Host cell protein (HCP) is a protein produced or encoded by a host cell for the production of recombinant therapeutic proteins. Recombinant therapeutic proteins are usually produced by genetically modified prokaryotic or eukaryotic host cells using cell culture or fermentation techniques. Genetic engineering enables host cells to be transformed to selectively express the target protein. In the process of recombinant protein production, host cells also jointly produce proteins essential for normal cell functions, such as cell growth, proliferation, survival, gene transcription, protein synthesis, and so on. Due to cell apoptosis, death, and lysis, other non-essential proteins can also be released into the cell culture medium.
As a foreign protein, although residual HCP in biological products is low in content, it may still trigger the body's immune response to varying degrees, and ultimately lead to allergic reactions or other adverse reactions. HCP constitutes the main part of process-related impurities in the production process of biological agents. The amount of residual HCP in a drug is usually considered a critical quality attribute (CQA) because it may affect product safety and efficacy. Therefore, one of the regulatory requirements is to monitor the removal of HCP in biopharmaceuticals during the development of bioprocesses.
Method of Host Cell Protein Analysis
ELISA
Enzyme-Linked ImmunoSorbent Assay (ELISA) is the most widely used HCP detection method, but this technique has limitations. The ELISA method is only semi-quantitative, does not guarantee the detection of every potential HCP impurity, and cannot determine whether it is multiple HCP residues or a high concentration residue of HCP. At the same time, it takes a long time and high cost to develop a dedicated ELISA kit for a specific process. It is not suitable for the comprehensive evaluation of biopharmaceutical products made with different cell types or purification schemes. In order to solve these problems, LC-MS/MS came into the field as an orthogonal and complementary means of the ELISA method, mainly used for the identification and quantification of trace HCP.
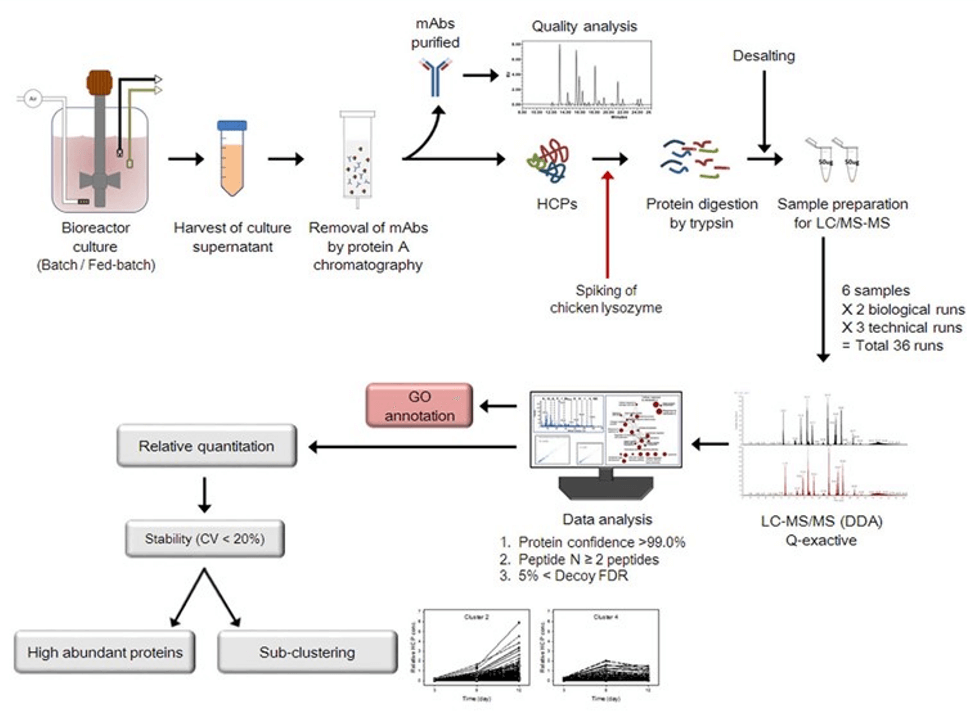
LC-MS/MS
When using LC-MS/MS for HCP detection, there are two main detection strategies according to different research purposes:
1. Target HCP determination: Applicable to the quality control of biological drugs whose production process has been determined. The MRM or high-resolution MRM (MRMHR) scanning mode of conventional resolution mass spectrometry is usually used to obtain sub-µg/mL or µg/mL sensitivity.
2. Unknown HCP determination: In process research and development, in order to design and optimize the purification process, it is necessary to conduct a high-throughput and stable evaluation of HCP in different purification processes or steps. Since these processes are often carried out through a specific combination or reaction, the residual HCP is often unknown. Therefore, in this type of research, a High-Resolution Mass Spectrometer (HRMS) is widely used. The selection of high-resolution mass spectrometry data depends on the acquisition mode, and thousands of proteins can be identified. The SWATH acquisition technology, with the sensitivity reaching 10μg/mL, can then be used for relative or absolute sample quantification. The reproducibility is good, the method is simple, and the measurement throughput is high.