As the global fervor for protein drug development continues to rise, Capillary Electrophoresis (CE), an emerging analytical technique, has garnered attention and recognition from the biopharmaceutical industry and regulatory bodies. Presently, a majority of global biopharmaceutical companies have incorporated capillary electrophoresis systems into the research and quality control analysis of protein drugs. From optimizing culture media, clone selection, studying formulation stability, monitoring purification processes, to protein characterization, impurity detection, and quality control of protein drug products, every stage of protein drug development relies significantly on the support of capillary electrophoresis technology.
For instance, in protein purity determination, there has been a shift from SDS-PAGE to Sodium Dodecyl Sulfate Capillary Electrophoresis (CE-SDS). Concerning the determination of protein isoelectric points, Capillary Isoelectric Focusing (CIEF) provides more accurate results than traditional gel-based methods. Additionally, for the analysis of glycoprotein drug glycosylation heterogeneity, capillary electrophoresis has become one of the high-resolution analytical techniques. The broad application spectrum of capillary electrophoresis underscores its pivotal role in advancing various facets of protein drug development and quality control.
In the analysis of monoclonal antibody drugs, research findings indicate that, in comparison to traditional SDS-PAGE techniques, Capillary Electrophoresis with Sodium Dodecyl Sulfate (CE-SDS) is more suitable for the characterization and quality control of monoclonal antibody drugs. The stability and reliability of results achieved through CE-SDS far surpass those obtained through SDS-PAGE. Consequently, it is recommended for biopharmaceutical companies to replace SDS-PAGE with CE-SDS as the platform for research and quality analysis.
In 2013, the United States Pharmacopeia (USP) issued guidelines specifically addressing the use of capillary electrophoresis (CE) for purity determination, isoelectric point/charge heterogeneity analysis, and glycosylation analysis of monoclonal antibody drugs such as Rituximab and Trastuzumab. These regulations provide explicit recognition of the pivotal role played by CE in the analysis of size variants, charge variants, identification, consistency, and glycosylation modifications of monoclonal antibody drugs.
Select Service
- Isoelectric Point Determination with cIEF
- Isoelectric Point
- Protein Isoform Analysis (Capillary Isoelectric Focusing)
- Charge heterogeneity analysis
- Glycosylation analysis
- Protein Purity Analysis
- Antibody Drug Characterization
- Monoclonal Antibody Characterization
- Antibody Drug Conjugate Characterization
Purity and Size Heterogeneity Analysis of Monoclonal Antibody Drugs
In the realm of monoclonal antibody (mAb) drug purity analysis, traditional SDS-PAGE methods have become inadequate for the evolving demands of biopharmaceutical research and quality control in terms of resolution, quantitative accuracy, and automation. In contrast, capillary electrophoresis with sodium dodecyl sulfate (CE-SDS) methods, leveraging the separation based on differences in protein molecular weight, prove suitable for purity analysis of both reduced and non-reduced forms of mAb drugs. This method eliminates intricate manual procedures, ensuring more accurate quantification, heightened resolution, and, particularly in the reduced mode, precise separation and quantification of non-glycosylated heavy chains.
![Figure 1. Purity analysis of reduced monoclonal antibody drugs by CE-SDS[1]](upload/image/application-of-capillary-electrophoresis-in-monoclonal-antibody-drugs-analysis-1.jpg) Figure 1. Purity analysis of reduced monoclonal antibody drugs by CE-SDS[1]
Figure 1. Purity analysis of reduced monoclonal antibody drugs by CE-SDS[1]Tailoring capillary length allows for both high-resolution and fast-mode purity analyses. In high-resolution mode, Capillary Electrophoresis with Sodium Dodecyl Sulfate (CE-SDS) coupled with polymer dispersant offers optimal resolution. Conversely, in the fast mode, the CE-SDS method reduces wash and separation times, consequently enhancing analysis throughput.
Moreover, the combination of Laser-Induced Fluorescence Detection (CE-SDS-LIF) with CE-SDS, employing labeling with 5-Tarma or FQ dyes for protein marking, enhances sensitivity. This synergistic approach allows accurate detection even with impurity fragments as low as 0.01%. Simultaneously, the use of LIF detectors minimizes baseline fluctuations, ensuring more precise integration and quantification.
Isoelectric Points and Charge Heterogeneity Analysis of Monoclonal Antibody Drugs
Monoclonal antibody (mAb) drugs undergo post-translational modifications such as glycosylation, deamidation, isomerization, and oxidation, leading to alterations in protein surface charges and subsequent charge heterogeneity. Each variant possesses distinct isoelectric points. Capillary isoelectric focusing (cIEF) based on isoelectric points enables high-resolution separation and quantification of mAb variants, with a separation difference as low as 0.03 pI units. This method utilizes an isoelectric point marker to construct calibration curves, facilitating precise determination of the isoelectric points of mAb variants. It has become a crucial approach for isoelectric point determination and charge heterogeneity analysis of mAb drugs.
