Glycosylation play an essential role in the stability, half-life, safety, and biological activity of antibody drugs. Different production processes can lead to variations in molecular glycoforms, which highlights the significance of accurately and promptly acquiring information about the N-glycan profile during the early stages of process development. Such information noticeably advances the development of cell lines and upstream processes.
N-glycan profile analysis strategies
In the realm of N-glycan profile analysis, there are currently three primary analytical strategies: the analysis at the level of free glycan, glycopeptide and molecular weight (Figure 1). The free glycan level analysis of N-glycan profiling involves steps such as enzymatic cleavage, derivatizing reagent labeling, and liquid or mass spectrometry analysis. The conventional methods for free glycan level N-glycan analysis mainly include the 2AB-UPLC and 2AA-UPLC methods.
Beyond analysis at the level of free glycan, the use of mass spectrometric technology to perform analysis at the glycopeptide or molecular weight level is an efficient strategy.
Taking monoclonal antibodies as an example, considering that monoclonal antibodies typically have only one glycosylation site in their heavy chain CH2 region, information about the protein's N-glycosylation type and glycoform abundance can be obtained through molecular weight analysis at the intact protein or subunit level.
In detail, the types of glycosylation can be determined by resolving the mass-to-charge ratio of ion clusters, while information about glycoform abundance is proportional to the corresponding ion cluster signal intensity. The smaller the glycosylated site in the subunit, the lower the complexity of the resulting mass spectrum. This aids the resolution of lower abundance glycoforms, thereby making the glycoform analysis results closer to reality.
You may interested in
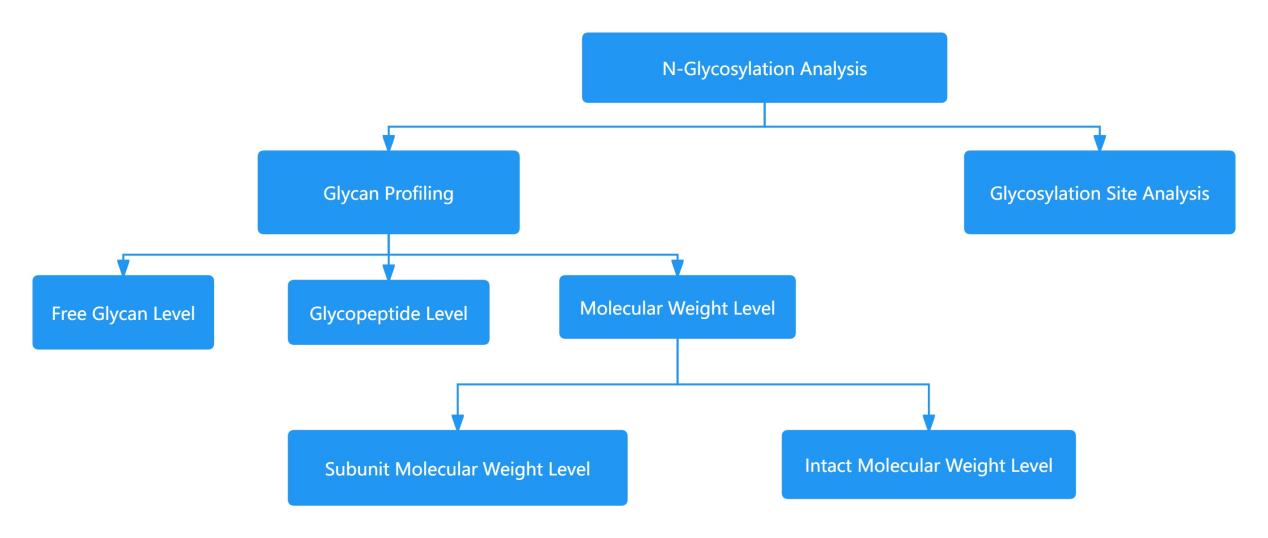 Figure 1 Schematic diagram of antibody N-glycan characterization analysis strategy
Figure 1 Schematic diagram of antibody N-glycan characterization analysis strategyFree Glycans Rapid Analysis Strategy of N-Glycans
Traditional N-glycan analysis involves enzymatic cleavage of the sugar chains via N-glycosidase, followed by 2-AA or 2-AB derivatization, and concludes with high-performance liquid chromatography (HPLC). Two key phases within this scheme are the enzymatic cleavage of N-glycans and the tagging with derivatization reagents; the efficacy of these steps decisively influences the pace of the entire analysis.
