What Is Capillary Electrophoresis
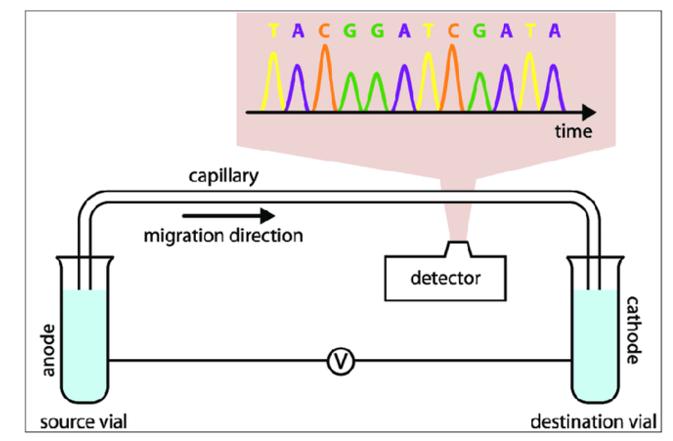
Capillary electrophoresis (CE), also known as high-performance capillary electrophoresis (HPCE), represents a class of liquid phase separation techniques. This method leverages high voltage direct current electric fields as the driving force and capillaries as the separation channels. It achieves separation based on the differences in migration rates and partitioning behaviors among various components within a sample. Notably, CE advanced analytical science from the microliter level to the nanoliter level, and further facilitated cellular analysis, even single-molecule analysis. Subsequent to high-performance liquid chromatography, CE has emerged as another significant advancement in the field of analytical science.
 Capillary Electrophoresis
Capillary ElectrophoresisDevelopment of cIEF
The advent of cIEF modality Since the 1980s, Capillary Electrophoresis (CE) technology has introduced a variety of separation modalities, including CZE, CGE, and MEKC. In 1985, Hjertén and Zhu, for the first time, realized Capillary Isoelectric Focusing (cIEF) on capillary electrophoresis instruments. Compared to slab-gel IEF, cIEF has the advantages of high automation and quantifiability.
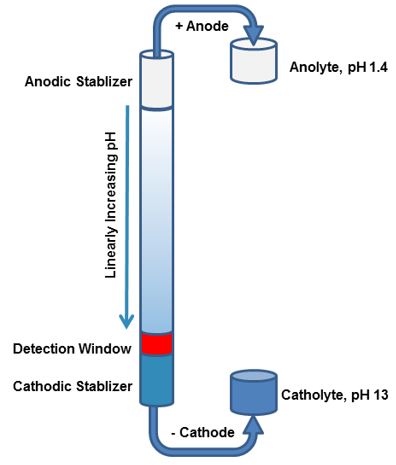
Capillary isoelectric focusing (cIEF) is significant for assessing charge heterogeneity attributes of protein therapeutics; however, after years of development, cIEF has not been widely acknowledged within the pharmaceutical industry. The primary challenge lies within the commercial capillary electrophoresis instruments. As since they are only equipped with single-point ultraviolet absorbance and fluorescence detectors, running cIEF methods can prove to be difficult. Post-separation, proteins are relocated using chemical and pressure methods, which range past single-point detectors. However, this relocation protocol may induce peak distortions, smearing or poor resolution and extend running times, all aggravating further method optimization. Furthermore, the internal proceedings of focusing and mobilization steps within the separation column remain absent from observation, resulting in a challenging optimization process.
Key Advantages of icIEF
In 1992 and 1994, scientists Wu and Pawliszyn from ProteinSimple first proposed the whole column imaging detection technology based on refractive index and UV absorption. They commercially launched cIEF with full column imaging UV absorption for the first time in 1998, calling it icIEF. The advent of icIEF somewhat compensates for the deficiencies in traditional capillary isoelectric focusing electrophoresis.
Capillary isoelectric focusing, otherwise known as icIEF, stands to offer numerous advantages, including simplification of method development, high throughput, reliable isoelectric point (pI) quantification, and sound reproducibility. This obviates the need for the traditional clEF migration step thereby reducing the analysis duration from 40 to 60 minutes to a mere 5 to 15 minutes per sample. Additionally, icIEF permits real-time monitoring of the entire focusing process, facilitating an effortless identification of causes for non-reproducible peaks, such as protein aggregation. This problem can be mitigated by optimizing the focusing time or by the addition of a stabilizer. Following its successful commercialization, icIEF gradually superseded traditional cIEF and due to its superior performance features, has been extensively utilized in the biopharmaceutical industry. This method is also now included in the 2020 edition of the Pharmacopoeia.
In the current realm of biopharmaceuticals, the characterization of charge variants is primarily executed through chromatographic separation coupled with fraction collection, desalting, and lyophilization before analysis by mass spectrometry. However, this approach falls short, particularly for proteins apart from monoclonal antibodies, mainly due to the low resolution of chromatographic separation and the method's complexity. In routine development, researchers often confront the predicament where liquid chromatography (LC) chromatograms and icIEF spectra are not in alignment. As such, there is an urgent need for more sophisticated technologies to meet these requirements.
Outlook on Capillary Electrophoresis
Since its inception in 1981, capillary electrophoresis has progressively evolved into a mature and reliable separation technique. Compared with traditional separation techniques such as gel electrophoresis and liquid chromatography, capillary electrophoresis boasts a simpler setup, smaller volume, faster separation speed, higher resolution and efficiency, as well as lower consumption of samples and solvents. As such, capillary electrophoresis has found widespread applications in diverse fields including biopharmaceutical development, medical research, forensic investigations, environmental monitoring, and food quality inspection.
Currently, within the realm of biopharmaceutics, there is a discernible surge in the prominence of monoclonal antibodies, large-molecule proteins, small molecule drugs, Proteolysis Targeting Chimera (PROTAC), and nucleic acid cell gene therapy. Capillary electrophoresis, recognized as an indispensable tool for characterizing protein drugs, is now entering a phase characterized by substantial advancements. The recent milestone achievement, involving the inclusion of the Imaged Capillary Isoelectric Focusing (icIEF) method in pharmacopeias, positions capillary electrophoresis for increased integration into the research and production workflows of numerous biopharmaceutical enterprises.
Reference
- Coşkun, Ö., & Öztopuz, Ö. Electrophoresis Applications Used in Medicine. Medical Sciences, 15(1), 12-25.