![Figure 2. Analysis of isoelectric point and charge heterogeneity of monoclonal antibody drugs by CIEF[5]](upload/image/application-of-capillary-electrophoresis-in-monoclonal-antibody-drugs-analysis-2.jpg) Figure 2. Analysis of isoelectric point and charge heterogeneity of monoclonal antibody drugs by CIEF[5]
Figure 2. Analysis of isoelectric point and charge heterogeneity of monoclonal antibody drugs by CIEF[5]For protein samples with diverse pI ranges, the selection of appropriate amphoteric electrolytes is crucial for achieving high-resolution analysis. For instance, considering that the pI values of most monoclonal antibodies fall within the range of 7-10, amphoteric electrolytes with a pH range of 3-10 are suitable for optimal resolution. When dealing with protein samples with a pI ranging from 5-7, a narrower pH range of 5-8 for the amphoteric electrolyte is recommended. In the case of acidic proteins with a pI below 5, enhanced analysis results can be achieved through reverse focusing and migration modes. This strategic choice in electrolytes ensures the refinement of protein isoelectric focusing, contributing to improved analytical outcomes.
Fast Analysis of Charge Heterogeneity in Monoclonal Antibody Drugs using CZE Method
Capillary zone electrophoresis (CZE), a separation technique based on the proportion of charge to volume for analytes, stands out in capillary electrophoresis due to its simplicity and speed. In the CZE separation mode, where the molecular volumes of various monoclonal antibody (mAb) drug variants are closely matched, the separation of charge variants relies predominantly on differences in surface charge. This principle aligns with the separation mechanism of variants in capillary isoelectric focusing (CIEF) mode. Consequently, CZE is considered a preferred method for the rapid analysis of charge heterogeneity in the biopharmaceutical industry. Moreover, owing to the simplicity and rapidity of CZE, it holds significant value in the realm of distinctive analysis of mAb drugs.
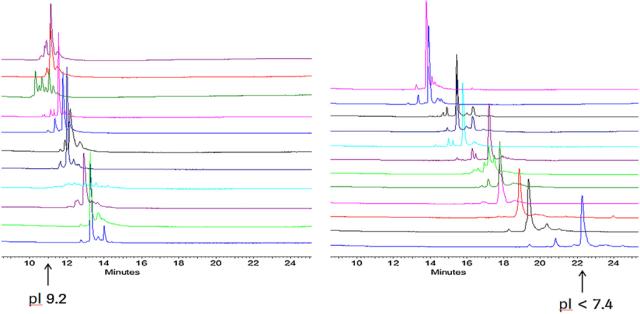 Figure 3. Charge heterogeneity analysis of 23 monoclonal antibody drugs using the same CZE method [3]
Figure 3. Charge heterogeneity analysis of 23 monoclonal antibody drugs using the same CZE method [3]Analysis of Glycosylation Heterogeneity in Monoclonal Antibody Drugs
In monoclonal antibody (mAb) drugs, such as glycoprotein pharmaceuticals, the type and arrangement of glycans contribute to glycosylation heterogeneity. Glycosylation modifications significantly impact the safety and efficacy of mAb drugs, making the quality control of glycosylation heterogeneity crucial. The process of using capillary electrophoresis (CE) technology for glycosylation heterogeneity analysis involves the release of glycans from glycoproteins, glycan labeling, and capillary electrophoresis separation.
By employing magnetic bead-assisted glycan release and labeling, the preprocessing time is reduced to 1 hour, accelerating the analysis process. The use of 8-aminopyrene-1,3,6-trisulfonic acid (APTS) as a fluorescence label not only enhances separation efficiency by increasing charge but also achieves high sensitivity glycan analysis through laser-induced fluorescence (LIF) detection. The advantages of capillary electrophoresis in glycan analysis lie in its high resolution and rapid speed. It can distinguish glycan differences, allowing the separation of glycan isomers with similar molecular weights in a process that takes only 5-20 minutes.
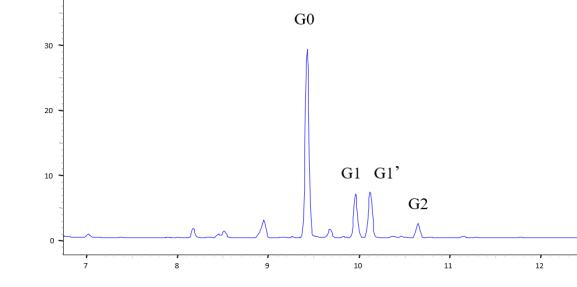 Figure 4. Monoclonal antibody glycosylation analysis using CE-LIF method.
Figure 4. Monoclonal antibody glycosylation analysis using CE-LIF method.References
- Brian Nunnally et al., A Series of Collaborations between Various Pharmaceutical Companies and Regulatory Authorities Concerning the Analysis of Biomolecules Using Capillary Electrophoresis, 2007, 66, 955-961.
- Oscar Salas-Solano et al., Intercompany Study to Evaluate the Robustness of Capillary Isoelectric Focusing Technology for the Analysis of Monoclonal Antibodies, Chromatographia, 2011, 73, 1137-1133.
- Bernd Moritz et al., Evaluation of Capillary Zone Electrophoresis for Charge Heterogeneity Testing of Monoclonal Antibodies, Journal of Chromatography B, 2015, 983, 101-110.
- Ákos Szekrényes et al., Multi-Site N-glycan Mapping Study 1: Capillary Electrophoresis – Laser Induced Fluorescence, 2016, 8,56-64.
- Application Guide: Capillary Isoelectric Focusing (cIEF) Analysis, Sciex.