary-phase interactions are acceptable and well characterized.
- Use SV-AUC for absolute sedimentation coefficients and when orthogonality with an independent physical principle is required.
- Choice depends on sample size, fragility, throughput needs, and the specific CQA to monitor.
Integrating AF4 with mass spectrometry and proteomics
For identity or post-translational modification (PTM) analysis, we collect AF4 fractions offline and route them to LC-MS workflows. We recommend sample stabilizers during fraction collection and document fraction handling to preserve analyte integrity.
Advantages of Our AF4 Services
- Wide measurable size range: Enables characterization of particles and macromolecules from approximately 1 nm to 1 µm.
- High-resolution aggregate profiling: Allows clear differentiation of monomers, oligomers, and higher-order aggregates in complex protein formulations.
- Multi-detector compatibility: Can be coupled with detectors such as DLS, UV, fluorescence, and refractive index to obtain molecular weight, particle size, and concentration data simultaneously.
- Orthogonal verification capability: Provides independent confirmation of results obtained from chromatographic techniques such as SEC.
- Flexible method development: Analytical workflows can be tailored for proteins, antibodies, viral capsids, and nanoparticle-based formulations.
Creative Proteomics' AF4 Workflow
- Method optimization: Adjust injection volume, focusing time, and cross-flow gradients for optimal separation (1 nm–1 µm).
- Size-based separation: Perform AF4 fractionation under controlled flow conditions to preserve sample integrity.
- Multi-detector analysis: Use light scattering, refractive index, UV, or fluorescence detectors for size, molecular weight, and concentration measurements.
- Quality control: Conduct recovery checks, system suitability testing, and replicate injections to ensure data reliability.
- Data processing: Analyze chromatographic outputs to generate size distribution, molecular weight, and structural parameters.
Sample Requirements
| Sample Type | Proteins, peptides, antibodies, viral vectors, nanoparticles (e.g., LNPs) |
| Concentration Range | Typically 0.1–10 mg/mL (protein); varies for nanoparticles |
| Injection Volume | 4–100 µL |
| Buffer Composition | Compatible with AF4 membrane (e.g., PBS, low-salt buffers) |
| pH Range | Typically pH 5–8 |
| Temperature Stability | Stable at 18–40°C |
| Storage Conditions | Fresh or properly stored samples (e.g., 2–8°C or frozen) |
Why Choose Creative Proteomics for AF4?
- Extensive industry experience: Decades of expertise in protein, antibody, and nanoparticle characterization ensure scientifically sound AF4 method design.
- Tailored method development: Customized AF4 workflows optimized for specific sample types (proteins, AAVs, LNPs, polymers) and project goals.
- Multi-detector integration: Combined use of MALS, DLS, UV, RI, and fluorescence enables comprehensive size, molecular weight, and concentration analysis.
- Orthogonal validation capability: Seamless integration with SEC, DLS, and SV-AUC for cross-validation and enhanced data confidence.
- High reproducibility and data quality: Strict system suitability testing and quality control ensure consistent and reliable results.
- Fast turnaround time: Efficient workflows deliver high-quality technical reports within project timelines.
FAQ
Q1: What is AF4, and how does it separate particles?
A1: AF4 separates particles in an empty channel by applying a perpendicular cross-flow. Particles establish a diffusion-driven equilibrium: smaller species occupy faster central streamlines and elute earlier than larger species. This is the reverse elution order of SEC.
Q2: When should AF4 be chosen over SEC or SV-AUC?
A2: Choose AF4 when stationary-phase interactions could bias SEC, when a broad size range must be covered in a single method, or when gentle, matrix-free separation better preserves fragile assemblies. SV-AUC remains valuable for absolute sedimentation and when an independent physical principle is required.
Q3: How are empty and full viral capsids distinguished in AF4?
A3: AF4 separates capsid populations based on hydrodynamic size and density-related differences. When combined with MALS and concentration detectors, relative quantitation of different capsid populations can be achieved, often supported by orthogonal assays.
Q4: Can AF4 analyze PEGylated or highly modified proteins?
A4: Yes. AF4 is well-suited for modified proteins, including PEGylated species, because it avoids interactions with stationary phases that may bias chromatographic methods.
Case Study
Case: Proteins and antibodies in serum, plasma, and whole blood—size characterization using asymmetrical flow field-flow fractionation (AF4)
Authors: Maruno T, et al.
Journal: Journal of Pharmaceutical Sciences
Year: 2021
DOI: 10.1016/j.xphs.2021.06.031
Background
Protein aggregation is a critical concern in biopharmaceutical development, as it can affect efficacy and safety. While aggregation is typically assessed in formulated products, understanding protein behavior in blood after administration remains challenging due to the complexity of biological matrices. Traditional methods, such as SEC, may introduce artifacts, and extensive sample preparation can alter native protein states. Therefore, a method capable of analyzing proteins in blood under near-physiological conditions is highly desirable.