Recognizing this, several leading manufacturers have launched quick N-glycan analysis kits offering commercially viable solutions for rapid N-glycosidase cleavage and derivatization agent tagging. These kits have powerfully advanced the capabilities for speedy analysis of N-glycans at free sugar levels.
Rapid Analysis Strategy for Subunit of N-glycans
To acquire subunit fragments with glycosylation sites, the common approach is the reduction of intact antibodies leading to the disconnection of the heavy chain (HC) and light chain (LC). Detailed analysis of the mass spectrum for the heavy chain (HC) facilitates the acquisition of glycomic information at the HC level. Further enzymatic cleavage of the heavy chain using the tool enzyme IdeS allows for the procurement of smaller subunit fragments (scFc) that include the glycosylation sites. Through the analysis of the mass spectra of these scFc fragments, more accurate N-glycan glycomic information can be obtained.
During the early stage of macromolecule process development, the need for N-glycan glycosylate detection stems primarily from cell line development and upstream culture processes. Particularly during the cell line development process, where the amount of provided detection samples is limited and may diminish further after the Protein A chromatography purification step.
To meet this detection requirement, Creative Proteomics has developed a new N-glycan analysis method based on CE-MS, which eliminates the need for Protein A purification step. Instead, it applies direct enzyme cleavage and MS sample injection to cell culture. Prior to sample loading, a specific dilution buffer is utilized for high-ratio dilution to diminish the interference from various HCPs, culture medium components, and salts on mass spectrometry. This method simplifies the sample pretreatment process, enabling large-scale operations easily. Additionally, this method allows for rapid separation and detection of samples, hence achieving high-throughput detection of N-glycan glycoforms.
Currently, this method has been applied for cell line screening and effectively promoted the acceleration of cell line development processes. However, due to sensitivity constraints, some low-abundance glycoforms may not be detected. In future work, we intend to further optimize this method.
Rapid Analytical Strategy for Glycopeptide Mapping of N-Glycans
Through rigorous technological research and development, many industry-leading corporations have proposed mature solutions for N-glycan detection at the glycopeptide level. Similarly, Creative Proteomics has developed a "Glycopeptide Mapping". Our strategy can efficiently complete protein denaturation, reduction, alkylation, and high temperature enzyme digestion within 1.5 hours. It is combined with a quick LC-MS detection method, where glycopeptide retrieval analysis is automatically completed via a mass spectrometry workstation. This technique effectively prevents the decleavage of N-glycans, while facilitating the accurate analysis of low-abundance glycan types, compared to the traditional 2AB method. The method has been successfully applied for the final confirmation of the glycan type of monoclonal antibody molecules, as well as for glycan analysis in upstream cell culture process samples.
For N-glycans less than 1%, conventional methods might face difficulties in resolution. In the quest to attain the sensitivity level of the classical 2AB method, analysis at the glycopeptide level is required; here, our glycopeptide level N-glycan rapid analysis strategy provides an excellent choice. Moreover, for dual-antibody or fusion proteins with multiple N-glycosylation sites, the complexity of their glycan modification might render mass spectral analysis challenging. In such situations, utilizing our "Glycopeptide Mapping Scheme" can accurately obtain glycan spectra information for each glycosylation site, potentially resolving the presence of O-glycan spectra information.
Summary
In summation, the 2AB-UPLC method, as a universal approach for determining N-glycan at free glycan levels, has been recognized as the gold standard in N-glycan profiling due to its broad applicability and high accuracy. Concurrently, our N-glycan analysis strategy, characterized by low sample consumption and simplified pretreatments, is an ideal choice for cell culture analysis. Particularly during cell line development, this strategy can swiftly meet the demands of sample testing. Furthermore, our unique glycopeptide mapping scheme excels in precisely analyzing low abundance glycotopes. Not only can this strategy provide information on N/O-glycosylation, but it can also resolve specific situations at various glycosylation sites, demonstrating enormous potential for application. For pharmaceutical companies with existing LC-MS configurations, this is undeniably an excellent choice.
As Creative Proteomics, we possess these three levels of rapid N-glycan characterization strategies. We are committed to offering tailored N-glycan analysis methods aligned with the specific project requirements of our clients, ensuring we deliver high-quality and cost-effective CDMO services.