Since antibody-drug conjugates (ADCs) have the molecular characteristics of both small molecule and macromolecule therapeutic drugs, when choosing analytical methods, the typical ones used for both should all be considered. There are many different molecules in ADCs, and the difference between these molecules lies in the drug-to-antibody ratio (DAR) value and/or the site where the antibody binds to the drug. In addition, ADCs prepared by the current production process usually contain non-ADC forms, including naked antibodies and free small molecule drugs. When the ADC molecule enters the body, the conjugated drug is released from the ADC over time during the circulation process, resulting in a change in DAR, and its effectiveness and safety may also be affected. For the above-mentioned different types of analytes, it is necessary to select different analytical methods to complete the detection according to the different purposes of drug development and evaluation and the characteristics of the test substance.
Drug-to-antibody ratio (DAR) analysis
With the dissociation of small-molecule drugs or the different DAR values of ADC in the body, the ADC clearance rate is different, and the DAR value distribution is also changing. Studying the distribution of DAR values helps to understand the overall fate of ADCs and provides a theoretical basis for ADC biotransformation in vivo. UV/VIS spectroscopy is widely used to determine antibody concentration and DAR value, but this method is a buffer system and cannot be applied to complex biological systems such as plasma/serum.
Scientists have developed two methods that can be used to determine the distribution of DAR values in plasma/serum samples: affinity capture LC-MS method and affinity capture hydrophobic interaction chromatography (HIC). Both methods can detect the complete molecular weight.
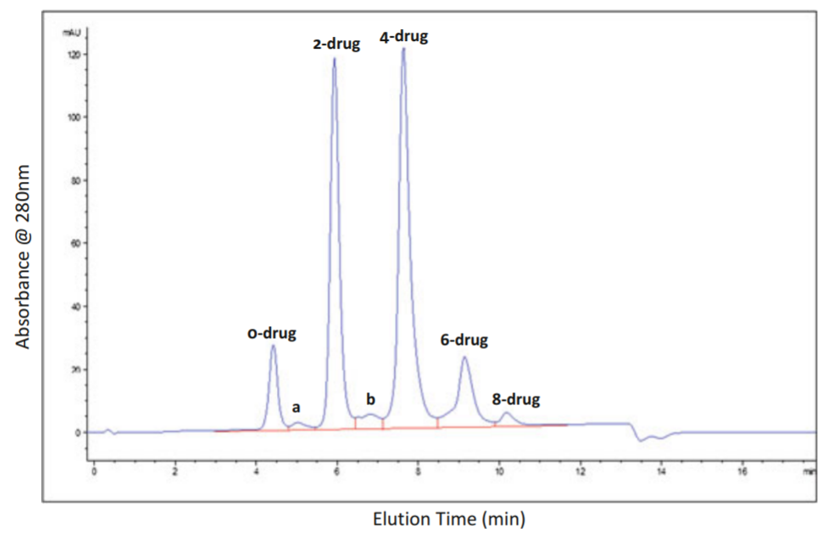
HIC chromatogram (Basa et al., 2013)
Total antibody analysis
Total antibody analysis measures all antibodies in the sample, including bound antibodies (DAR≥1) and unbound antibodies (DAR = 0), which can be used to assess whether the ADC meets the general PK characteristics of monoclonal antibodies. A rapidly cleared ADC, if shown by the total antibody analysis, indicates its ineffective binding to the target and is not suitable for later development.
ELISA is traditionally used for total antibody analysis. For non-clinical PK and TK experiments, specific reagents are difficult to obtain, and general analytical methods are usually used. The capture reagent and detection reagent are both anti-human IgG antibodies.
In recent years, an affinity capture LC-MS/MS method has been developed for detecting total antibodies. It combines the characteristics of ELISA and LC-MS/ MS. First, affinity enrichment of antibodies is achieved by capturing reagents (such as protein A, recombinant antigen, anti-ID antibody, etc.). After enzymatic hydrolysis, the specific peptide is determined by LC-MS/MS for quantitative analysis. A similar affinity capture LC-MS/MS method has been used for the quantification of multiple monoclonal antibody drugs.
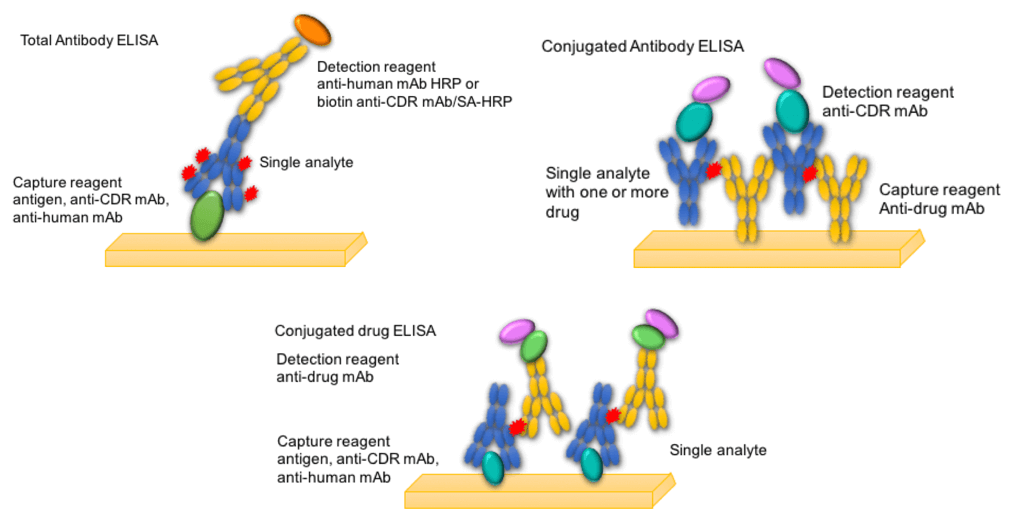
In addition to total antibody analysis, pharmacokinetic studies are also needed to monitor a variety of analytes, including conjugated-antibody, antibody-conjugated drug, nonconjugated drug, and toxin-related metabolites.
Using ELISA and LC-MS/MS technology platforms, the above analytes can be quantitatively analyzed. For antibody conjugates, labeled avidin biotin (LAB) technology is often used. For bound small molecule drugs, free small molecule drugs, and small molecule drug-related metabolites, LC-MS/MS-based methods are mostly used.
Anti-therapeutic antibody (ATA) analysis
Like other biological products, ADC may also cause immunogenicity after entering the organism and produce anti-therapeutic antibodies (ATA), affecting its safety, PK, and efficacy.
ATA has a variety of research platforms, including bridging ELISA, electrochemiluminescence (ECL), radio immunoprecipitation (RIP), surface plasmon resonance (SPR), etc.
Reference
- Basa, L. (2013). Drug-to-antibody ratio (DAR) and drug load distribution by LC-ESI-MS. In Antibody-Drug Conjugates (pp. 285-293). Humana Press, Totowa, NJ.