Methods
AF4 was performed using a multi-detector setup including UV, fluorescence, multi-angle light scattering (MALS), and differential refractive index (dRI). Key methodological features include:
- Minimal preprocessing: no centrifugation or filtration
- Detection strategy: UV/dRI/MALS for general protein profiling. Fluorescence detection for selective monitoring of FITC-labeled antibodies
- Validation standards: reference proteins (myoglobin, BSA, IgG)
- Separation conditions: controlled cross-flow AF4 with physiological buffer conditions
Results
- AF4 successfully separated major proteins (e.g., albumin, IgG) in serum and plasma.
- Plasma showed additional components compared to serum.
- The labeled antibody exhibited altered size behavior in plasma, indicating interactions and increased dimer formation.
- Whole blood could be analyzed with minimal preprocessing, though large components introduced some complexity.
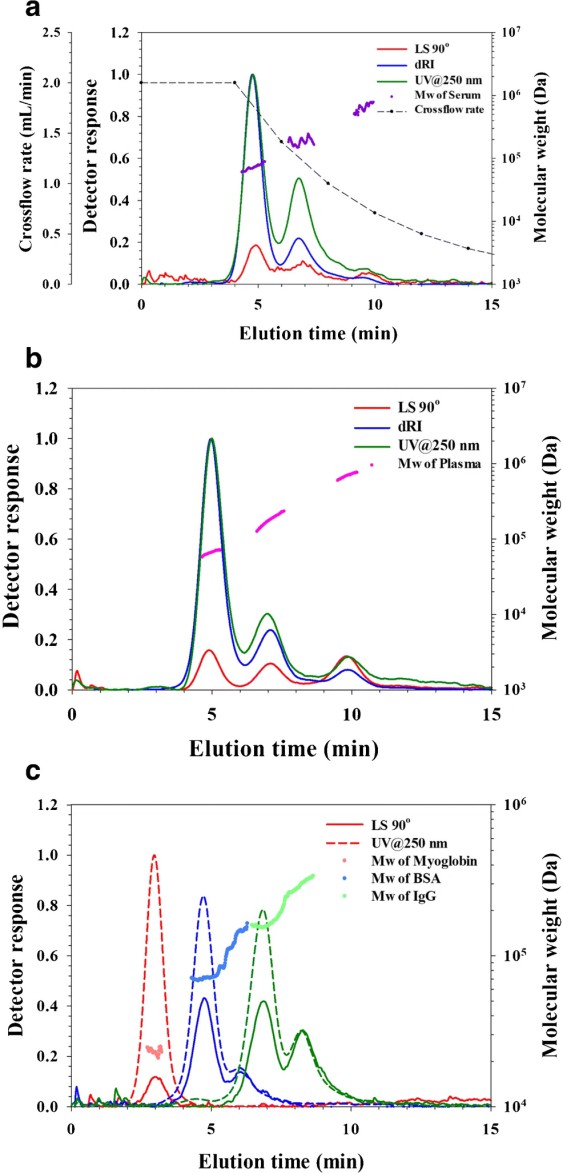 Figure 2. AF4 separation of different-sized spherical bacteriophages.
Figure 2. AF4 separation of different-sized spherical bacteriophages.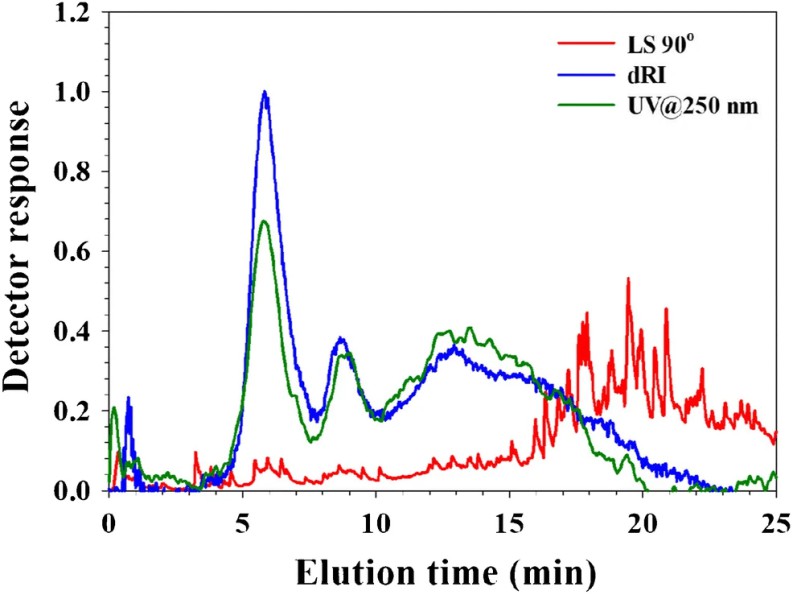 Figure 3. Analysis of whole blood by AF4-UV-MALS-dRI.
Figure 3. Analysis of whole blood by AF4-UV-MALS-dRI.Conclusion
AF4 enables direct, high-resolution protein size analysis in complex biological matrices under near-native conditions. It is a powerful orthogonal tool for studying protein aggregation and interactions in blood.
Demo
Demo: Asymmetrical Flow Field-Flow Fractionation on Virus and Virus-Like Particle Applications
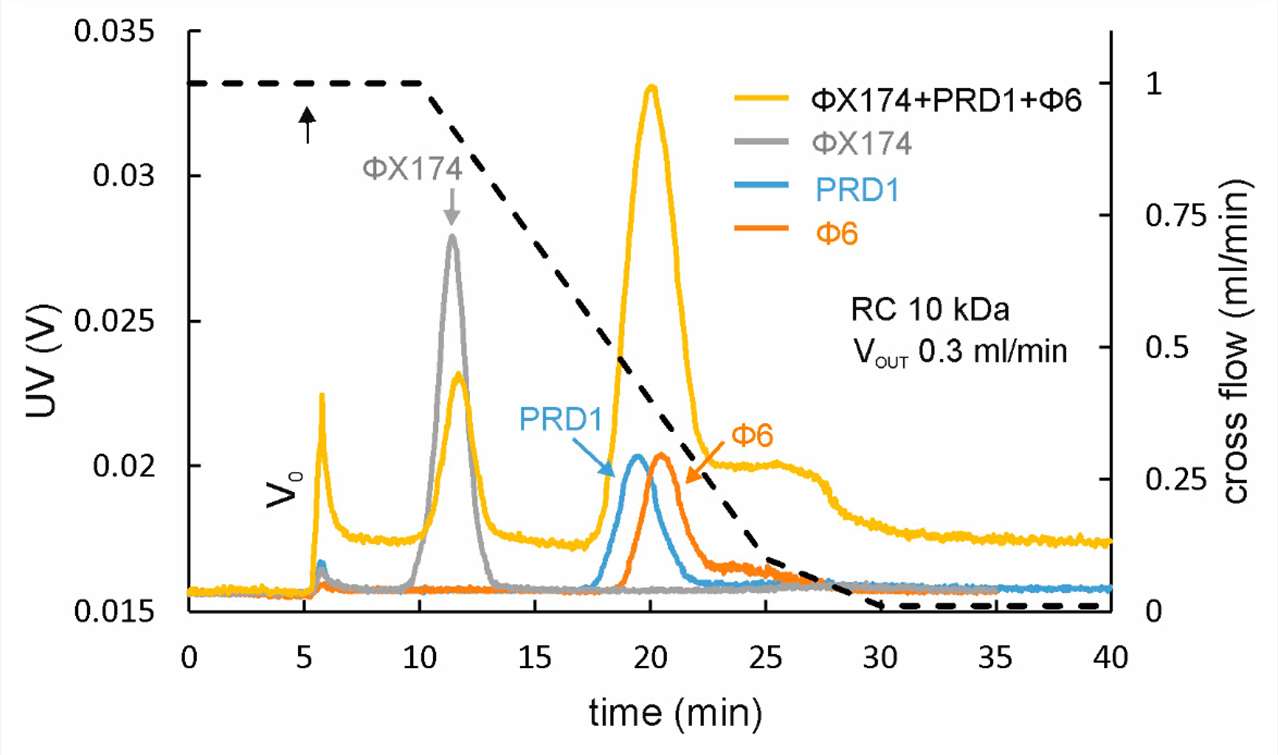 Figure 4. AF4 separation of different-sized spherical bacteriophages.
Figure 4. AF4 separation of different-sized spherical bacteriophages.References
- Schneider C M, Haffke D, Cölfen H. Band sedimentation experiment in analytical ultracentrifugation revisited. Analytical chemistry, 2018, 90(18): 10659-10663. DOI: 10.1021/acs.analchem.8b02768.
- Leeman M, et al. Proteins and antibodies in serum, plasma, and whole blood—size characterization using asymmetrical flow field-flow fractionation (AF4). Analytical and bioanalytical chemistry, 2018, 410(20): 4867-4873. DOI: 10.3390/molecules28104169
- Maruno T, et al. Comprehensive size distribution and composition analysis of adeno-associated virus vector by multiwavelength sedimentation velocity analytical ultracentrifugation. Journal of Pharmaceutical Sciences, 2021, 110(10): 3375-3384. DOI: 10.1016/j.xphs.2021.06.031
- Eskelin K, Poranen M M, Oksanen H M. Asymmetrical flow field-flow fractionation on virus and virus-like particle applications. Microorganisms, 2019, 7(11): 555. DOI: 10.3390/microorganisms7110555
Related Services
Support Documents
KNOWLEDGE CENTER